The coronavirus disease (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), continues to spread across the globe. To date, there have been more than 95 million reported cases of SARS-CoV-2 infection worldwide. Over the past few months, new variants of SARS-CoV-2 have emerged that are more virulent.
One new SARS-CoV-2 variant of concern, called 202012/01, started to spread in England in late October. A new study published on the pre-print server medRxiv* found that 60 percent of daily COVID-19 cases screened by the Alderley Park Lighthouse Labs were tied to 202012/01, also known as lineage B.1.1.7 or 20B/501Y.V1. This suggests a rapid spread of the new variant across the country.
The new variant
On December 14, 2020, health authorities in the United Kingdom and Northern Ireland reported Variant of Concern 202012/01 to the World Health Organization (WHO) after identifying it through viral genomic sequencing. The new variant has subsequently been identified in other countries
While 202012/01 is considered fast-spreading and easily transmissible, it does not cause more severe symptoms.
Scientists fear that newly developed SARS-CoV-2 vaccines, some of which are already in use, may not protect against 202012/01.
The variant 202012/01 contains 14 mutations that lead to amino acid changes and three deletions. One of the mutations, called N501Y, alters an amino acid within the six key residues in the receptor-binding domain (RBD), making it bind more tightly to the cellular receptors. This means it can more easily infect people.
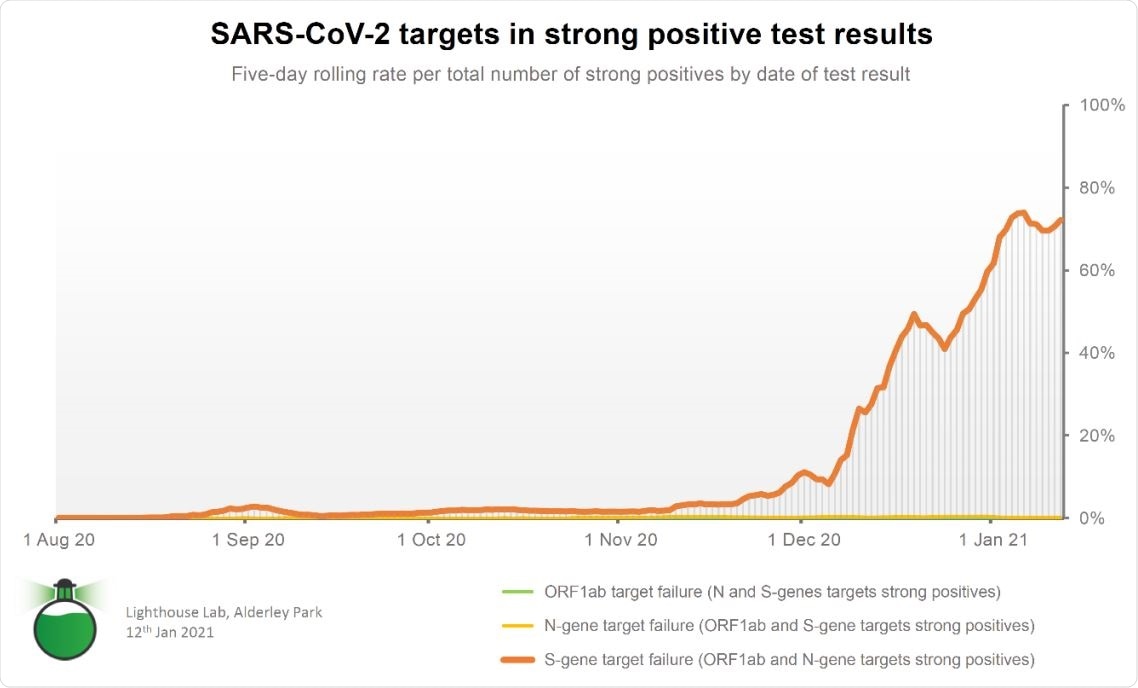
TaqPath™ COVID 19 Assay, S-gene detection in comparison to ORF1ab and N-gene.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
In the United Kingdom alone, more than 1,100 cases of the new variant were detected by December 13, 2020. To date, 202012/01 has been reported in at least 31 countries, including Australia, Denmark, Canada, Philippines, France, Italy, the Netherlands, Hong Kong, Japan, Singapore, and Nigeria, among others.
The study
The study, spearheaded by scientists at the Medicines Discovery Catapult, Lighthouse Labs, Alderley Park in the United Kingdom, reported that by the end of December 2020, 60 percent of daily positive test results in the laboratory were tied to the new variant under investigation.
The Alderley Park (AP) Lighthouse Lab (LHL) tests for the presence of the RNA of SARS-CoV-2 by using the ThermoFisher TagPath COVID-19 test for real-time reverse transcription-polymerase chain reaction (RT-qPCR) detection of SARS-CoV-2 in nasal and throat swabs.
When the team collated and studied the tests they processed over the past months, the new variant's impact on diagnostic testing increased rapidly in December 2020, rising to over 70 percent of strong positive test results detected at the beginning of January 2021.
