Integral membrane proteins are proteins are among the most valuable, but also most challenging, targets in drug discovery. However, the right approaches can overcome commonly encountered challenges.
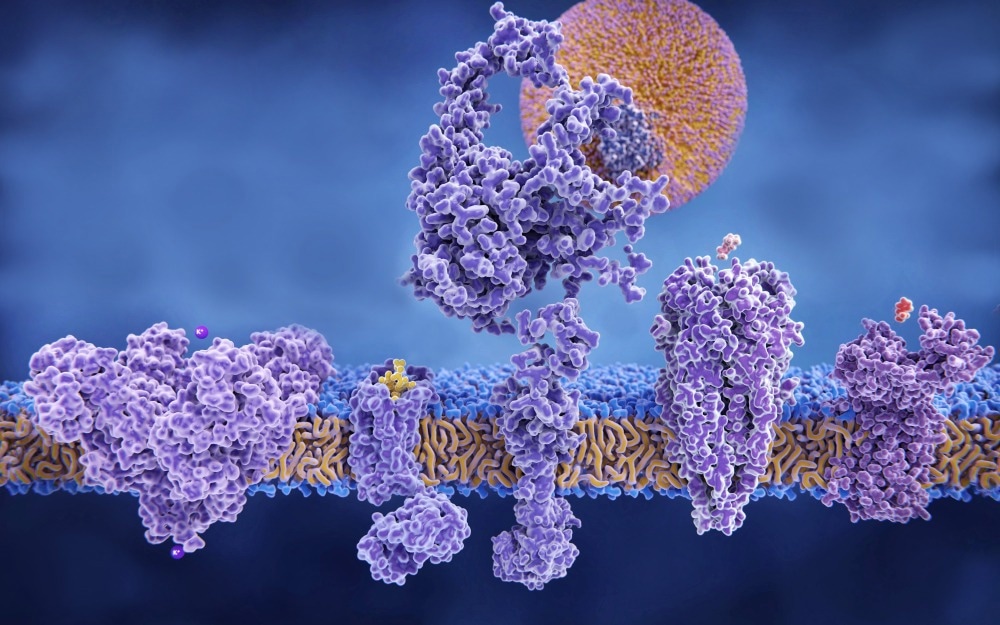
Image Credit: Juan Gaertner /Shutterstock.com
Despite comprising only about 25% of the human genome's proteins,2 membrane proteins account for more than half of all therapeutic targets.1
This notable overrepresentation as therapeutic targets exists because of their location at the beginning of many signaling cascades, and the fact that they are frequently found on the cell surface, making them easier to approach with biologic and small-molecule medications than other targets.
How to overcome the challenges of working with membrane proteins
Several characteristics of membrane proteins complicate drug discovery and development against these valuable targets. However, using the right approaches can overcome common challenges.
Low native expression
Membrane proteins are substantially less common than soluble proteins since they are only located in membranes' limited 2D region. In addition, each membrane protein may be expressed solely in a certain tissue or cell type, as well as in a single subcellular membrane.
To address this, a heterologous expression system such as E. coli (for simple targets), insect cells (for most GPCRs), or mammalian expression systems (for complicated targets requiring proper glycosylation) might increase functional expression levels.3
Careful construct design (using in silico approaches) and, when possible, screening of several constructs and orthologs can improve success rates even further.
Low stability, purity, and activity
Membrane proteins have evolved to reside in a phospholipid bilayer, and detergent extraction frequently leads to protein unfolding or aggregation, making it unsuitable for downstream processes.
There are several techniques that can be employed to address protein stability concerns. Careful detergent selection for extraction can assist in keeping the protein folded and active. The use of stabilized constructs and stabilizing point mutations can also result in increased yields and stability.
Membrane mimetics4 such as SMA, amphipols, peptidiscs, and nanodiscs offer another option for providing long-term stability in the absence of destabilizing detergents. Alternatively, a membrane environment can be maintained by reconstituting into liposomes for functional tests.
It can be possible to avoid isolating the proteins altogether. Activity assays, such as membrane transport, can be performed in overexpressed systems or relevant cell types, eliminating the requirement for purification, and minimizing interference from excess membrane mimetic molecules.
Due to limited expression and stability, sub-milligram yields purified from many liters of protein are frequently reported; obtaining high purity and activity is also challenging. Low yields can be overcome by applying extremely sensitive biophysical techniques for interaction analysis, such as SPR, GCI,5 and nanoDSF, to reduce protein consumption.
