Introduction:
Quantifying amino acids from DBS using optical detection typically requires complex mixtures of buffers and/or ion-pair reagents, along with lengthy analytical run times. This study demonstrates the utility of the RUO Kairos Amino Acid Kit for analyzing 42 amino acids in DBS using LC-MS-based technologies.
For comprehensive instructions on using the Kairos Amino Acid Kit (p/n: 176004379), refer to the Care and Use Manual (p/n: 720006448), which details sample preparation, analytical methodology, and performance characteristics for solvent Calibrators and Quality Controls. This study summarizes the method performance of Waters in-house DBS Quality Controls prepared with the Kairos™ Amino Acid Kit and analyzed using the ACQUITY™ UPLC™ I-Class PLUS System and Xevo™ TQ-S micro Mass Spectrometer.
Waters’ in-house DBS Quality Controls were prepared with the Kairos Amino Acid Kit before being analyzed using the ACQUITY UPLC I-Class PLUS System and Xevo TQ-S micro Mass Spectrometer.
Waters' in-house and external CDC DBS materials were each extracted before being incubated with the Kairos Internal Standard Solution. Extracts were then transferred to a 96-well collection plate that contained borate buffer. These were derivatized using the AccQ•Tag™ Ultra “3X” Derivatizing Reagent.
The Kairos Amino Acid Kit Calibrators were used to inject and quantify a two µL sample extract.
UHPLC-MS/MS analysis was used to quantify a total of 37 amino acids extracted from a single DBS for clinical research. The five remaining amino acids were reported for semi-quantitative purposes only.
Chromatographic separation was completed using an ACQUITY UPLC I-Class PLUS System and a CORTECS™ UPLC C18 Column, with detection then performed using a Xevo TQ-S micro Mass Spectrometer (Figure 1).

Figure 1. The ACQUITY UPLC I-Class PLUS System with Xevo TQ-S micro Mass Spectrometer. Image Credit: Waters Advanced Diagnostics
Experimental
Sample description
Reagent kit
The Kairos Amino Acid Kit (p/n: 176004379) guided by the Care and Use Manual (p/n: 720006448) was used to reconstitute the Kairos Amino Acid Kit Calibrator and Quality Control Sets, Kairos Amino Acid Kit Reagents, Borate Buffer, and AccQ•Tag Ultra “3X” Derivatization Reagent.
The Kairos Internal Standard (IS) was reconstituted. This was performed using 2 mL of 80:20 MeOH:H2O (80:20 methanol/water, v/v), mixed at room temperature for a total of 10 minutes to ensure that all material fully dissolved.
The vial’s contents were then transferred to a volumetric flask and made up to 50 mL with 80:20 MeOH/H2O.
Calibrators and QC sample preparation
Calibrator and QC samples were freshly prepared on the day of analysis.
- Add 75 µL of Kairos IS to a 1.5 mL microfuge tube
- Add 20 µL of Calibrator/QC sample to the previous sample
- Vortex for five seconds
- Leave samples aside until needed (see Step 5 below)
DBS sample preparation
DBS samples were freshly extracted on the day of analysis:
- Punch a 3.2 mm DBS disc into a 350 µL collection plate
- Add 75 µL of Kairos IS to the disc
- Seal the plate with foil adhesive before incubating at room temperature for 20 minutes at 500 rpm
- Add 70 µL of borate buffer containing 0.5 M NaOH to a 350 µL collection plate
- Transfer 10 µL of incubated DBS extract/Calibrator/QCs into borate buffer (note that the DBS sample extracts are translucent and light yellow in the borate solution, while Calibrator and QC extracts are colorless)
- Pipette to mix
- Vortex theAccQ•Tag Ultra “3X” Reagent and add 20 µL to each sample
- Seal the plate with a silicone cap mat
- Allow the plate to rest at room temperature for a total of one minute
- Incubate the sample plate for 10 minutes at 55 °C and 500 rpm
- Inject two µL onto the UHPLC-MS/MS System
Method conditions
The Kairos Amino Acid Kit Care and Use Manual (p/n: 720006448) details the LC Conditions, Gradient Table, MS Conditions, and MRM Parameters.
