Congestive heart failure, as it is properly called, affects over 5 million Americans, and is caused by a failure of the heart muscle to pump blood strongly enough to supply the body with enough oxygenated blood.
The problem
Heart failure worsens over time, because of the input from diverse pathways such as the sympathetic system, the renin-angiotensin-aldosterone system (RAAS) and others.
RAAS activation is linked to retention of water and sodium, dilation of the heart, dysfunction of the left ventricle, and cardiac cachexia associated with muscle wasting. This leads to discomfort, difficulty with breathing, swelling or edema, and tiredness. Edema appears not only as fluid accumulation around the lung tissue (pulmonary and pleural edema), but also in the body cavities and soft tissues (ascites and peripheral edema, such as ankle swelling).
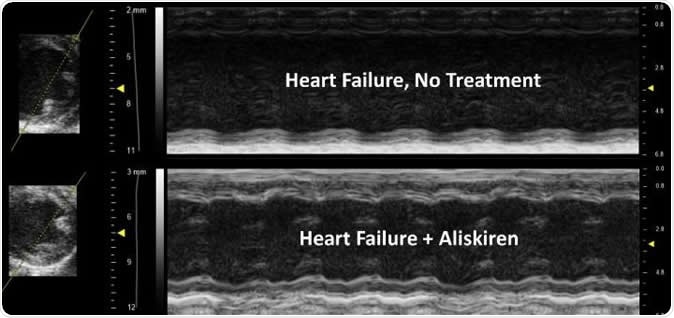
The treated image shows improved contractility of the heart, less ventricle/chamber dilation and improvement in wall thickness compared to the untreated heart. Image Credit: UA College of Medicine Phoenix
However, it is not easy to measure to what extent these changes are occurring because neither body swelling or muscle wasting are visible from the outside in many patients, leading to a lack of awareness of the severity of the heart failure. In particular, extracellular water retention is not routinely measured in current clinical practice, in a non-invasive way, even though this occurs before any clinical symptoms occur and before edema is detectable. The present study showed that this single feature was adequate to predict mortality beyond a certain limit.
Without independent and reproducible ways to measure these clinical features, it is difficult to accurately gauge the response to different medical interventions for symptoms like shortness of breath, swelling, lung congestion and other features of cardiac failure, or for cardiac cachexia, the involuntary wasting that occurs in these patients.
Better techniques to measure these effects are badly needed to help develop improved preventive and therapeutic methods. The need for precision medicine is being increasingly recognized in heart failure, because different patients have varying degrees of left ventricular pump failure, and different rates of progression, despite being put in the same diagnostic category.
The solution
The researchers from the Cardiovascular Disease Research group from the UA College of Medicine - Phoenix Department of Internal Medicine looked at the use of noninvasive technology in the form of QMR (quantitative magnetic resonance) in a mouse model with heart failure due to dilated cardiomyopathy, which mimics the progression of heart failure through stages A to D in human beings. They treated female mice with stage B heart failure onwards with aliskiren in a randomized blinded manner. All the mice had high plasma renin at the beginning of the study.
In this study, aliskiren was used orally rather than subcutaneously, to better mimic actual treatment conditions and to ensure that the drug distribution in the body could be followed, in accordance with what is known already about oral administration.
The use of QMR showed how Aliskiren was of benefit in congestive heart failure. Aliskiren is a direct renin inhibitor (DRI), a drug which inhibits renin, one of the enzymes involved in blood pressure regulation via the RAAS. The use of this drug suppressed plasma renin selectively, and reduced other markers of heart failure like neprilysin, without detectable impact on angiotensin II and aldosterone, two essential components of normal RAAS function.
They examined body composition in the mice every ten days to find out how fluid retention increases progressively with heart failure, using QMR. In fact, this is the first study to examine the use of QMR as a way to evaluate systemic edema. This provided an objective measure of this symptom in terms of the weight of extracellular water, rather than merely relying on imaging findings. In addition, they measured body fat and lean muscle mass longitudinally to assess the changes in these parameters over time. Wasting of body fat and loss of muscle mass forecasts a poor outcome in patients with heart failure. In this study, normalized renin levels were linked to better heart contraction, relief of fluid retention and swelling, and less cachexia and muscle wasting, with fewer deaths.
Implications
Being able to reliably assess such changes can help greatly to improve care for such patients by providing a means of monitoring and making required adjustments to treatment protocols, thus eventually leading to precision medicine in these cases. QMR offers advantages such as ease of application, speed of recording, reproducibility of findings, and measurement accuracy that obviates the need for specialist interpretation of images.
Researcher Ryan Sullivan says, “That's an extra 5.6 years with loved ones that otherwise would not be possible. Obviously, further studies are needed, along with human clinical trials, but we are excited about our research direction and what those outcomes could mean for the college and the people of Arizona and beyond. These exciting findings demonstrate new approaches that may guide future care for heart failure patients.”
Source:
Journal reference:
Normalizing plasma renin activity in experimental dilated cardiomyopathy: effects on edema, cachexia, and survival. Ryan D. Sullivan, Radhika M. Mehta, Ranjana Tripathi, Inna P. Gladysheva, and Guy L. Reed. International Journal of Molecular Science 2019, 20 (16), 3886. https://www.mdpi.com/1422-0067/20/16/3886/htm