Feb 27 2018
BGN Technologies, the technology transfer company of Ben-Gurion University of the Negev, announced today its collaboration with Biosensorix, a Singapore-based company in the field of medical diagnostics, to develop next-generation, quantitative, point-of-care diagnostics based on research conducted at Ben-Gurion University (BGU). The new technology is an electrochemical lateral flow immunosensor that conducts quantitative diagnosis of disease related biomarkers and pathogens, in the form of a smart USB-like drive.
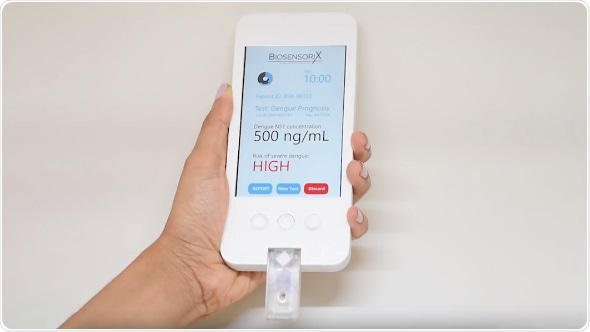
Next-generation, point-of-care diagnostic kit in the form of a smart USB-like drive
The business collaboration was created following joint research of researchers at BGU and Nanyang Technological University of Singapore to further develop the technology, as part of the Singapore-Israel NRF CREATE program for interdisciplinary research.
“There are fast-acting testing kits today that can be used both in the clinic and at home to diagnose a wide range of conditions and diseases, but none of them can discern the severity of the condition or disease,” says Prof. Robert Marks, of the Avram and Stella Goldstein-Goren Department of Biotechnology Engineering at BGU. “In order to determine the severity of a disease, the patient needs to conduct a blood test at a hospital or specialized laboratory, with results taking between half to an entire day, and sometimes even longer.”
“At the price range of $5 per kit, our new diagnostic kit presents a breakthrough in diagnosis,” says Dr. Lukas Fajs, CEO of Biosensorix. “Medical staff will be able to receive immediate results and decide on a course of action, instead of sending the patient to the lab and waiting for the results.”
The first application developed by Biosensorix is for the detection of dengue hemorrhagic fever, a disease endemic in South East Asia and South America. “Most people with dengue fever can be released to home care, yet are kept at the hospital until results come in,” says Prof. Marks. “Our kit provides physicians with true triage power: with the new diagnostic kit, the physician can release the patient within half an hour, saving time and money.”
The second application under development is assessment of stroke severity as well as early detection of a secondary stroke, which often occurs after the patient is released from hospital. The current diagnosis of ischemic stroke that relies today on clinical assessment and neuroimaging, can be significantly speeded up by using this novel diagnostic kit. The kit detects specific biomarkers that are present in the blood following ischemic stroke.
“The new kit can help achieve faster and more accurate diagnosis even when MRI results are available, and can certainly help in cases where diagnosis is unclear,” says Dr. Ora Horovitz, Senior VP Business Development at BGN Technologies. “With stroke every minute counts. A quick quantitative test means rapid diagnosis that is necessary for accurate timely treatment. This can save brain functions and even lives,” adds Dr. Horovitz.
The new kit has the potential to help distinguish ischemic stroke from stroke mimics, to determine the stroke etiology, its severity and outcome (such as neurological deterioration and hemorrhagic complications), and help decide on the correct treatment.