A landmark NEJM outbreak study shows how Andes virus can move from rodents to people and then through close human contact, offering urgent context for the MV Hondius cruise ship cluster now under WHO investigation.

Image Credit: Dedi rinaldi08 / Shutterstock
In a 2020 study published in The New England Journal of Medicine, researchers analyzed clinical and sociodemographic data linked to a significant outbreak of Andes virus (ANDV) hantavirus pulmonary syndrome in Epuyén, Argentina, between late 2018 and early 2019. The findings are newly relevant following the recent MV Hondius cruise ship cluster, in which the WHO reported 11 hantavirus cases, including three deaths, with current evidence suggesting onboard human-to-human transmission of ANDV.
The 202 study combined next-generation (“next-gen”) sequencing technologies and stochastic modeling to identify the specific viral strain and epidemiological route that allowed the outbreak to infect 34 individuals and claim 11 lives. Study findings revealed that a single zoonotic introduction led to four generations of human infection. Transmission was shown to be primarily driven by three symptomatic "super-spreaders" during social gatherings.
Study data further revealed that transmission risk was associated with higher viral titers and markers of liver injury or impaired hepatic function, rather than viral adaptation, suggesting that social contact patterns and host clinical factors appeared to have greater influence than viral genetic change in person-to-person spread in this typically zoonotic pathogen.
Andes Virus Transmission Background
Hantavirus pulmonary syndrome is an acute zoonotic disease caused by viruses in the genus Orthohantavirus. In South America, the Andes virus (ANDV) is particularly notorious due to its high case-fatality rate (21% to 50%).
While primary hantavirus infection typically occurs via human inhalation of aerosolized excreta from the long-tailed colilargo rodent (Oligoryzomys longicaudatus), ANDV has uniquely demonstrated a capacity for person-to-person transmission since its first documentation in 1996.
However, despite this more than 2-decade-old precedent, epidemiological reviews revealed that sustained human transmission chains remained extremely rare and therefore poorly understood until the 2018–2019 Epuyén outbreak. The more recent MV Hondius cluster has renewed attention on this risk in enclosed, high-contact settings. The Epuyén outbreak provided researchers with an unexpected opportunity to investigate whether the virus was evolving to better infect humans or if extrinsic factors, such as host-pathogen interactions and social dynamics, were driving the pathogen’s spread.
MV Hondius Cruise Cluster Context
On May 13, 2026, the WHO reported that the MV Hondius cluster involved 11 cases, including three deaths, for a case fatality ratio of 27%. Eight cases were laboratory-confirmed as ANDV infection, two were probable, and one remained inconclusive pending further testing. WHO’s working hypothesis was that the first case acquired infection before boarding through land-based exposure, while current evidence suggested subsequent human-to-human transmission onboard. Preliminary sequencing revealed closely related, near-identical viral sequences across cases, supporting a likely shared outbreak source or a limited transmission chain.
WHO assessed the public health risk for people onboard the ship as moderate but the global risk as low, noting that close living quarters, shared indoor spaces, prolonged exposure, and frequent interpersonal interactions likely facilitated transmission. For the general public, including those not exposed on board or through close contact with a confirmed case, the WHO considered the risk of infection low.
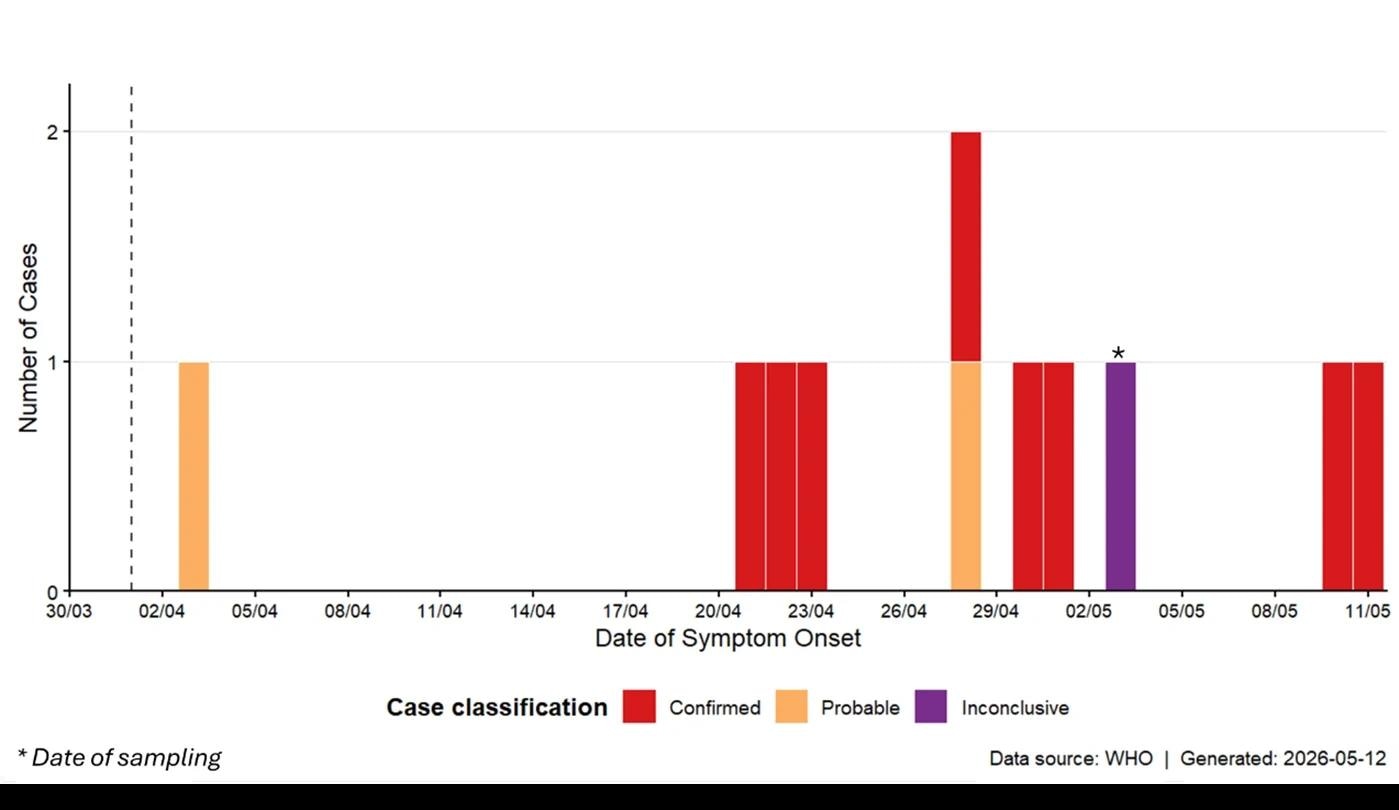
Epidemiological curve of Andes hantavirus cases (n = 11) reported to WHO as of 13 May 2026, 17:00.
