From Alzheimer’s risk to cardiovascular prediction, this review shows how protein profiling and AI could help detect disease earlier, guide treatment more precisely, and move medicine closer to truly personalized care.
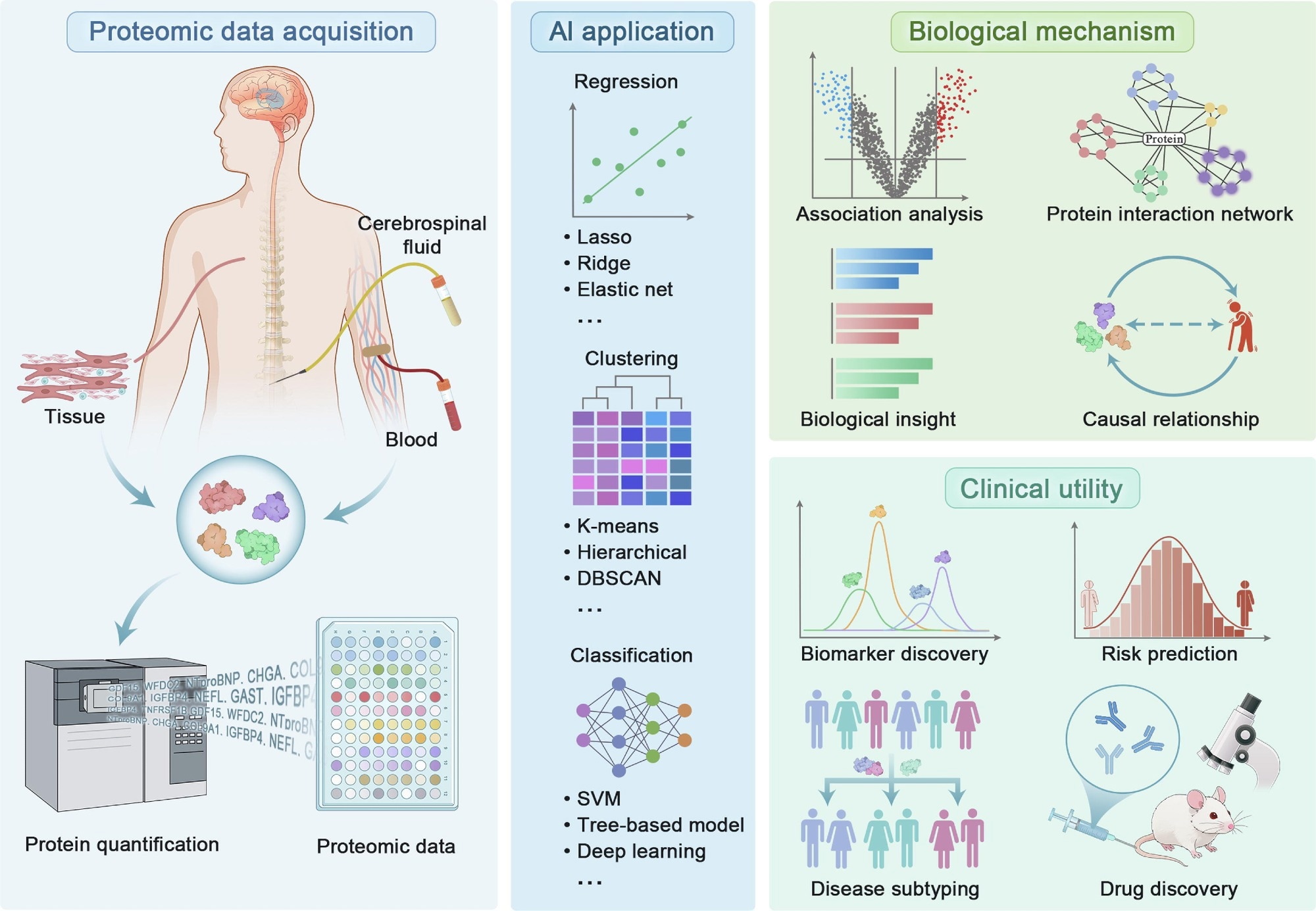
Proteomics analytical pipeline in precision medicine. Review: The emerging role of high-throughput proteomics in advancing precision medicine
In a recent review published in the journal Science Bulletin, a group of Chinese researchers explored how high-throughput proteomics and artificial intelligence (AI) are advancing biomarker discovery, disease prediction, and personalized treatment strategies in precision medicine.
Proteomics Background and Biomarker Discovery
What if diseases like Alzheimer’s or heart failure could be detected decades before symptoms appear? There has been a growing focus in medicine on earlier detection, prevention, and more individualized care, particularly through precision medicine approaches tailored to individual biological variability.
