The Waters ACQUITY™ UPLC™ I-Class/Xevo™ TQD IVD System enables the quantification of organic compounds in human biological liquid matrices.
This article demonstrates the system’s analytical performance for the analysis of cyclosporine, sirolimus, everolimus, and tacrolimus in whole blood.
Experimental
The ACQUITY UPLC I-Class/Xevo TQD IVD System was controlled by MassLynx IVD (v4.1) and the data processed using the TargetLynx Application Manager.
Whole blood Calibrators and Quality Controls were processed according to the following conditions:

ACQUITY UPLC I-Class/Xevo TQD IVD System. Image Credit: Waters Advanced Diagnostics
Sample preparation conditions
A total of 50 μL of sample was processed with aqueous zinc sulfate and acetonitrile, then centrifuged. Each analyte was individually analyzed.
LC conditions
Source: Waters Advanced Diagnostics
| |
|
| Column: |
ACQUITY UPLC HSS C18 SB 1.8 μm, 2.1 mm x 30 mm |
| Mobile phase A: |
2 mM Ammonium acetate + 0.1 % formic acid in water |
| Mobile phase B: |
2 mM Ammonium acetate + 0.1 % formic acid in methanol |
| Flow rate: |
0.4 mL/min |
| Gradient: |
50 % B for 0.2 minutes, 50–100 % B over 0.4 minutes, hold 100 % B for 0.6 minutes, equilibrate with 50 % B for 0.6 minutes at 0.6 mL/min |
MS conditions
Source: Waters Advanced Diagnostics
| |
|
| Resolution: |
MS1 (0.75 FWHM), MS2 (1.2 FWHM) |
| Acquisition mode: |
MRM |
| Polarity: |
ESI (+) |
Results and discussion
Table 1 features the performance characteristics of everolimus, sirolimus, cyclosporine, and tacrolimus on the ACQUITY UPLC I-Class/Xevo TQD IVD System. Figure 1 illustrates the system’s analytical sensitivity.
Table 1. Performance characteristics of cyclosporine, everolimus, sirolimus, and tacrolimus. Range defined by linear fit where r2 > 0.99. LLOQ defined by S/N (PtP) > 10 and %RSD ≤ 20 %. % RSD at LLOQ determined through analytical sensitivity experiments performed over five occasions (n=50). Total precision and repeatability of QCs performed over five occasions in whole blood (n=25). EQA mean bias determined by comparison of obtained values to the LC-MS all laboratories trimmed mean (LC-MS ALTM) value (n=33). Note: The EQA mean bias for everolimus (-11.9 %) is based on EQA results from only two laboratories. Source: Waters Advanced Diagnostics
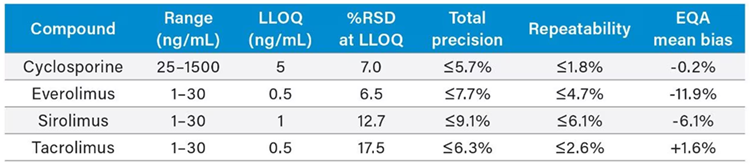
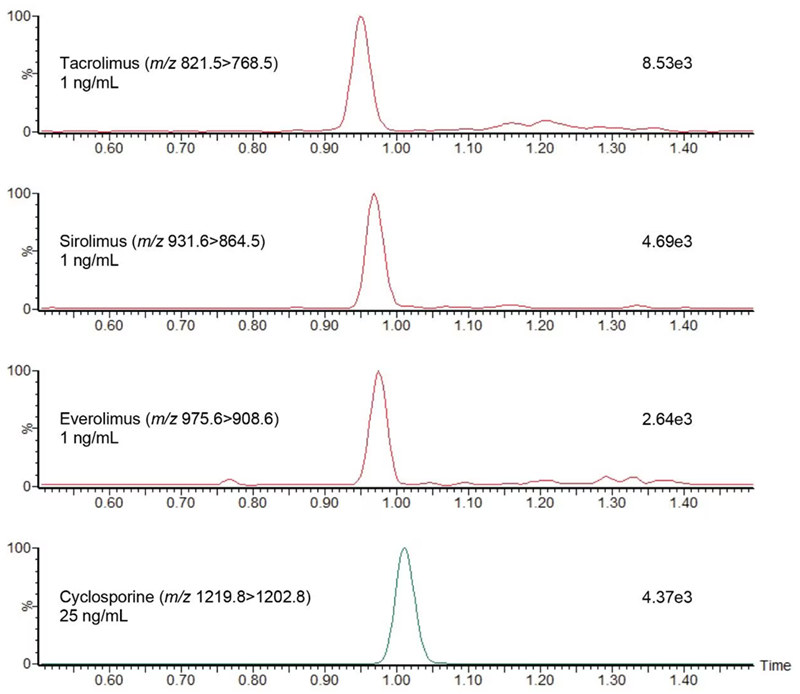
Figure 1. Chromatogram showing the analysis of 25 ng/mL cyclosporine and 1 ng/mL everolimus, sirolimus, and tacrolimus using the ACQUITY UPLC I-Class/Xevo TQD IVD System. Image Credit: Waters Advanced Diagnostics
Conclusion
The ACQUITY UPLC I-Class/Xevo TQD IVD System from Waters Corporation was able to deliver excellent analytical accuracy, precision, and sensitivity when used to analyze cyclosporine, sirolimus, everolimus, and tacrolimus in whole blood.
For in vitro diagnostic use. Not available in all countries.
Disclaimer
The analytical performance data presented here is for illustrative purposes only. Waters does not recommend or suggest analysis of the analytes described herein. These data are intended solely to demonstrate the performance capabilities of the system for analytes representative of those commonly analyzed using liquid chromatography and tandem mass spectrometry. Performance in an individual laboratory may differ due to a number of factors, including laboratory methods, materials used, intra-operator technique, and system conditions. This document does not constitute a warranty of merchantability or fitness for any particular purpose, express or implied, including for the testing of the analytes in this analysis.
Acknowledgments
Produced from materials originally authored by Waters Corporation.
About Waters Advanced Diagnostics
At Waters, we recognize the critical role hospital laboratories play in delivering timely, accurate diagnostics that drive patient care. Our solutions are built to support your lab’s evolving needs - whether you’re expanding test menus, improving turnaround times, or meeting rigorous regulatory standards.
Rooted in three foundational pillars, our approach is designed specifically for clinical lab environments like yours:
Flexibility – Integrate seamlessly into your existing systems and workflows.
Efficiency – Help your team deliver high-quality results faster and with fewer hands-on steps.
Trust – Proven technologies backed by decades of scientific innovation and global support.
From LC-MS/MS systems to automated sample preparation and data management tools, Waters offers comprehensive solutions that enable your lab to operate with confidence - today and in the future.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.