Please can you give a brief introduction to the CLIRSPEC network?
The CLIRSPEC network is a UK Engineering and Physical Sciences Research Council (EPSRC – EP/L012952/1) funded network in clinical infrared and Raman spectroscopy.
Infrared and Raman spectroscopy can identify the hallmarks of disease and distinguish between diseased and non-diseased samples based upon inherent chemistry.
In order to realise the potential of clinical infrared and Raman spectroscopy there are many hurdles to overcome to move from the proof of concept stage through the translational stage and into the clinical setting.
The network has been set up to overcome these hurdles by bringing together academics, industrialists, and clinicians to pool our resources and work on these generic problems together.
 (a) Grayscale image mosaic of 50 unique serum biopsies (row 1 non-cancer controls, 2 brain, 3 breast, 4 lung, and 5 skin cancer patient samples). The scale bar displays 500 pixels (equivalent to a spatial resolution of 2.1 mm at a ? = 5.5 µm). (b) Classification outcomes for correct cancer diagnosis were accurate to 90%, 90%, 90%, and 80%, highlighted by color for brain (blue), breast (pink), lung (yellow) and skin (red) cancer samples respectively. Figure credits: Caryn Hughes, University of Strathclyde, Benjamin Bird, Daylight Solutions, Matthew J Baker, University of Strathclyde
(a) Grayscale image mosaic of 50 unique serum biopsies (row 1 non-cancer controls, 2 brain, 3 breast, 4 lung, and 5 skin cancer patient samples). The scale bar displays 500 pixels (equivalent to a spatial resolution of 2.1 mm at a ? = 5.5 µm). (b) Classification outcomes for correct cancer diagnosis were accurate to 90%, 90%, 90%, and 80%, highlighted by color for brain (blue), breast (pink), lung (yellow) and skin (red) cancer samples respectively. Figure credits: Caryn Hughes, University of Strathclyde, Benjamin Bird, Daylight Solutions, Matthew J Baker, University of Strathclyde
What do you see as the critical steps to transitioning infrared microscopy and imaging techniques into clinical use?
This is a very difficult question and one that will have many answers. I currently see two of the main requirements as outreach and evidence.
I think we need to let people know more about the excellent potential of IR microscopy and imaging in the clinic and we need to move towards a large-scale clinical trial – the main factor in this approach will be identifying the clinical issue that will be able to enable IR translation quickly.
Can you provide an overview of your recent publication in Spectroscopy Europe related to infrared imaging of blood serum?
Biofluids provide an easily accessible sample source that can be collected worldwide. Using IR to characterize and diagnose biofluids is a rapidly emerging field.
Following on from previous work where we were able to discriminate the serum from non-cancerous patients and from brain tumour patients using ATR-FTIR, we wanted to see if we could push the speed and focus in on the salient information required for serum diagnostics.
Using Spero, a new laser-based infrared microscope from Daylight Solutions, we were able to rapidly collect spectral images that represent 14 discrete frequencies, identified as diagnostic in our previous study, in only 2 minutes per tile.
From these frequencies we were able to diagnose to similar powers as were possible when analysing 900 frequencies when collecting in a Fourier Transform Infrared (FTIR) set up. This has moved the spectral collection onto truly clinical timescales.
We hope to expand this study to prove the power of Discrete Frequency Infrared (DF-IR) for rapid patient spectroscopic serum diagnostics and enable clinical translation.
Spero Highlights for SciX 2014
How has the Spero microscope supported your research efforts?
The main way in which Spero has supported my research efforts is really providing a versatile system that can collect the required information for my diagnostic research within clinically relevant times.

This helps me to open dialogue with clinicians and speak about current technology as opposed to very fundamental instrumentation development that is required.

How do you plan to take advantage of the live and discrete frequency imaging capabilities of Spero?
Both of these capabilities will provide excellent opportunities for the future and I will capitalize on the progress made so far to carry on with the discrete frequency tissue and serum analysis to drive forward the clinical translation of IR imaging and microscopy.
But I am particularly looking forward to the live frequency imaging to enable rapid intra-operative diagnostics and also, combined with the brilliant source, to look at live cell imaging.
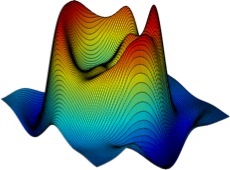
3-D mesh plot showing the total IR absorbance (1800 – 1000 cm-1) collected from a dried human pooled serum drop (ca. 1000 µm in diameter) acquired using a single frame from the Spero™ Infrared Microscope with a low magnification objective (FOV = 2 mm × 2 mm, 4X magnification, numerical aperture (NA) = 0.15, pixel size = 4.25 µm, spatial resolution = ca. 25 µm at ? = 5.5 µm). Figure credits: Caryn Hughes, University of Strathclyde, Benjamin Bird, Daylight Solutions, Matthew J Baker, University of Strathclyde
How do you believe the availability of rapid high resolution imaging in desktop instruments will impact the field?
The possibilities are endless across domains for this sort of responsive imaging. In my field I think it will enable us to illuminate new information concerning biological processes. This information would be obtainable across many different biological issues as the sample preparation is relatively simple. The impact could be significant.
What new features would you like to see in next-generation laser-based infrared microscopes?
I would like to be able to analyse from 4000 – 400 wavenumbers and then push the limits of portability that can be achieved. A handheld high-resolution microscope with chemical imaging capability would be great.
What other applications do you plan to pursue with the chemical sensing capabilities of quantum cascade lasers?
In addition to the biological research I would be very interested in stand-off detection of chemicals for defence, security, and forensics purposes and also investigating the ability for non-invasive monitoring of health status.
Where can readers find more information?
About Dr Matthew Baker
For just over a decade, I have been researching the use of spectroscopy and spectrometry geared towards chemical and biological detection challenges.
Following my PhD at the University of Manchester on the “Application of Time-of-Flight Secondary Ion Mass Spectrometry to Cellular Analysis”, I held an EPSRC Life Science Interface Fellowship targeted at “Spectrometric and Spectroscopic Molecular Pathology and Diagnosis” at The University of Manchester, Robert-Koch Institute, Berlin and Harvard Medical School.

After this, I worked for the UK Ministry of Defence and the Defence Science and Technology Laboratory focusing on research within the Chemical, Biological, Radiological, and Nuclear (CBRN) field and I am currently at The University of Strathclyde where I perform research to understand the composition and behavior of molecules within complex matrices related to real-world detection challenges using spectroscopy and spectrometry.
I am also Co-Investigator and Younger Members Lead for the CLIRSPEC Network, Clinical Infrared and Raman Spectroscopy for Medical Diagnosis.