While genomics has been an important part of medicine, it does not capture biological changes occurring in real time. Proteomics provides a dynamic view of disease-related biology by studying proteins, the functional molecules that link genes to physiology. Recent technological developments enable the simultaneous analysis of thousands of proteins, helping to better understand human health and disease.
These research areas are important given the rise in chronic diseases and the aging population. However, further research is needed to develop standardized processes to use these approaches in daily clinical care.
Proteins act as the functional drivers of biological processes, linking genetic information to real-world physiological outcomes. Genomics only provides static information, but proteomics tracks changing biological activity, making it crucial for spotting early signs of disease. High-throughput technology can measure thousands of proteins in a single sample, improving diagnostic tools and enabling faster, more accurate disease detection.
Different biological samples provide unique insights. Blood-based proteomics enables large-scale population studies, while cerebrospinal fluid (CSF) provides more precise information for neurological diseases such as Alzheimer’s and Parkinson’s. Other sources, including urine and tissue samples, provide disease-specific insights, though they may be limited by variability, technical constraints, or invasiveness.
Proteomic Disease Prediction and Risk Models
One of the most impactful applications of proteomics is its ability to predict disease risk. Traditional tools, such as polygenic risk scores (PRS), estimate genetic susceptibility but cannot account for environmental and lifestyle factors. Proteomics bridges this gap by reflecting real-time biological states shaped by both genetics and external factors.
Proteomic models show strong predictive potential and, in some studies, outperform conventional risk scores for cardiovascular disease. Studies show that protein-based models can identify high-risk patients several years before the onset of the disease. Likewise, in the case of patients with neurodegenerative diseases, proteomic profiles can identify individuals at risk of developing dementia, or those who could develop symptoms of Parkinson’s disease, up to a decade or more prior to a formal diagnosis, helping support earlier risk stratification and intervention.
Proteomics also supports multi-disease prediction. Large-scale studies have shown that smaller panels of proteins can reliably predict multiple disease processes simultaneously, including metabolic, cardiovascular, and neurodegenerative diseases, as well as multiple chronic conditions.
Another emerging concept is the “biological aging clock.” By analyzing protein patterns, researchers can estimate biological age, which may differ from chronological age. Individuals with accelerated biological aging are at higher risk of diseases and mortality, highlighting the potential of proteomics in preventive healthcare.
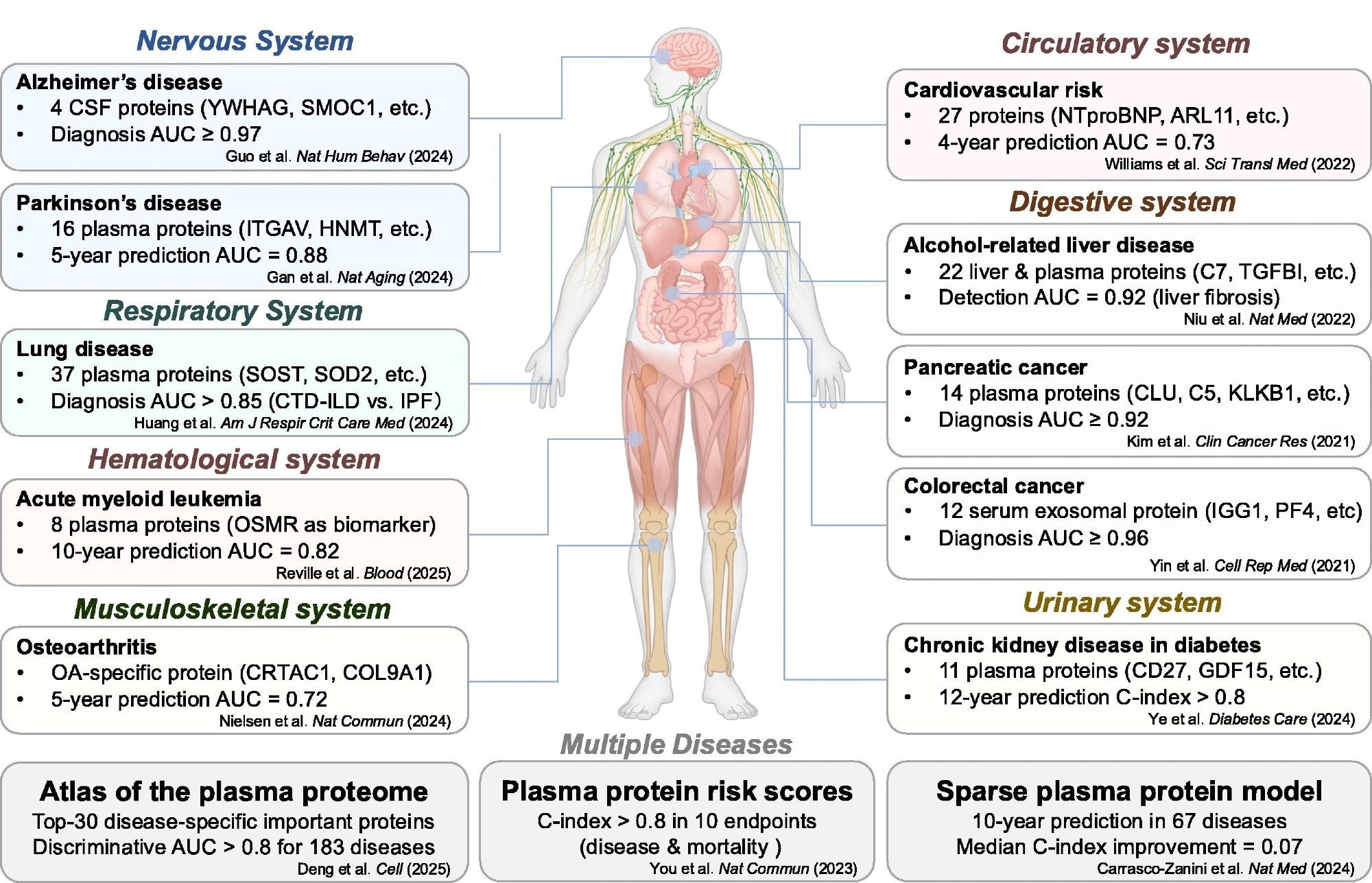
Proteomics advancing biomarker discovery and disease risk prediction. (Created with BioRender.com). AUC: area under the ROC curve; CSF: cerebrospinal fluid; CTD-ILD: connective tissue disease-associated interstitial lung disease; IPF: idiopathic pulmonary fibrosis; OA: osteoarthritis.
Proteomics in Drug Discovery and AI Analysis
Proteomics is changing drug discovery by enabling better therapeutic target identification, complementing rather than simply replacing the slow, trial-and-error process seen in parts of traditional drug development. Mass spectrometry helps researchers see how drugs affect biological systems, and methods such as activity-based protein profiling (ABPP) and thermal proteome profiling (TPP) improve drug safety and effectiveness by identifying both targets and side effects.