Epuyén Hantavirus Outbreak Study Design
The 2020 study aimed to leverage data from the Epuyén outbreak to better understand hantavirus, specifically ANDV, epidemiology, and inform future public health containment strategies by analyzing data from 34 confirmed cases across Argentina and one linked case in Chile.
The study's data collection included participants’ sociodemographic and geographic data, temporal data, medical history data, and biological samples for viral sequencing and biomarker monitoring.
The study employed next-generation sequencing to map complete or nearly complete viral genomes from 27 patients, thereby enabling the assessment of viral diversity and possible adaptation during person-to-person transmission. Interview-based contact tracing approaches were used to identify routes of viral spread.
Statistical analyses specifically computed: 1. The reproductive number (R), the average number of secondary cases per infected individual, and 2. Individual reproductive numbers (Z), the number of people infected by a single individual, are used herein to identify "super-spreaders.”
In parallel, the study monitored biological markers in the patients’ blood, for example, viral load and cytokine levels. Specifically, reverse-transcriptase quantitative polymerase chain reaction (RT-qPCR) assays were used to detect viral RNA, which was then compared with clinical outcomes, for example, hepatic injury markers and respiratory distress, across different patient groups.
Super-Spreading and Viral Load Findings
Study outcomes identified the outbreak strain as the Epuyén/18-19 strain and traced the outbreak's origin to a single rodent-to-human spillover event involving the index patient. The outbreak's primary transmission event was shown to be a birthday party attended by approximately 100 guests.
Genomic analysis revealed high viral stability, and patient-derived sequences showed 99.8% to 100% identity, with no evidence of host-mediated adaptation. Transmission dynamics evaluations revealed that R = 2.12 before control measures were implemented and decreased to 0.96 following the government's implementation of isolation and quarantine measures.
The study further identified three individuals who acted as super-spreaders (Z > 4) and accounted for 64% of secondary cases (21 of 33). Notably, patient-specific Z values were found to have a positive correlation with patients’ alanine aminotransferase (ALT; r = 0.51) and lactate dehydrogenase (LDH; r = 0.54 before control measures). However, overall disease severity itself had no clear association with secondary transmission.
Epidemiological data showed that, among patients assessed before isolation, higher viral loads were associated with a greater likelihood of transmission (odds ratio [OR] = 1.7). The case fatality rate was found to be 32% (11 of 34) with incubation periods ranging from 9 to 40 days.
Finally, spreaders exhibited significantly higher levels of Interleukin-1β (IL-1β; OR = 2.4) and lower levels of Stem Cell Growth Factor β (SCGF-β; OR = 0.27) than non-spreaders.
Andes Virus Public Health Implications
The 2020 NEJM article confirms that the Andes virus can sustain person-to-person transmission chains in the absence of genetic adaptation, provided that social and ecological factors allow for super-spreading events. The data suggest that the Epuyén/18–19 strain appeared to carry baseline genetic traits compatible with person-to-person transmission, without requiring newly detected adaptation during the outbreak.
Notably, the successful reduction of the R value below 1.0 following enforced isolation underscores the efficacy of rapid public health interventions in mitigating zoonotic outbreaks. This is particularly relevant to the current MV Hondius response, in which WHO has emphasized contact tracing, quarantining high-risk contacts, rapid case isolation, infection prevention, and supportive clinical management. WHO recommended active monitoring and home or facility quarantine of high-risk contacts for 42 days after last exposure, while low-risk contacts were advised to self-monitor and seek medical evaluation if symptoms occurred.
Because no specific antiviral treatment is approved for HPS, WHO emphasized early recognition, prompt isolation, transfer to an emergency or intensive care unit when needed, and supportive care focused on careful fluid management, hemodynamic monitoring, and respiratory support. Future research must continue to focus on the biological risk assessment of orthohantaviruses to prevent similar spills from escalating into community or enclosed-setting outbreaks.
Download your PDF copy by clicking here.
Source:
Journal reference: