Vaccines against coronavirus disease 2019 (COVID-19) have been granted emergency use approval by various regulatory bodies around the world since December 2020. Further, COVID-19 vaccines continue to be administered on mass scales to the elderly and the general public.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Background
COVID-19 vaccines have shown sufficient effectiveness and protection from most variants of the severe acute respiratory distress syndrome coronavirus (SARS-CoV-2), particularly in reducing severe outcomes and hospitalization rates in infected individuals. However, there is limited data on the variant-specific efficacy among immunocompromised or those living with life-threatening co-morbidities.
Patients on maintenance dialysis are one type of vulnerable population who are at an increased risk of infection, severe disease, hospitalization, and even death due to COVID-19. Previous studies on vaccine efficacy among patients dependent on maintenance dialysis indicate that two doses of a SARS-CoV-2 messenger ribonucleic acid (mRNA) vaccine elicit a sero-response in almost 90% of dialysis patients, though the level of antibody titers is lower than those in the general population.
The SARS-CoV-2 Delta variant has increased the transmissibility of this virus in the United States since its original introduction into this country in June 2020. Rising concerns about the Delta variant emerged after reports demonstrated the increased virulence of the variant, as well the increased rate of breakthrough infections in fully vaccinated patients.
Multiple studies have reported a reduction of protection offered by the Pfizer-BioNTech BNT162b2 vaccine from 93% at baseline to 53% after at least four months. Further, there are reports of 10-15% of dialysis patients not responding to two doses of a messenger ribonucleic acid (mRNA) vaccine. It is also well established that the protection offered by vaccines wanes in about half of the population of those responding to treatments by four-six months, particularly those whose initial response was less.
In a recent study published on the preprint server medRxiv*, researchers assess the impact of lesser initial vaccine response and subsequent waning antibody levels on clinical outcomes among maintenance dialysis.
About the study
In the current study, the researchers collected details on unvaccinated, partially vaccinated, and fully vaccinated adult dialysis patients treated by a national dialysis provider within the U.S. between February 1, 2021, and October 2, 2021. Their aim was to evaluate the incidence of COVID-19 diagnoses, severe COVID-19-related hospitalization, or death among these patients in both the pre-Delta and Delta dominant periods.
The researchers also analyzed the subset of vaccinated patients with available data on immunoglobulin G (IgG) anti-spike antibody titers post-vaccination who were diagnosed with ‘breakthrough’ SARS-CoV-2 infections to determine the association between antibody levels and clinical outcomes.
Study findings
The researchers conducted a retrospective, observational study with data from Dialysis Clinic, Incorporated (DCI). DCI is the largest non-profit provider of kidney care and dialysis care in the United States, with 260 operating outpatient dialysis clinics in 29 states, 20 chronic kidney disease clinics in five states, and 3 organ procurement organizations in 3 states.
A total of 15,718 patients receiving dialysis during the study period were analyzed. Among these individuals, 71% (11,191) were fully vaccinated, 5% (733) were partially vaccinated, and 24% (3,794) were unvaccinated.
Researchers identified 967 COVID-19 cases in this cohort, out of which 511 (53%) occurred among the unvaccinated patients and 579 (60%) occurred during the Delta dominant period. COVID-19-related hospitalization or mortality was less likely among vaccinated patients in comparison to unvaccinated patients for all vaccines. The adjusted hazard ratio (HR) for individual vaccines including the Pfizer-BioNTech BNT162b2, Moderna mRNA-1273, and Johnson & Johnson Ad26.COV2.S vaccines were 0.25, 0.14, and 0.34 respectively.
While analyzing the correlation between anti-spike IgG levels and clinical outcomes, those with IgG levels greater than or equal to seven had a significantly lower risk of a COVID-19 diagnosis. None of these individuals experienced a COVID-related hospitalization or death.