Data management and processing
Source: Waters Advanced Diagnostics
| |
|
| MS software: |
MassLynx™ v4.2 Software (SCN 1042) |
| Informatics: |
Informatics: TargetLynx™ XS v4.2 Application Manager waters_connect™ Software with QUAN Review Application |
| Data processing: |
TargetLynx Software (User factor of 6.45 must be applied for quantitation of DBS samples only) |
Results and discussion
Kairos Amino Acid Kit performance
No significant carryover from the Kairos Amino Acid Kit Calibrator 6 was observed in subsequent blank samples. This was performed for all amino acids that exhibited a detector response of ≤ 20 % of LOQ.
No functionally significant carryover was noted from in-house-prepared QC High DBS-extracted samples into subsequent Endogenous DBS extracts.
Matrix effects were evaluated on six independent sources of contrived DBS endogenous samples that had been incubated in triplicate using the Kairos IS Solution.
The IS peak area was compared with that of the IS peak area that had not been exposed to the DBS matrix. No significant ion enhancement (< ± 15 % compared to control, % CV < 10 %) was observed, except for histidine.
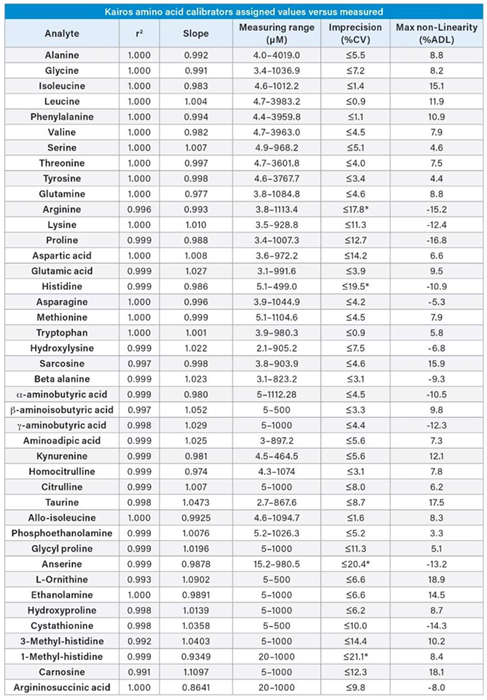
Linearity was evaluated using the Kairos Amino Acid Kit Calibrators on a single day (n=4), with regression analysis showing a linear fit with one/x weighting across the measuring range for the 42 amino acids (Table 4).
The coefficient of determination (R2) for each amino acid was > 0.99, and the percentage imprecision for each calibrator was found to be ≤ 15 %, except at the lower limit of the measuring interval (LLMI), which was ≤ 20 %.
Samples prepared in 0.1 M HCl over the range of 0.5–20 µM were analyzed in order to assess analytical sensitivity. These were evaluated in 10 replicates per level across five analytical runs (Table 3).
The LOQ for the amino acids analyzed was lower than the Calibrator 1 measured values, except for anserine (20 µM), 1-methyl-histidine (10 µM), histidine (9 µM), 3-methyl-histidine (10 µM), and argininosuccinic acid (20 µM).