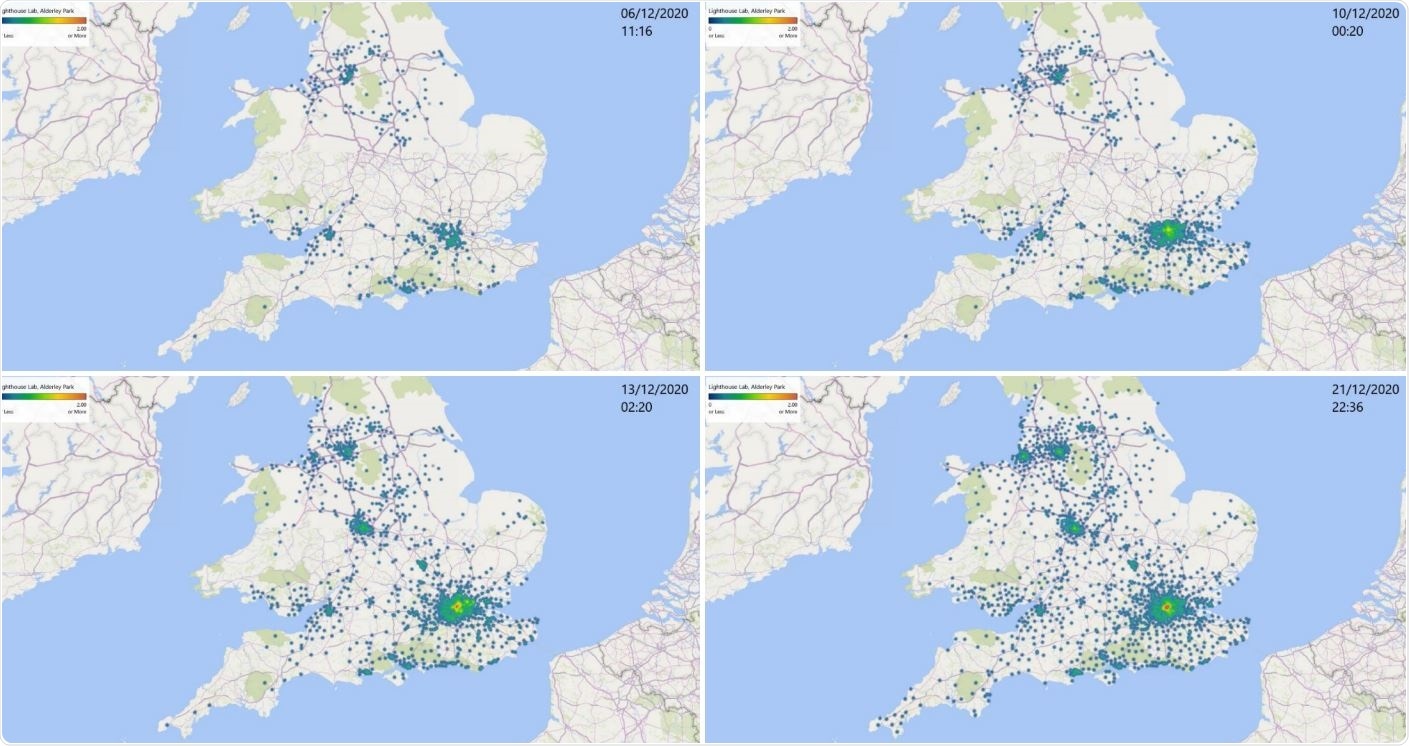
Map of S-gene detection failure, associated with the new SARS-CoV-2 variant. Data collected at Alderley Park, Lighthouse Labs, from 1st to 21st of December 2020.
The laboratory also noted that Spike protein mutations, usually seen in the VOC 202012/01, were present in the S-gene target detection failure cases. The failure of S-gene target detection is linked to the emergence of the new variant of concern. Further, some regions where the new variant has been detected are illustrated by the absence of S gene detection in the TagPath assay used in the study. This matches with areas that are now reporting skyrocketing levels of patient hospitalization.
"Ongoing molecular monitoring will be needed to ensure that no further new S gene variants are co-emerging with VOC 202012/01," the researchers note in the paper.
Fast-spreading variant
The current emergence and spread of the new SARS-CoV-2 variant highlight the need for strengthened infection control measures. Some countries have imposed lockdowns to prevent viral spread. Countries with surging COVID-19 cases related to the new variant should also boost compliance with health protocols, including regular handwashing, social distancing, and avoiding crowded places.
To date, there are more than 2 million deaths tied to the pandemic worldwide. The United States reports the highest number of cases, reaching over 24 million.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Source:
Journal references:
- Preliminary scientific report.
Gravagnuolo, A.M., Faqih, L., Cronshaw, C., et al. (2021). Epidemiological Investigation of New SARS-CoV-2 Variant of Concern 202012/01 in England. medRxiv. https://www.medrxiv.org/content/10.1101/2021.01.14.21249386v1
- Peer reviewed and published scientific report.
Gravagnuolo, Alfredo Maria, Layla Faqih, Cara Cronshaw, Jacquelyn Wynn, Paul Klapper, and Mark Wigglesworth. 2021. “High Throughput Diagnostics and Dynamic Risk Assessment of SARS-CoV-2 Variants of Concern.” EBioMedicine 70 (August): 103540. https://doi.org/10.1016/j.ebiom.2021.103540. https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(21)00333-9/fulltext.