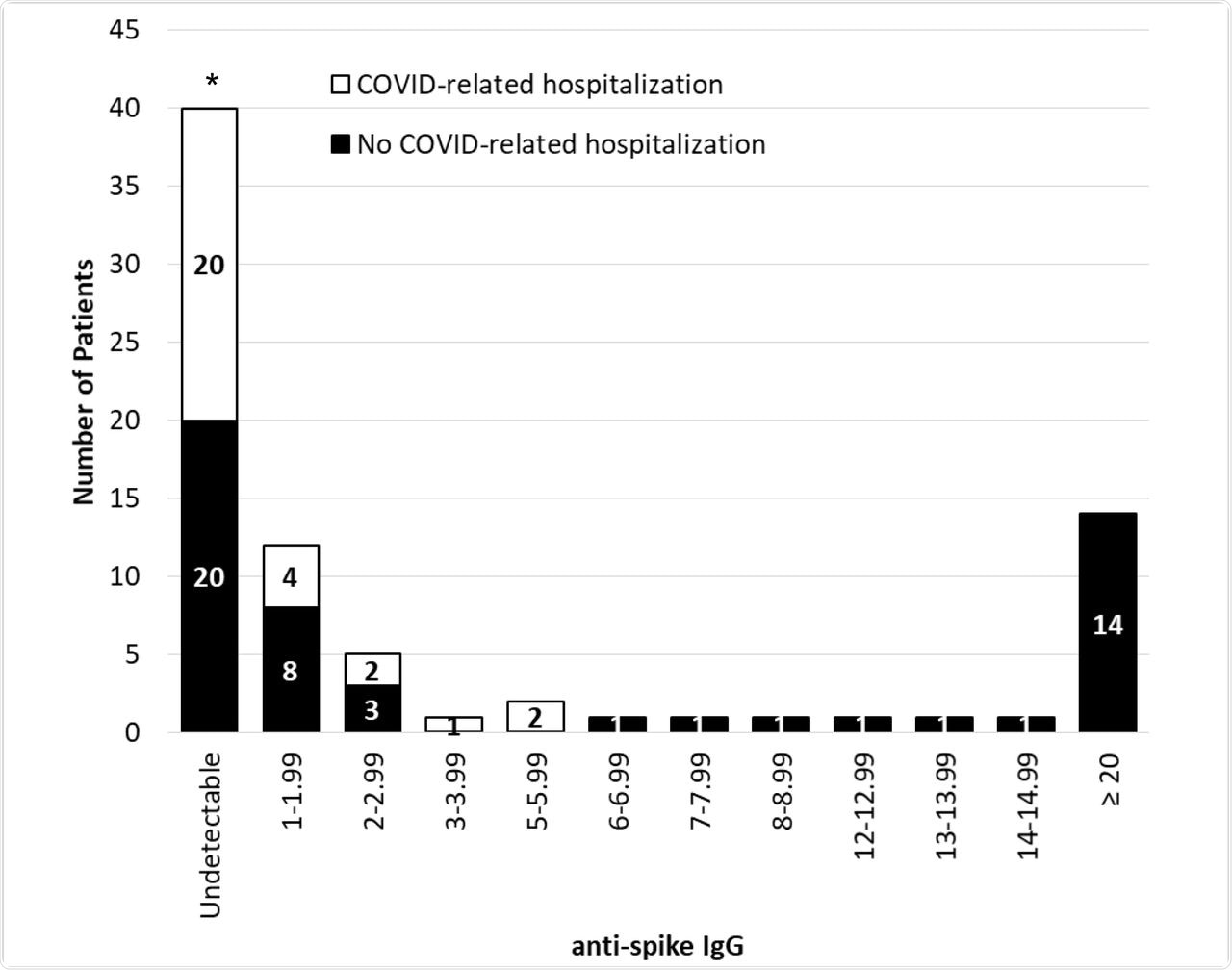
Patient anti-spike IgG value at the time of COVID infection diagnosis (N=80).
Implications
The current study was crucial in highlighting the importance of vaccinations among those with severe co-morbidities. For patients who were on maintenance dialysis patients, SARS-CoV-2 vaccination was associated with a lower risk of COVID-19 diagnosis, severe disease, hospitalization, and even death. Among vaccinated patients, low anti-spike IgG levels were associated with worse COVID-19 related outcomes.
The study results can be used to encourage vaccination among those still reluctant to undergo COVID-19 vaccination. The results of this study also add evidence for booster vaccination doses to maintain adequate immunity in the community.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Manley, H. J., Aweh, G., Hsu, C. M., et al. (2021) SARS-CoV-2 vaccine effectiveness and breakthrough infections in maintenance dialysis patients. medRxiv. doi:10.1101/2021.12.20.21268124. https://www.medrxiv.org/content/10.1101/2021.12.20.21268124v1.
- Peer reviewed and published scientific report.
Manley, Harold J., Nien Chen Li, Gideon N. Aweh, Caroline M. Hsu, Daniel E. Weiner, Dana Miskulin, Antonia M. Harford, Doug Johnson, and Eduardo Lacson. 2023. “SARS-CoV-2 Vaccine Effectiveness and Breakthrough Infections among Patients Receiving Maintenance Dialysis.” American Journal of Kidney Diseases 81 (4): 406–15. https://doi.org/10.1053/j.ajkd.2022.10.010. https://www.ajkd.org/article/S0272-6386(22)01051-4/fulltext.