We are neuroscientists, so it was a bit of a shift in direction! For several years TJ and I had been testing out how to use nanobodies to make brain imaging better. When the pandemic broke, we thought this is a once in a lifetime, all-hands-deck situation and joined the fight.
The labs were all closed down in April, and we thought we would much rather work on COVID-19 rather than staying at home.
SARS-CoV-2 virus binding to ACE2 receptor. Image Credit: Kateryna Kon/Shutterstock.com
What are ‘nanobodies’ and how do these differ from regular antibodies?
Nanobodies are derived from a special type of antibodies naturally produced by the immune systems of camelids, i.e. camels, llamas, and alpacas. Most antibodies are made from 4 proteins bound together: two heavy chains and two light chains.
Camelids make special antibodies that are made from just 2 proteins: 2 heavy chains. Nanobodies are created in the laboratory by isolating just the tips of the heavy chains, where the binding occurs. On average, these nanobody proteins are about a tenth the weight of a typical human antibody.
Because nanobodies are more stable, less expensive to produce, and easier to engineer than typical antibodies a growing body of researchers, including our group, have been using them for medical research. For instance, a few years ago scientists showed that humanized nanobodies may be more effective at treating an autoimmune form of thrombotic thrombocytopenic purpura, a rare blotting clotting disorder, than current therapies.
One of the exciting things about nanobodies is that, unlike most regular antibodies, they can be aerosolized and inhaled to coat the lungs and airways. We think this could be especially beneficial for preventing COVID-19 transmission through the air.
Can you describe how you carried out your research into COVID-19 antibodies?
We immunized a llama, named Cormac, five times over 28 days with a purified version of the SARS-CoV-2 spike protein. Then we isolated the DNA that includes the instructions for making antibodies from Cormac’s blood. (By the way, Cormac lives on a beautiful farm in Eastern Washington state with quite a few other llamas. He has a catheter in one of his veins so that people can draw his blood without sticking him with any needles).
Back in the lab here in Bethesda, we tested out hundreds of nanobodies and found that Cormac produced 13 nanobodies that might be strong candidates. We selected nanobodies that could block the attachment of the SARS-CoV2 spike protein to the human ACE2 receptor.
The virus gets inside of the human body by binding to the ACE2 receptor on the outer surface of human cells, so blocking this attachment was a logical target.
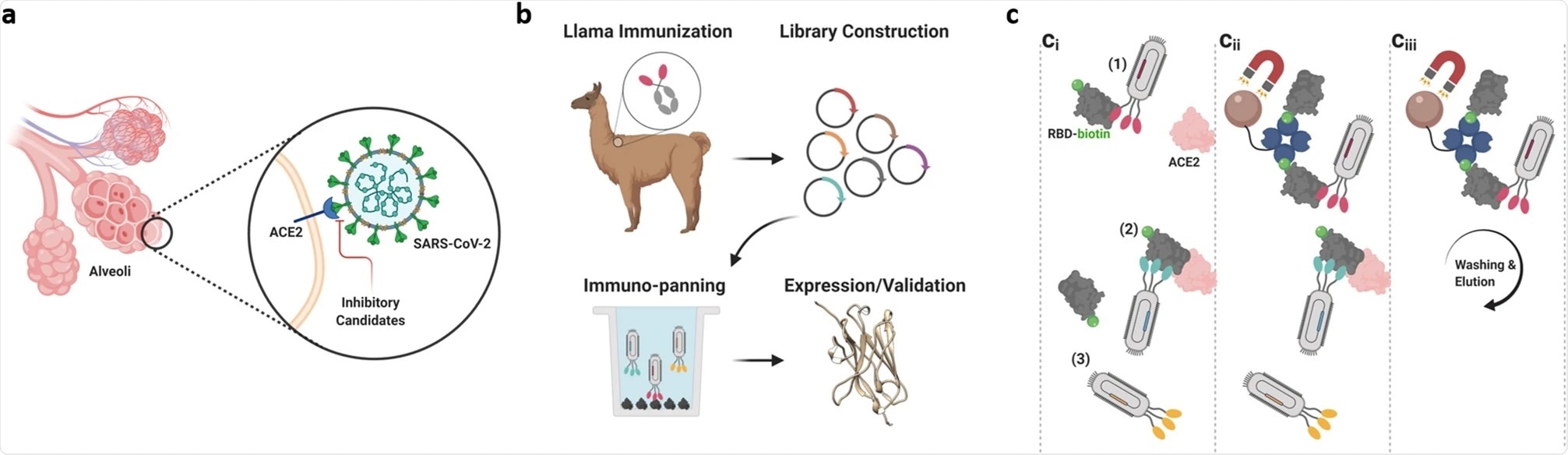
Image Credit: High affinity nanobodies block SARS-CoV-2 spike receptor binding domain interaction with human angiotensin converting enzyme
What did you discover?
We discovered that one candidate nanobody, called NIH-CoVnb-112 bound very tightly to the part of the SARS-CoV2 spike protein responsible for interacting with the ACE2 receptor.
We next showed that the NIH-CoVnB-112 nanobody could be effective at preventing infections in a petri dish. To mimic the COVID-19 virus, our collaborators in North Carolina genetically mutated a harmless “pseudovirus’ so that it can use the SARS-CoV-2 spike protein to infect cells that produce the human ACE2 receptor. They found that relatively low levels of the NIH-CoVnb-112 nanobodies prevented the pseudovirus from infecting these cells in Petri dishes.