Challenges with structural characterization
Structural biology is often used to evaluate the molecular features of protein-protein or protein-small molecule interactions, allowing for the rational design of therapeutic molecules through Structure-Based Drug Design (SBDD).
Membrane proteins are naturally flexible, have conformational heterogeneity, and are unstable over time, making it difficult to create well-ordered crystals for X-ray crystallographic structure determination.
Suitable construct design, careful purification, and detailed biophysical characterization can all improve the odds of structure identification, which in turn improves crystallographic success.
Using cryo-EM eliminates the need for ordered crystal formation and enables the determination of many structures from a single sample, making it especially beneficial for large, flexible, full-length membrane proteins and multi-subunit complexes. 6
How Concept Life Sciences can support discovery
Despite the challenges involved in characterizing membrane proteins, they continue to be promising targets for small chemical and biologic therapy development.
Concept Life Sciences has substantial hands-on experience expressing and purifying these tough targets, as well as their biophysical and structural characterization.
Concept Life Sciences provides construct design; protein production in bacterium, insect, and mammalian systems; and purification using detergents and other membrane mimetics. The company also offers a suite of QC and biophysical characterization, binding procedures, cryo-EM, and crystallography services.
References
- Overington, J.P., Al-Lazikani, B. and Hopkins, A.L. (2006). How many drug targets are there? Nature Reviews Drug Discovery, (online) 5(12), pp.993–996. DOI: 10.1038/nrd2199. https://www.nature.com/articles/nrd2199.
- The Human Protein Atlas.The human proteome in membrane proteome - The Human Protein Atlas. Available at: https://www.proteinatlas.org/humanproteome/tissue/membrane+proteome.
- Kesidis, A., et al. (2020). Expression of eukaryotic membrane proteins in eukaryotic and prokaryotic hosts. Methods, (online) 180, pp.3–18. DOI: 10.1016/j.ymeth.2020.06.006. https://www.sciencedirect.com/science/article/abs/pii/S1046202320301080.
- John William Young (2023). Recent advances in membrane mimetics for membrane protein research. Biochemical Society Transactions, (online) 51(3), pp.1405–1416. DOI: 10.1042/bst20230164. https://pmc.ncbi.nlm.nih.gov/articles/PMC10317169/.
- Sharma, P., et al. (2021). Kinetic analysis of antibody binding to integral membrane proteins stabilized in SMALPs. BBA Advances, 1, pp.100022–100022. DOI: 10.1016/j.bbadva.2021.100022. https://www.sciencedirect.com/science/article/pii/S2667160321000211.
- Nygaard, R., Kim, J. and Mancia, F. (2020). Cryo-electron microscopy analysis of small membrane proteins. Current Opinion in Structural Biology, 64, pp.26–33. DOI: 10.1016/j.sbi.2020.05.009. https://www.sciencedirect.com/science/article/abs/pii/S0959440X20300774?via%3Dihub.
Acknowledgments
Produced using materials originally authored by David Wright from Concept Life Sciences.
About Concept Life Sciences
Concept Life Sciences is a leading contract research organisation (CRO) serving the global life sciences industry. For over 25 years, the company, and its heritage companies, have provided consultative and collaborative drug discovery and development services. Our approach, supported by passionate scientists and world-leading capabilities, enables clients to overcome complex scientific challenges across a broad range of therapeutic areas, improving program success rates. The company has successfully helped 29 candidates advance to the clinic.
The company offers sophisticated translational biology services coupled with exceptional end-to-end chemistry capabilities across all modalities, including small molecules, biologics, peptides and cell & gene therapies, with the ability to seamlessly integrate capabilities and provide bespoke solutions to address client needs.
Collectively, the company’s high-quality services and commitment to customer service across the drug development pathway enhance efficiency in drug discovery, helping clients advance their drugs to clinic in as little as 32 months, well ahead of the industry average of 60 months.
Driven by a passion for science, Concept Life Sciences has around 230 employees, with around 70% holding PhDs. The company operates from state-of-the-art UK facilities, headquartered near Manchester, with additional operations in Edinburgh, Dundee, and Sandwich. The headquarters is one of the UK’s largest medicinal chemistry CRO sites with key discovery services all under one roof.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.