Being made up of flesh and bone, the human body often suffers injuries due to tendon damage caused by various acute or chronic events. As these damaged tissues recover, scar tissue builds up, causing a slow and often painful recovery. Such sequelae are frequently observed following injuries such as tears of the shoulder rotator cuff, or inflammation of the patellar tendon in front of the lower part of the knee joint (jumper’s knee). If the tendon could heal without such scarring, the result might be a stronger, cleaner tissue. The fact is that tendons hardly even recover completely, once damaged.
Now, a new study published in the journal Nature Cell Biology shows that tendons contain stem cells that could be put to good use to stimulate the healing of the tendon, to such an extent that it may even avoid surgery. By improving tendon healing, these cells could help restore full joint mobility and avoid regular use of pain relievers as well.
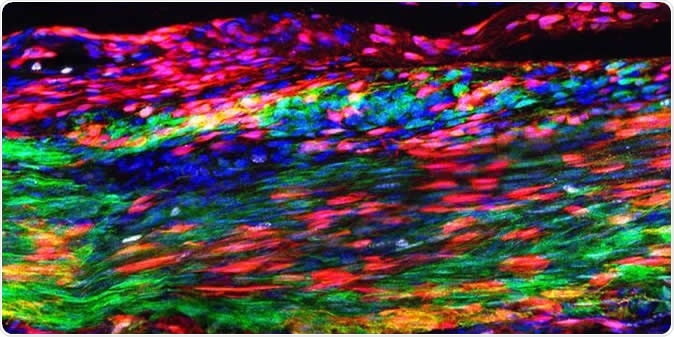
This image shows the Patellar tendon 30 days after an injury. The red marks newly discovered tendon stem cells that have self-renewed and are layered over green marked, original tendon cells. During regeneration, some tendon stem cells differentiate to make newly regenerated tendon cells--a process during which they transition into a yellow-orange color. The blue indicates cellular nuclei. Image Credit: Tyler Harvey
Tendons
Tendons are tough thin bands of connective tissue that extend from the end of the muscles to merge with the fibrous covering over the bone, called the periosteum. Thus, these structures form a strong connection between bone and muscle and allow the muscles to move the bones to execute various movements. On the other hand, tough as they are, they can be easily injured or even torn by excessive force.
Most tendon injuries seem to occur all at once but are the consequences of multiple smaller injuries that have occurred over time. With increasing age, degenerative changes set in that increase the chances of injury.
The slow and partial healing of tendons is notorious for the consequences it causes for the patients. For instance, the tendon structure may be spoilt because of the presence of a fibrous scar in the middle of the tendon. Especially with older patients and those who use their muscles fiercely, as with competitive athletes, tendon repair is required but entails a long period of rehabilitation and may still not ensure complete restoration of function.
Tendon degeneration has been described by previous researchers as a failure of adaptation and remodelling of the tendon matrix because of the lack of balance between the decomposition and synthesis of the matrix.
The study
The current study was aimed at establishing the presence of stem cells within tendon tissue. The researchers uncovered all the cell types present in samples of the patellar tendon. They found a new population of cells – tendon stem cells.
Stem cells are found in almost all tissue types in the body. These cells are characterized by their ability to divide almost indefinitely, as well as the ability to develop into mature cells of different types. Each tissue has its own pool of stem cells, which either divides to keep up the supply, or matures into any of the different kinds of cells that make up the tissue. This helps the tissue to heal when injured.
Until now, despite much searching by many researchers, nobody had ever seen a tendon stem cell in human tendon tissue. In fact, many doubted their existence, because of the slow incomplete healing of these tissues.
However, the current study showed that not only do they exist, they come from the same location as do the cells that make up the fibrous scar – the cells in the protective surface layer of the tendon. The two types of cells compete with each other to produce healing of an injured tendon. This could be why it takes so long for a strong tendon scar to form following an injury.
When tendon tissue is disrupted, a protein called platelet-derived growth factor A is released from platelets, which stimulates the activity of both tendon stem cells and scar precursor cells. If anything causes the tendon stem cells to stop responding to this growth factor, on the other hand, the scar that results is purely fibrous, without the formation of new tendon cells.
Implications
The challenge is to stimulate tendon stem cells selectively so that they respond faster and more intensely than scar precursor cells. This prevents the scar from being a fibrous one, and instead promotes healthy tendon tissue growth. Other scientists have suggested that human ageing processes cause depletion of the tendon stem cells pool in numbers as well as in fitness, impairing tendon recovery.
Researcher Chen-Ming Fan says, “Finding a therapeutic way to block the scar-forming cells and enhance the tendon stem cells could be a game-changer when it comes to treating tendon injuries.”
Journal reference:
Harvey, T., Flamenco, S. & Fan, C. A Tppp3+Pdgfra+ tendon stem cell population contributes to regeneration and reveals a shared role for PDGF signalling in regeneration and fibrosis. Nat Cell Biol (2019) doi:10.1038/s41556-019-0417-z, https://www.nature.com/articles/s41556-019-0417-z