In cancer treatment, proteomics may help guide personalized therapy, which is particularly important for heterogeneous diseases like metastatic cancers, where treatment responses vary widely. By tailoring therapies based on protein profiles, clinicians can potentially improve outcomes and reduce unnecessary treatments. It also allows the repurposing of existing drugs, which can be crucial in urgent healthcare challenges, such as emerging diseases or drug resistance.
The vast amount of data generated by proteomics requires advanced analytical tools. AI is essential for extracting meaningful insights from complex datasets, using techniques such as least absolute shrinkage and selection operator (LASSO), recursive feature elimination (RFE), and support vector machines (SVMs) to identify key biomarkers and build predictive models. AI helps to create protein panels that improve accuracy and detect diseases early by spotting patterns.
AlphaFold is another deep learning method being utilized to improve proteomics by predicting the structure of proteins. By predicting protein structures, researchers can better understand how proteins function and interact at the molecular level.
In addition to its ability to predict protein structure, AI can also provide an integrated view of proteomics, combined with other data, including genomics and clinical records, to give a comprehensive picture of a patient's overall health.
By providing a complete picture of a patient's health, this integrated view could support the development of personalized treatment plans and improve clinical decision-making.
Proteomics Challenges and Clinical Future
Sample preparation variability, the absence of standardized protocols, and variations in analytical methods can affect data reliability. High costs and technical complexity also limit widespread clinical adoption.
Statistical challenges, such as overfitting in machine learning models and data heterogeneity, and regulatory barriers, slow the translation of proteomic medicine research into clinical practice, as a standardized validation method is currently lacking.
Future successful use of proteomics will depend upon addressing these issues by developing standardized workflow processes, improving the ability to share data, strengthening external validation across diverse cohorts, accounting for differences between proteomic platforms, and integrating proteomics with multi-omics techniques.
Conclusion
High-throughput proteomics is helping to shape precision medicine by enabling earlier disease detection, accurate risk prediction, and personalized treatment strategies. By capturing dynamic biological changes, proteomics provides insights that go beyond traditional genetic approaches.
Integrating AI can lead to more effective identification of biomarkers and therapeutic targets, with potential benefits for both patients and health systems. However, challenges related to standardization, validation, and clinical implementation remain, and addressing these barriers will likely make proteomics a key element in modern healthcare, leading to improved outcomes and proactive disease management.
Journal reference:
- You, I., Deng, Y.-T., Chen, L., Wang, L.-B., Guo, Y., Li, Z.-Y., Feng, J.-F., Yu, J.-T., & Cheng, W. (2026). The emerging role of high-throughput proteomics in advancing precision medicine. Science Bulletin. 71(7). 1798–1811. DOI: 10.1016/j.scib.2026.02.054 https://www.sciencedirect.com/science/article/pii/S2095927326002112