Importantly, as a team, we showed that the nanobody was equally effective in preventing the infections in Petri dishes after being sprayed through the sort of nebulizer that patients might use.
How effective were these nanobodies at combating coronavirus?
We do not know how effective the nanobodies will be in combating coronavirus. There is still a lot of work to do before we will have the answers.
What are the advantages of using nanobodies compared to typical antibodies?
There are a lot of advantages of nanobodies.
- Nanobodies are very stable. We can keep them at room temperature for a long time, they resist extreme heat and cold, and they can be dried down into a powder for storage then reconstituted with water when needed. It is much trickier to handle and store typical antibodies.
- Nanobodies are less expensive to produce. They can be made in bacteria or yeast, which is much easier than the mammalian cells used to produce regular antibodies.
- Nanobodies are easier to engineer than typical antibodies. They are smaller and made from just 1 protein instead of 2.
- Nanobodies can be aerosolized and inhaled to coat the lungs and airways. This does not usually work well for typical antibodies.
- Nanobodies do not typically activate the human immune system. This could be an advantage or a disadvantage depending on whether we want to activate the immune system or not.
Nanobody. Image Credit: StudioMolekuul/Shutterstock.com
Do you believe that with further research, these nanobodies could potentially be used to combat the coronavirus pandemic?
We think that with further research, the nanobodies could potentially be used to combat the coronavirus pandemic in several ways:
- People who are going into a high-risk situation could use a nebulizer at home to coat their airways and lungs with a protective layer of nanobodies.
- People who have been exposed to someone with COVID-19 could start treating themselves at home to reduce the chances that they will get sick
- People who have COVID-19 could coat their airways and lungs with nanobodies to reduce the transmission of the active virus to others around them. A virus that has nanobodies stuck to it would be expected to be less infectious than a naked virus.
- The nanobodies could be used to spray environmental surfaces, providing a coat that will neutralize the active virus that lands on the surfaces.
- The nanobodies could be used for environmental sensors to detect the virus in the atmosphere, in fluids, or on surfaces.
- The nanobodies could be used to make very inexpensive test kits that are stable at extreme temperatures.
What are the next steps in your research?
There are several important next steps that are underway:
- Testing the nanobodies in animal models of COVID-19 infection.
- Scaling up production to make enough high-quality nanobodies for larger studies
- Testing to see whether the nanobodies will block the new variants of the virus.
Where can readers find more information?
We strongly encourage everyone to read the original paper. It is important for the public to understand the data and be well-educated on this topic.
For this reason, we have published it in a journal called Scientific Reports that is available to everyone in the world with internet access and we have written the paper in a way that we hope is relatively easy to understand.
Here is the link:
https://www.nature.com/articles/s41598-020-79036-0
Several other groups around the world are also working on nanobodies.
Here is a link to a news story from last fall:
https://www.nature.com/articles/d41586-020-02965-3
One company in China called Shanghai Novamab has already scaled-up production of its nanobody.
https://www.researchgate.net/publication/343580508_A_potent_neutralizing_nanobody_against_SARS-CoV-2_with_inhaled_delivery_potential
About Dr. David Brody
David L. Brody, MD, Ph.D., is the Director of the Center for Neuroscience and Regenerative Medicine (CNRM) and a Professor of Neurology within the Uniformed Services University of the Health Sciences (USU) in Bethesda, Maryland. He is a board-certified neurologist with both a research and a clinical specialization in traumatic brain injury (TBI) and neurodegenerative diseases. His research focuses on accelerating better outcomes for U.S. military TBI patients.
Prior to his current position, Dr. Brody was the Norman J. Stupp Professor of Neurology at the Washington University School of Medicine in St. Louis, Missouri. Dr. Brody was also the Washington University site director for the National Football League's Neurological Care Foundation. He has developed and authenticated advanced imaging technologies to detect injury in the brain's white matter and showed, for the first time, how to predict neurological function by measuring amyloid, an abnormal protein in the brain. He also helped discover that diffusion tensor imaging, an advanced magnetic resonance imaging technique, can reveal blast-related damage.
Dr. Brody previously led a team that worked in partnership with U.S. Department of Defense (DoD) researchers at the Landstuhl Regional Medical Center in Germany and at two sites in Afghanistan, treating U.S. military personnel with TBIs.
In 2011, he served as a consultant to the medical advisor of the Chairman of the Joint Chiefs of Staff. He traveled to Afghanistan at the request of then-JCS Admiral Michael Mullen with the "Gray Team", a group of civilian and military experts evaluating the status of TBIs in troops within the combat zone. His achievements have been recognized with several awards, including a Career Development Award from the National Institute of Neurological Disorders and Stroke, and a Burroughs Wellcome Career Award in the Biomedical Sciences.
Dr. Brody is a member of the Editorial Board of the Journal of Neurotrauma and Acta Neuropathologica and a permanent member of the National Institute of Health Acute Neural Injury and Epilepsy Study Section. His clinical monograph entitled "Concussion Care Manual: A Practical Guide" was published by Oxford University Press in 2014.
Dr. Brody earned a B.A. in Biological Sciences from Stanford University and his MD and Ph.D. from the Johns Hopkins University School of Medicine. He completed his neurology residency at Washington University in St. Louis.