New research shows further evidence between COVID-19 infection and the development of long-term neurological symptoms. The findings found COVID-induced neuroinflammation and microvascular injury contributed to dementia-like symptoms. However, they also found evidence indicating a low possibility of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) directly invading the brain.
Some people with COVID-19 infection experience persistent symptoms — even when the virus has been cleared from the body. A prominent 'long-COVID' symptom is brain fog, where people have a hard time thinking and concentrating. The current findings could further enhance scientists understanding of the neurological symptoms associated with long-COVID.
The study "Network medicine links SARS-CoV-2/COVID-19 infection to brain microvascular injury and neuroinflammation in dementia-like cognitive impairment" is available as a preprint on the bioRxiv* server, while the article undergoes peer review.
.jpg)
Network-based multimodal omics analytic methodology

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Designing the study
The researchers used a protein-protein interaction assay and CRISPR-Cas9 genetic results to construct a network-based, multimodal SARS-CoV-2 virus-host interactome. They then investigated COVID-19-associated cognitive impairments by evaluating transcriptomic profiles using single-cell and nuclei RNA sequencing for Alzheimer's disease marker genes in people with COVID-19 infection. Areas vulnerable to SARS-CoV-2 entry in patients with Alzheimer's disease without previous infection were also explored.
The team hypothesized that the neurological complications originate from the localization of the SARS-CoV-2 host cell's genes and proteins in the human protein-to-protein interaction (PPI) network. And they may be targeting genes and proteins most vulnerable to disease or indirectly through PPI.
Altered expression of protein and gene associated with Alzheimer's
Based on the results of PPI assays and CRISPR-Cas9 genetic assays, the researchers found SARS-CoV-2 host proteins and genes strongly affected the expression of proteins and genes associated with Alzheimer's. The altered proteins included RAB7A, TGFB1, and VCAM1.
Low chance SARS-CoV-2 directly invades the brain
The team also looked at whether SARS-CoV-2 directly affects the brain. They observed the expression of SARS-CoV-2 entry in the tissues, brain regions, and several brain cells.
There was a low expression of ACE2 and TMPRSS2 in both brains and neurons. In contrast, BSG, NRP1, and FURIN increased their expression in endothelial cells in the prefrontal cortex in both Alzheimer's brains and healthy controls.
"Our results suggest that it is unlikely for SARS-CoV-2 to target neurons directly via ACE2. However, we cannot rule out the possibility that SARS-CoV-2 may enter the brain through the cerebral endothelium using receptors such as BSG and NRP1 or other unknown entry factors," wrote the researchers.
Inflammation in the brain found in patients with COVID-19 infection
Given that neuroinflammation is a common symptom of Alzheimer's disease, the researchers sought to find if this also occurred in patients with SARS-CoV-2. When measuring markers in the blood and cerebrospinal fluid of patients with COVID-19 infection, they found genetic changes — in NKTR, GSTM3, TGFB1, TNFRSF1B, SPP1, and CXCL10 — that have been linked to Alzheimer's. Endothelial cells also had increased expression of the following antiviral defense genes: LY6E, IFITM2, IFITM3, and IFNAR1. The STAT3 and JAK1 genes seem to play a role in neuroinflammation, which is consistent with previous studies implicating the JAK-STAT signaling pathway in exacerbating COVID-19 symptoms.
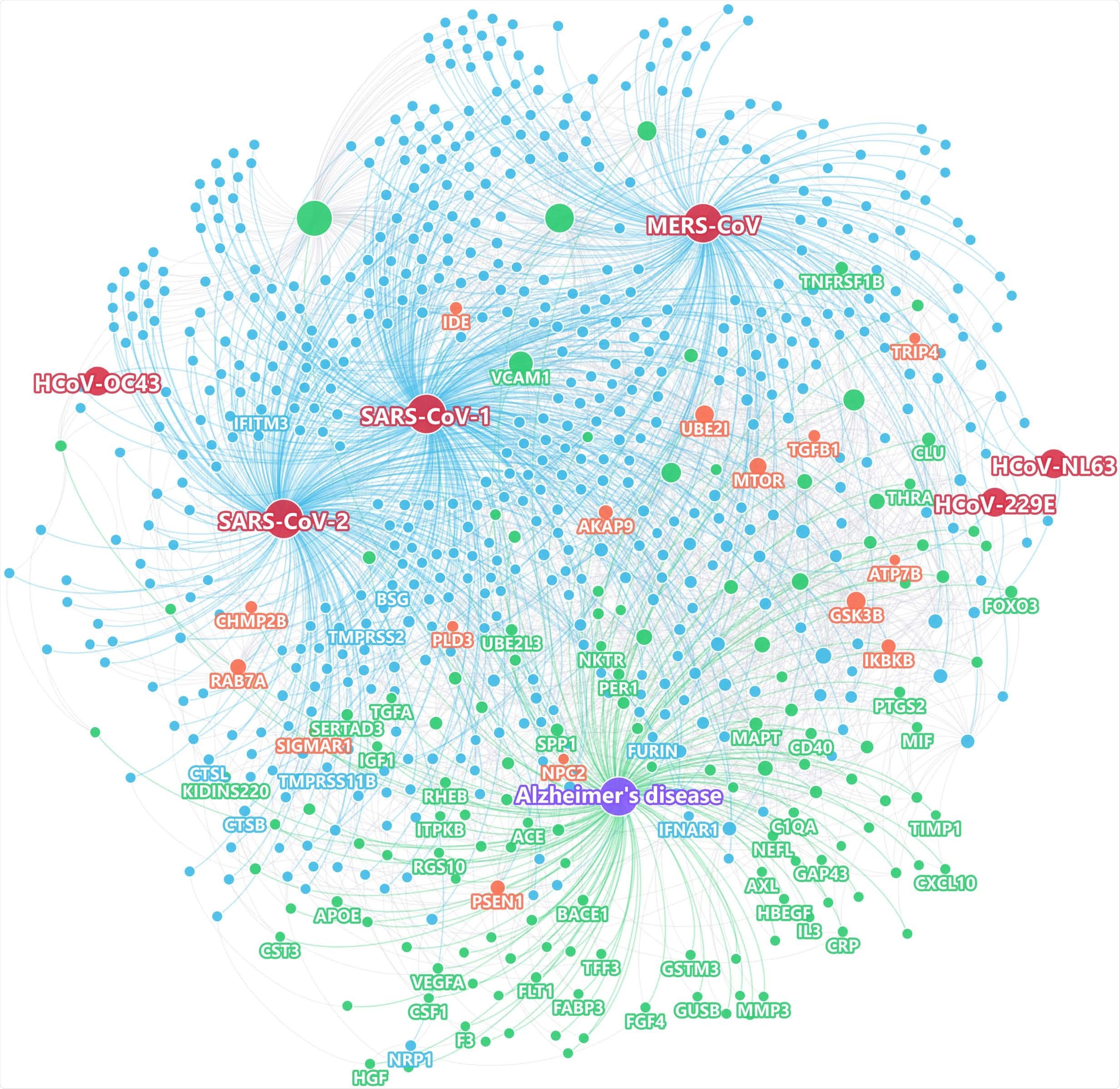
Using state-of-the-art network proximity measures to evaluate the network-based relationship for the gene/protein sets between virus-host factors and each disease/condition under the human interactome network model.
People with APOE E4/E4 show a lower expression of antiviral defense genes than people with the APOE E3/E3 gene, indicating a lack of gene expression and an elevated risk for COVID-19 illness. The researchers suggest more studies looking at the relationship between APOE gene expression and the likelihood of developing SARS-CoV-2 infection.
Study limitations to consider
Despite using high-quality data from multiple sources, the researchers acknowledge their human protein-protein interactome is incomplete, indicating a possibility that network associations could be either positive or negative. Also, the researchers found low expression of ACE2 and TMPRSS2 but could not rule out the chance of another key SARS-CoV-2 entry factor. Another limitation was that the current study did not assess possible neuroinflammation pathways in patients with both Alzheimer's and COVID-19, which could have affected the findings.
"The significance of our findings in the context of the general population of COVID-19 frequently suffering from "brain fog" without a formal diagnosis of AD needs further investigation," concluded the research team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Zhou Y, et al. Network medicine links SARS-CoV-2/COVID-19 infection to brain microvascular injury and neuroinflammation in dementia-like cognitive impairment. bioRxiv, 2021. doi: https://doi.org/10.1101/2021.03.15.435423, https://www.biorxiv.org/content/10.1101/2021.03.15.435423v1
- Peer reviewed and published scientific report.
Zhou, Yadi, Jielin Xu, Yuan Hou, James B. Leverenz, Asha Kallianpur, Reena Mehra, Yunlong Liu, et al. 2021. “Network Medicine Links SARS-CoV-2/COVID-19 Infection to Brain Microvascular Injury and Neuroinflammation in Dementia-like Cognitive Impairment.” Alzheimer’s Research & Therapy 13 (1). https://doi.org/10.1186/s13195-021-00850-3. https://alzres.biomedcentral.com/articles/10.1186/s13195-021-00850-3.