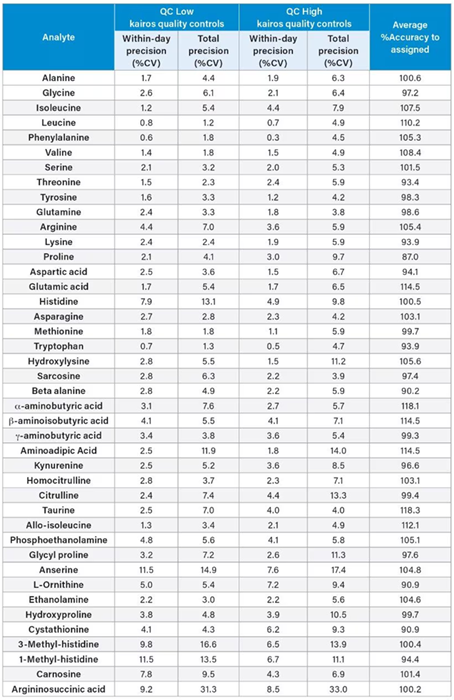
The precision and accuracy of the Kairos Amino Acid Kit QC solvent samples (QC LOW and QC HIGH) were also assessed in five replicates across five analytical runs.
For each analyte, average within-day precision (%CV) and % Accuracy to assigned values were calculated. The Kairos QC within-day precision was found to be ≤ 11.5 % CV and total precision was found to ≤ 14.0 % CV, other than anserine, 3-methyl-histidine, and argininosuccinic acid which were found to exceed the precision goals of ≤ 15 % CV.
The %Accuracy to QC-measured values was within ±15 % of the assigned values (Table 5).
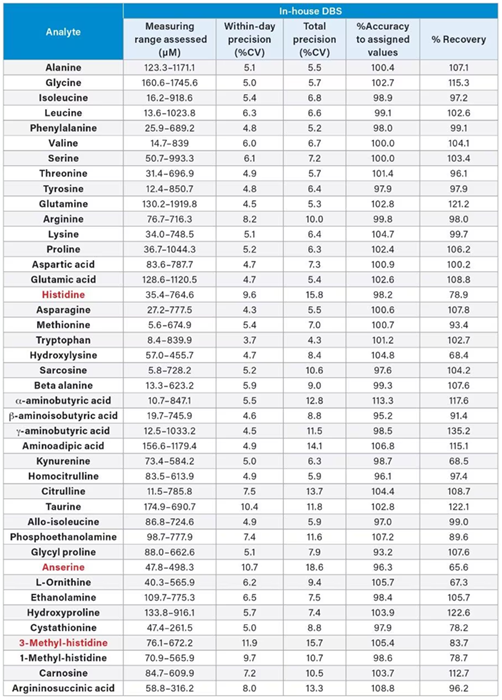
In-house DBS amino acid precision, accuracy, and recovery
In-house DBS QCs were prepared (55 % hematocrit) at five different concentrations (QCs A–E) in order to cover the measuring range of the Kairos Amino Acid Kit.
The in-house DBS QCs were assessed for precision, accuracy, and recovery performance, with five replicates per QC across five analytical runs (n=25).
The average within-day precision and total precision values were ≤ 15 % CV, other than anserine, histidine, and 3-methyl-histidine (total precision ≤ 18.6 % CV). The %Accuracy to assigned values was <15 % for all amino acids.
Recovery was evaluated using QC B-E. This was performed by deducting the endogenous QC-A-calculated concentrations and comparing the results to the spiked amino acid amount. Each amino acid was found to recover within 40-140 %, with the average recovery ranging from 65.5-135.2 % (Table 1).
A single 3.2 mm DBS punch contains around 3.1 µL of blood. A user factor of 6.45 is applied in TargetLynx Software to correct amino acid measurements from extracted DBS. This is required when quantifying using the Kairos Amino Acid Calibrators solvent.
Table 1. Precision, accuracy, and recovery of in-house DBS QC materials using the Kairos Amino Acid Kit. Source: Waters Advanced Diagnostics
External DBS material from the CDC was assessed for precision and accuracy to evaluate method performance against the CDC’s reported values. This involved analyzing five replicates across five analytical runs (n=25).
Within-day precision was found to be ≤ 6.8 %, total precision was found to be ≤ 14.4 %, and %Accuracy to CDC measurement concentrations was within ± 15 % (Table 2).
