Sponsored Content by AbselionReviewed by Olivia FrostApr 2 2026
His-tagged proteins are central to assay development, structural biology, and drug discovery projects. Their small tag facilitates rapid construct iteration and straightforward purification.
Teams require quick, quantitative readouts from crude lysates when screening many constructs, enabling decisions on what to scale and what to drop. Everyday tools such as spectrophotometry and SDS-PAGE are essential, but can be ambiguity at the crude stage.
Teams can capture reproducible concentration measurements directly from unpurified samples with a His-tag assay on the Amperia™ platform. In doing so, expression and purification decisions can be made at an earlier stage.
Fig 1. Amperia provides quantitative data in expression screening. Image Credit: Abselion
Direct measurements also allows:
- Decoupling of bottlenecks
- Early readouts, resulting in better decisions
- Distinguishing expression limits from purification issues
- Earlier ranking of constructs due to quantification in crude lysates
- Guiding purification by confirming trends and detecting incomplete elution
Image Credit: Abselion
About the assay
The Amperia™ His-tag assay uses a premix competition format in which His-tagged proteins in the sample compete with a labeled His-tagged detection reagent for binding sites.
Using an inverse signal relationship in this way enables accurate quantification across a wide dynamic range, even with crude lysates.
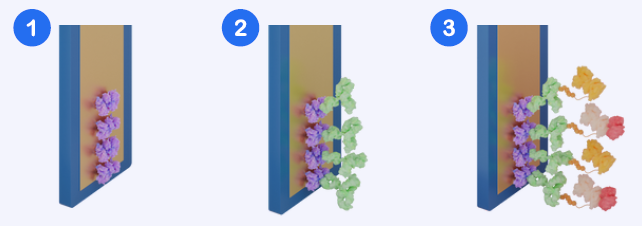
Fig 2. Assay Workflow Schematic. (1) Sensor with streptavidin surface. (2) Biotinylated anti-His antibody bound to the sensor. (3) Premixed sample and HRP-labelled His-tagged detection reagent applied, enabling signal generation. Image Credit: Abselion
Note: In the commercial kit, steps 1 & 2 are completed during manufacturing.
Project context
A total of eight His-tagged protein constructs were expressed in E. coli and assessed on the Amperia™ platform as part of an evaluation with a global pharmaceutical company.
Each construct carried an 8-His tag. Cells were lysed, crude lysates were directly analyzed, and purified fractions were prepared to enable comparison. In this instance, the target protein had an approximate molecular weight of ∼30 kDa.
Results
Screening crude lysates
The eight constructs (S1-S8) were expressed and analyzed as crude lysates. Several samples (S1-S6) produced strong but overlapping bands on SDS-PAGE (Figure 3, top left), while other samples (S7, S8) demonstrated no visible band near the 30 kDa target.
It was difficult to interpret relative yields, but Amperia™ delivered a clear ranking in a single run (Figure 3, top right, blue).
S4 was observed to express at the highest level (∼2230 µg/mL), followed by S5 (∼670 µg/mL) and S1 (∼400 µg/mL). S2 and S6 were noted to give only low signals, while the other constructs demonstrated little or no expression.
Analyzing purified fractions
Samples were purified using spin columns (200 µL crude → two 200 µL elutions). S1, S4, and S5 showed clear bands at ∼30 kDa on SDS-PAGE (Figure 3, bottom), with S4 being the strongest. Other constructs, however, produced no distinct band.
Amperia™ verified an identical ranking in the first elution (Figure 3, top right, red), measuring ∼750 µg/mL for S4, ∼150 µg/mL for S5, and ∼120 µg/mL for S1. Other constructs were either at or near the baseline.
Concentrations in the first elution were found to be lower than those measured in crude lysates. Additional protein was visible in the second elution by gel (Figure 3, bottom). This was determined to be consistent with incomplete recovery from the resin.
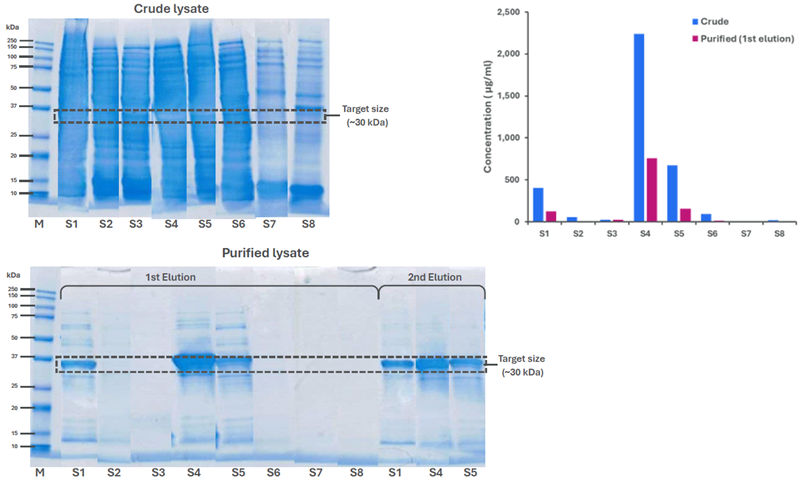
Fig 3. Crude lysates and purified fractions were analyzed by SDS-PAGE and Amperia™. Amperia™ provided quantitative ranking in crude and 1st elution samples, while SDS-PAGE also showed protein in the 2nd elution. Image Credit: Abselion
Workflow insights
Amperia provided the team with a clear construct ranking in crude lysates (S4 > S5 > S1), whereas the gels were more ambiguous. The same order was also confirmed following purification, though at lower concentrations. However, gels also showed protein in the second elution.
This combination of results demonstrates Amperia’s capacity to provide quantitative insight into His-tag protein expression, facilitating more informed downstream decisions.
Acknowledgments
Produced from materials originally authored by Abselion.
About Abselion
Abselion started in 2018, at that time under the name HexagonFab, in a small corner of a laboratory at the University of Cambridge.
It set out with the humble goal to make protein research simpler. Scientists should be able to pursue their passion for discovery and innovation, rather than spend their valuable time on tedious, manual tasks. With RED, Abselion had access to the ideal technology to create this product. A product that is so compact that it could fit on every bench, and so affordable that it is accessible to everyone. Over the years, Abselion has designed, built, and tested its first product, Amperia™, and is proud to introduce it to the world.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.