A team of international scientists has recently investigated the efficacy of PVP-1, a complex of polyvinylpyrrolidone and iodine, in preventing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Using in vitro cell culture settings, the scientists observe that PVP-1 formulation used in a commercially available nasal spray (Nasodine) readily inactivates SARS-CoV-2 within a short period of time. The study is currently available on the bioRxiv* preprint server.

 *Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
Background
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the causative pathogen of coronavirus disease 2019 (COVID-19), primarily spread from person to person via large respiratory droplets. However, there is evidence suggesting that airborne transmission of the virus is also possible. The virus is known to replicate more efficiently in the upper respiratory tract than that in the lower respiratory tract. Two host cell proteins, namely angiotensin-converting enzyme-2 (ACE2) and transmembrane protease serine 2 (TMPRSS2), which are required for the viral envelop – host cell membrane fusion and viral entry, are highly expressed in the nasal and alveolar epithelial cells, supporting the fact of preferential viral replication in the upper respiratory tract.
Because viral shedding from the nasal cavity and upper respiratory tract is the primary mode of infection transmission, early therapeutic elimination of SARS-CoV-2 in the nasal cavity can significantly reduce the risk of person-to-person transmission as well as propagation of the infection to the lower respiratory tract and other organs.
In the current study, the scientists have examined the efficacy of PVP-1 in eliminating SARS-CoV-2 infection by conducting a series of in vitro experiments. PVP-1 is known to have a broad range of antimicrobial effects and is routinely used as a skin and mucous membrane disinfectant. In this study, the scientists have specifically evaluated the efficacy of commercially available Nasodine antiseptic nasal spray, which contains a clinically approved formulation of 0.5% of PVP-1.
Study design
Of two SARS-CoV-2 isolates used in the study, one was incubated with Nasodine for 1 minute. The other one was incubated with either Nasodine or 0.5% PVP-1 in saline for 15 seconds, 5 minutes, or 15 minutes. Both PCR-based and cell culture-based methods were used to measure the residual viral titers and determine the efficacy of PVP-1 formulation.
Important observations
By conducting the cell culture-based assay, the scientists observed that Nasodine can completely inhibit the replication of SARS-CoV-2. In contrast, a robust replication was observed for SARS-CoV-2 treated with control media (without Nasodine).
For further confirmation, the scientists used a cytopathic effect (CEP)-based assay to determine the effect of Nasodine on a different SARS-CoV-2 strain. They considered two timepoints (15 seconds and 5 minutes) as clinically relevant for nasal use, because the activity of PVP-1-based nasal formulation gradually declines with time due to the inactivation of iodine by nasal proteins.
The assay findings revealed that Nasodine significantly reduces SARS-CoV-2 replication within 15 seconds of exposure. Moreover, no viable virus was detected after 15 minutes of treatment. Importantly, Nasodine formulation showed comparatively better efficacy than the PVP-1 preparation in saline in terms of reducing viral titers. The efficacy of Nasodine formulation was higher at both clinically-relevant timepoints (15 seconds and 5 minutes).
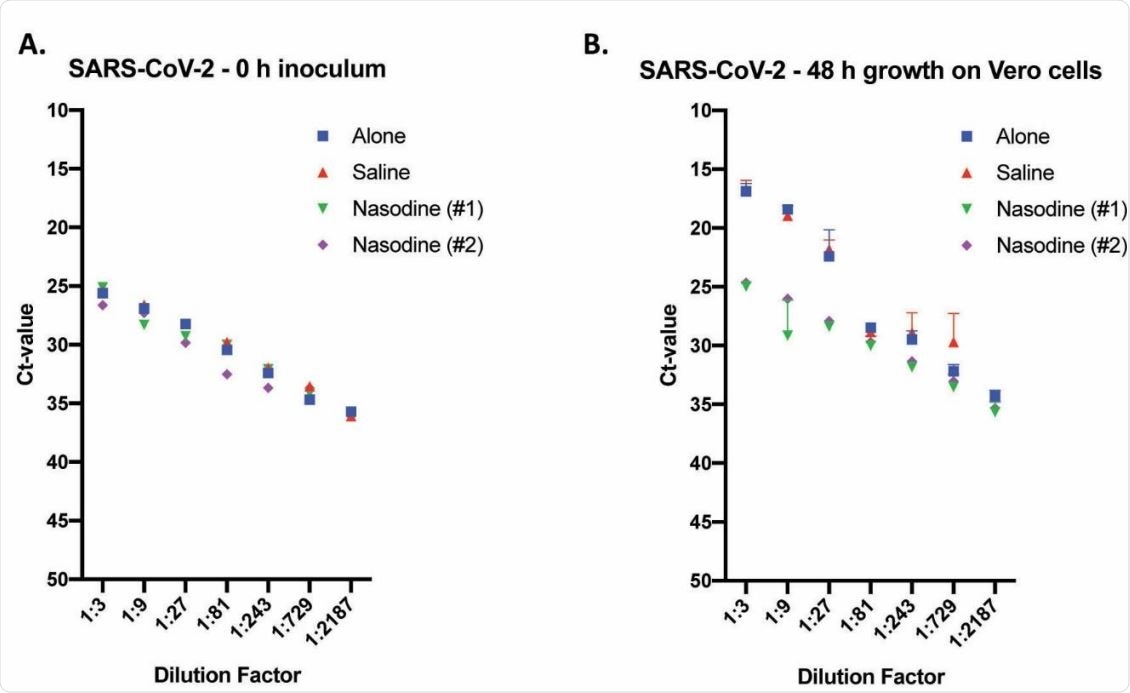
Titre of Nasodine and control-treated SARS-CoV-2 via TCID50 assay and RNA detection via real-time TaqMan RT-PCR. SARS-CoV-2 was exposed to the indicated test solution(s) for 1 minute before serial dilution (1:3) and incubation on Vero cells for either 0 (zero) or 96 h. Values expressed as mean cycle threshold (Ct) value + SEM (n=3) versus dilution factor. (A) Time point zero (0 h) inoculum titration used to determine baseline Ct-values of treated samples prior to replication in Vero cells. (B) Titres associated with cultures harvested 96 h post inoculation of Vero cells.
Study significance
The study reveals that Nasodine, a clinically-approved nasal spray formulation with 0.5% PVP-1, has significant antiviral activity against SARS-CoV-2, and can be potentially used for early eradication of SARS-CoV-2 in the nasal cavity. To better manage the COVID-19 pandemic, Nasodine can be potentially used along with other control measures to prevent the viral transmission.
The scientists are currently conducting a clinical trial to check if these in vitro effects are equally relevant for COVID-19 patients. They believe that Nasodine could be an effective complementary intervention for newly diagnosed COVID-19 patients in terms of reducing disease progression and viral shedding.

 *Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.