Table 2. Precision and Accuracy of 12 Amino Acids from CDC QC material using the Kairos Amino Acid Kit. Source: Waters Advanced Diagnostics
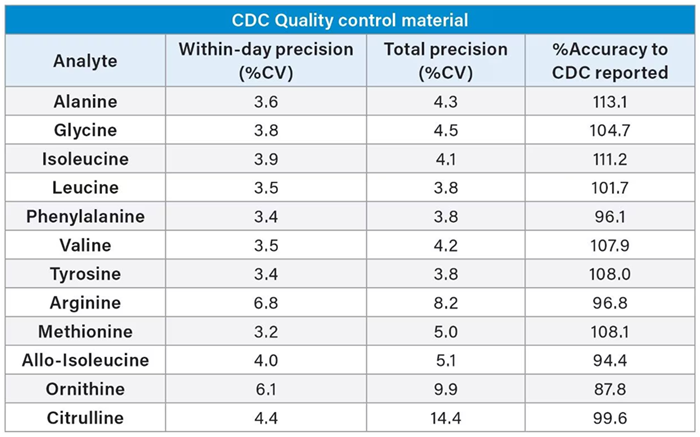
Figure 2 features chromatographic traces of isobaric leucines (leucine, isoleucine, and allo-isoleucine) in the external DBS material covering four QC levels:
- Endogenous (red trace)
- 100 µM (green trace)
- 200 µM (purple trace)
- 400 µM (black trace)
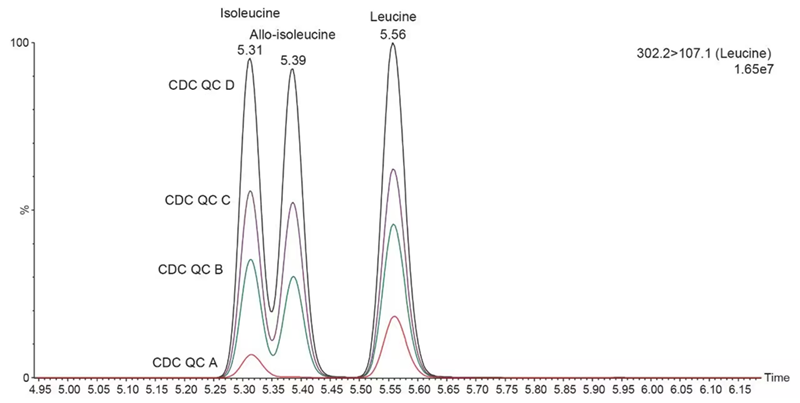
Figure 2. Detection and separation of isobaric Leucines in CDC QC material. Red trace = endogenous DBS; Green, Purple and Black traces represent 100, 200, and 400 µM of Leucines spiked DBS, respectively. Image Credit: Waters Advanced Diagnostics
Figure 3 shows the Total Ion Chromatogram (TIC) for the 12 amino acids detected in the external DBS sample.
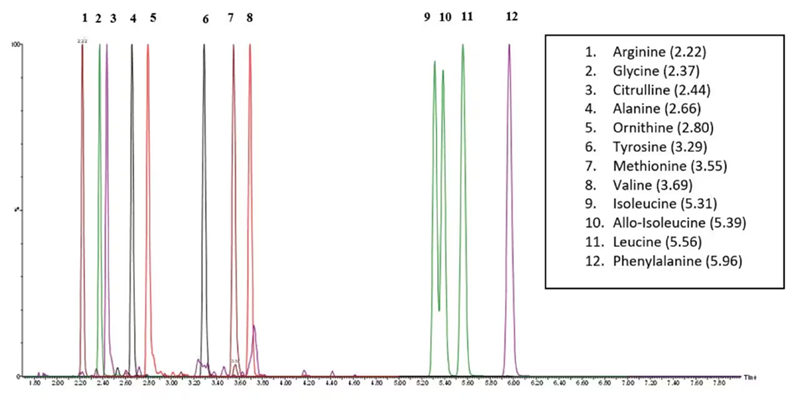
Figure 3. TIC of 12 Amino Acids present in CDC QC Material. Image Credit: Waters Advanced Diagnostics
Conclusion
The Kairos Amino Acid Kit empowers clinical researchers to achieve reliable results with just one flexible kit. The kit exhibits no significant matrix effects or carryover, demonstrating good analytical sensitivity, linearity, precision, and accuracy.
