Recently, a group of nanoengineers at the University of California San Diego discuss their development of cell membrane-based nanoparticles that successfully delivered dexamethasone directly to inflamed lung cells in a murine model of inflammation.
Schematic of genetically engineered cell membrane-coated nanoparticles for drug delivery to inflamed lungs. Image Credit: Zhang Lab
An overview of inflammation
The immune system induces a wide variety of both chemical and physiological changes in response to inflammation. For example, proinflammatory cytokines such as interleukin-6 (IL-6) and tumor necrosis factor (TNF) can be released during inflammation to improve the immune response at a site of stress or injury. Some of the different effects that these cytokines have on the local vasculature include vasodilation and increased vascular permeability to promote more efficient recruitment of immune cells to the affected site.
In addition to the macroscopic changes that arise as a result of these proinflammatory cytokines, several cellular level changes also occur. To this end, the release of cytokines can lead to the upregulation of specific surface markers including vascular cell adhesion molecule-1 (VCAM-1) or intracellular adhesion molecule-1 (ICAM-1). Both VCAM-1 and ICAM-1 promote the adhesion of immune cells at the site of inflammation.
Nanoparticle-based platforms to tackle inflammation
Despite the utility of inflammation for survival, a dysregulated immune system is often associated with various diseases ranging from cancer to cardiovascular diseases. To manage a wide range of diseases that are associated with inflammation, several nanoparticle-based platforms have been developed. The use of nanoparticles to deliver anti-inflammatory agents to the vasculature by specifically targeting cellular adhesion molecules like VCAM-1 and ICAM-1 has emerged as an appealing approach to treating a wide range of diseases affected by inflammation.
More recently, researchers have developed cell membrane coating technology that utilizes virtually any type of cell membrane to coat nanoparticles. The utility of this approach is that the final nanoformulations can be custom-tailored to specific applications while simultaneously increasing their functionality as drug delivery systems.
Cell membrane-based dexamethasone nanoparticles
In a recent study published in Science Advances, a group of nanoengineers at the University of California San Diego discuss their development of immune cell-mimicking nanoparticles that specifically target lung inflammation for drug delivery purposes.
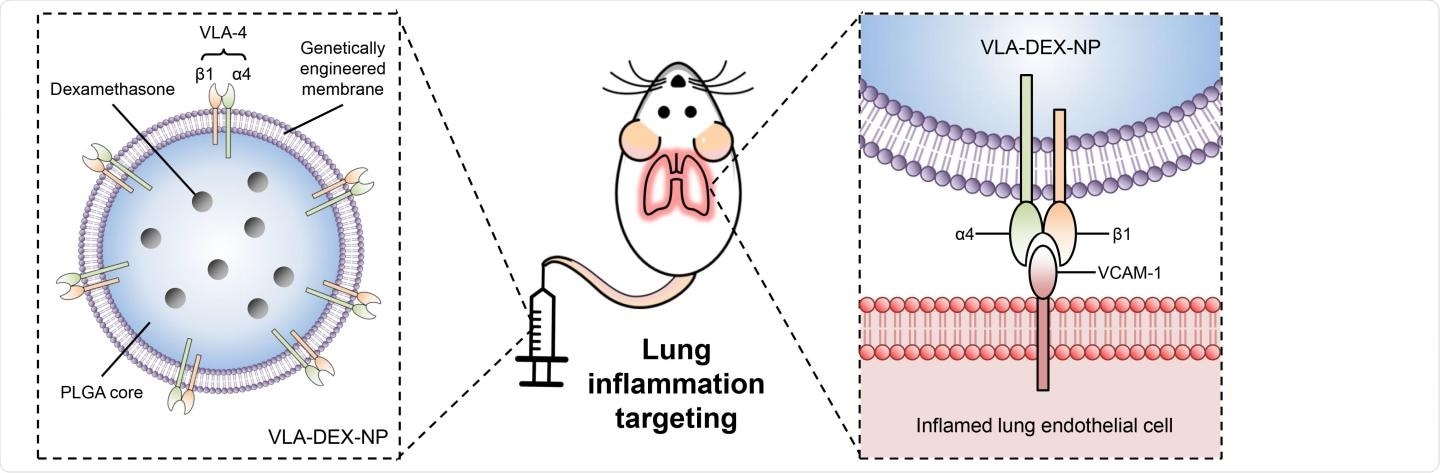
In their work, the researchers genetically modified a cell line to express very late antigen-4 (VLA-4), which is a cognate ligand of VCAM-1. To do this, the researchers packaged VLA-4 genes into a viral vector, which was then inserted into lab-grown host cells that originated from mice. The cells incorporate the genes from the viral vector into their own genome, which results in the production of cell membranes that constantly overexpress VLA-4.
The engineered membrane was then used to coat polymeric nanoparticles that were loaded with dexamethasone (DEX), which is a potent anti-inflammatory drug. The efficacy of these nanoparticles was then studied in vivo using a murine model of bacterial lipopolysaccharide (LPS)-induced lung inflammation.
The overexpression of VLA-4 on the nanoparticles allows for them to seek out VCAM-1, which is often overexpressed on vascular endothelial cells during inflammation. Although the nanoparticles themselves do not directly enhance the efficacy of DEX, their unique ability to bring this anti-inflammatory drug directly to the site of inflammation allows for smaller doses to be used. Ultimately, the researchers found that the cell membrane-coated nanoparticles successfully delivered DEX to the site of interest at high levels and more rapidly as compared to when standard drug delivery approaches were used.
We’re delivering the exact same drug used in the clinic, but the difference is we’re concentrating the drugs to the point of interest. By having these nanoparticles target the inflammation site, it means a larger portion of the medicine will wind up where it is needed, and not be cleared out by the body before it can accumulate and be effective.”
Journal reference:
- Park, J. H., Jiang, Y., Zhou, J., et al. (2021). Genetically engineered cell membrane-coated nanoparticles for targeted delivery of dexamethasone to inflamed lungs. Science Advances 7(25); eabf7820. doi:10.1126/sciadv.abf7820.