Sep 13 2017
For a cancer drug to be successful, it needs to reach the malignant tumor site. Researchers in Japan have now found a way for increasing the effectiveness of drug delivery to certain types of brain tumors, by packing epirubicin, a known antitumor agent, in specially designed polymeric micelles. In a mouse model, use of the micelles led to a 12-fold increase in antitumor activity, making it a highly promising, translationable approach for decreasing the drug resistance of brain tumors.
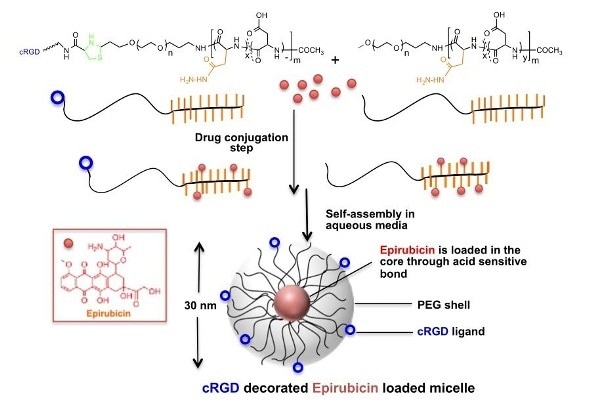
Synthesis and structure of cRGD-decorated epirubicin-loaded micelles, shown to lead to the suppression of gliobastoma multiforme tumors in a mouse model.
Kazunori Kataoka and colleagues at the Kawasaki Institute of Industrial Promotion, The University of Tokyo and Tokyo Institute of Technology applied their method to gliobastoma multiforme (GBM), an aggressive brain cancer for which less than 5% of patients survive more than 3 years. Its resistance to drugs is mainly due to the blood–brain barrier, a layer of tightly packed cells, the function of which is to separate the brain from the circulatory system. In order for a drug to reach the brain tumor site in therapeutic concentration, it needs to penetrate this barrier.
Epirubicin is a potent antigliobastoma drug, more effective than temozolomide, the current clinical medicine, but it penetrates poorly through the blood–brain barrier. In order to improve the delivery of epirubicin, the researchers studied the effect of decorating epirubicin-containing micelles with cyclic Arg-Gly-Asp (cRGD) peptides. The latter are biomolecules implicated in cell-attachment mechanisms involving integrins — receptors embedded in cell membranes. The cRGD molecules target so-called ανβ3 and ανβ5 integrins that are generated in GBM cells and its vascular system; molecular recognition between cRGD and the integrins leads to better penetration.
An in vitro penetration study on multi-cellular glioma spheroid revealed cRGD-decorated-epirubicin-loaded micelles penetrate deep into the spheroids in higher quantity compare to micelles without cRGD. Bioluminescence imaging of mice injected with the cRGD-decorated micelles also showed that in vivo, the therapy led to the effective suppression of the growth of orthotopic GBM. Given that clinical trials are already underway for untreated epirubicin micelles, the researchers concluded that “the straightforward modification of their surface with cRGD peptide could potentially extend their clinical application as an effective brain tumor therapy.”