The method outlined here uses the ACQUITY UPLC I-Class System and the Xevo TQ-S micro Mass Spectrometer to quantify 42 amino acids in a single DBS in less than 10 minutes.
The approach removes the need for ion-pair reagents or mobile-phase buffers, while ensuring that derivatized samples remain stable. The method’s chromatographic conditions ensure confident peak identification by effectively separating isobaric amino acids.
The clinical research method outlined good precision, accuracy, and recovery for the range of 37 amino acids evaluated in in-house DBS material. The remaining five analytes (anserine, histidine, 1- and 3-methyl histidine, and argininosuccinic acid) were also included, but they did not meet all method performance criteria.
Table 3. Analytical Sensitivity of the Kairos Amino Acid Kit in solution using the ACQUITY UPLC I-Class PLUS System and Xevo TQ-S micro Mass Spectrometer. Source: Waters Advanced Diagnostics
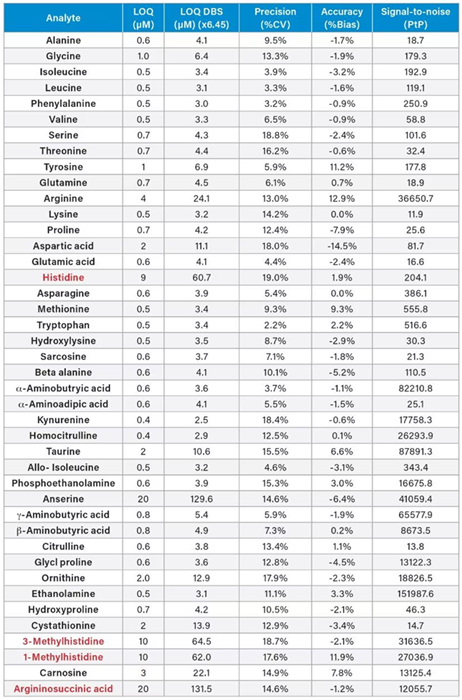
Table 4. Linearity of the Kairos Amino Acid Kit in solution. Source: Waters Advanced Diagnostics
*Denotes maximum %CV at LLMI.
Table 5. Precision and Accuracy of the Kairos Amino Acid Kit QCs in solution. Source: Waters Advanced Diagnostics
Kairos, AccQ•Tag, ACQUITY, UPLC, Xevo, CORTECS, MassLynx, TargetLynx, and waters_connect are trademarks of Waters Corporation or its
affiliates.
Acknowledgments
Produced from materials originally authored by Rachel McBrinn, Gareth Hammond, Padhraic Rossiter, and Lisa J. Calton from Waters Corporation.
For research use only. Not for use in diagnostic procedures.
About Waters Advanced Diagnostics
At Waters, we recognize the critical role hospital laboratories play in delivering timely, accurate diagnostics that drive patient care. Our solutions are built to support your lab’s evolving needs - whether you’re expanding test menus, improving turnaround times, or meeting rigorous regulatory standards.
Rooted in three foundational pillars, our approach is designed specifically for clinical lab environments like yours:
Flexibility – Integrate seamlessly into your existing systems and workflows.
Efficiency – Help your team deliver high-quality results faster and with fewer hands-on steps.
Trust – Proven technologies backed by decades of scientific innovation and global support.
From LC-MS/MS systems to automated sample preparation and data management tools, Waters offers comprehensive solutions that enable your lab to operate with confidence - today and in the future.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.