A new Russian study shows the vital role played by HLA class I molecules in the risk of lethal COVID-19.
Several risk factors for a poor outcome following severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection have been identified. Among these, age is critical. Chronic underlying disease has also been shown to have a deleterious impact on the course and severity of the illness.
The study, published on the preprint server medRxiv*, explores the ability of HLA class I molecules to mediate the outcome in COVID-19 patients. These molecules are important in the immune response. They bind viral proteins, acting as receptors within the host cell. The HLA class I-viral antigen complex is then transferred to the cell surface, signaling the presence of the infection to any patrolling CD8+ T cell. The T cell receptor (TCR) interacts with the bound antigen, responding by T cell activation.
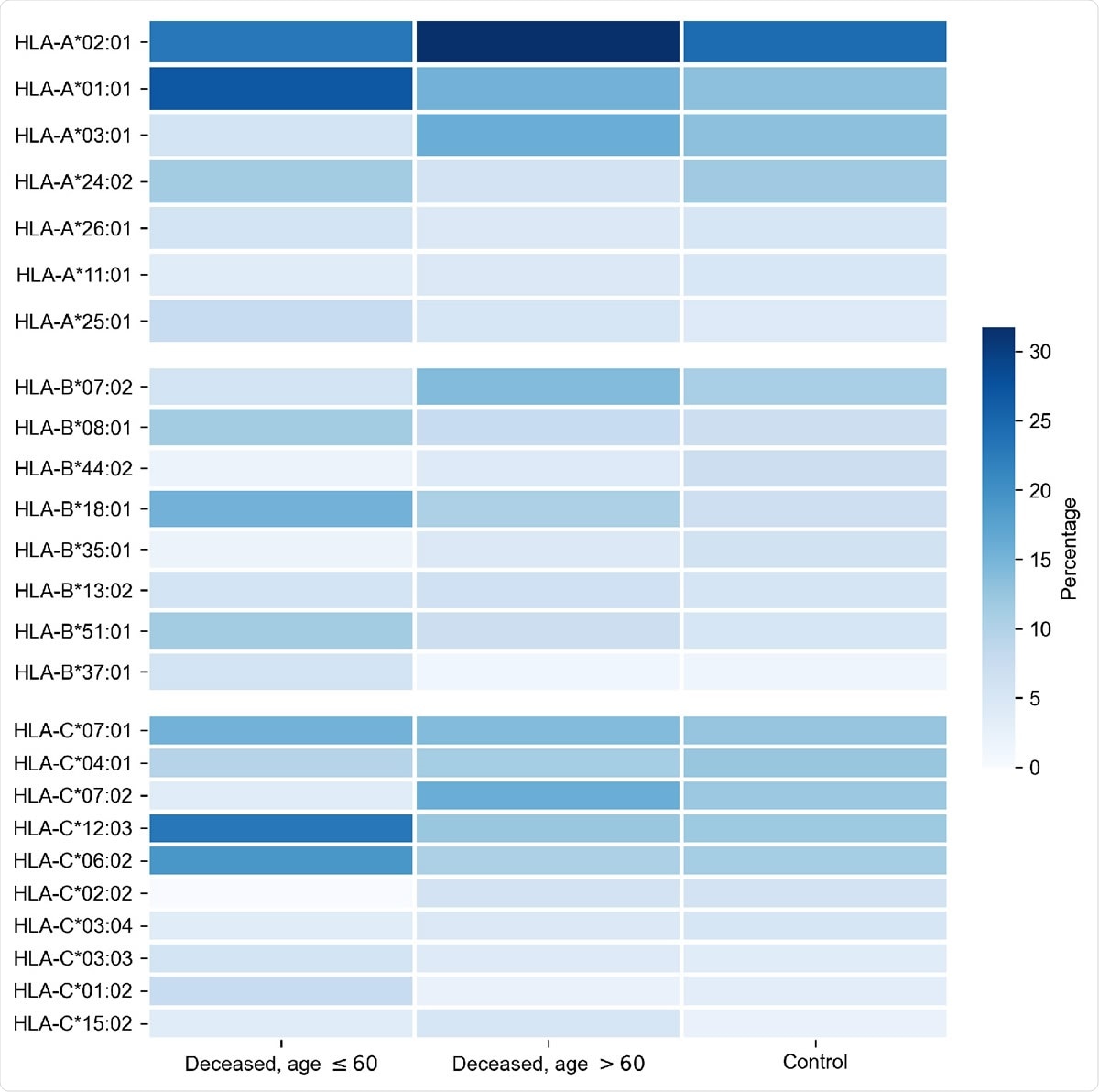
Distribution of HLA-A, HLA-B, HLA-C alleles in cohorts of deceased COVID-19 patients and the control group. Alleles with frequency over 5% in at least one of three considered groups are presented.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
The activated T cell begins to divide, resulting in the expansion of a clone of cytotoxic CD8+ T cells specifically targeting the virus. These are called cytotoxic because they can destroy the infected cells, using their powerful perforin and serine protease enzymes to break down the cell membrane. This is one way the innate immune system limits the infection by destroying the infected cells before the virus can replicate further and spread to other cells.
HLA class I receptors are of three types, A, B and C, each of which has two copies in any given individual, one from each parent. Each copy or allele has many different variants and can recognize many different foreign proteins. The pattern of distribution of these alleles is characteristic across various populations and countries.
Earlier reports suggest that different HLA gene profiles affect the susceptibility to SARS-CoV. However, there is little to be known about how they impact the individual risk of COVID-19. One small study showed that a higher frequency of HLA-A*01:01 and HLA-A*02:01 alleles correlates with the number of infections and the mortality rate in various parts of Italy.
The current study aimed to identify the contribution of HLA class I genotypes to the severity of COVID-19. The researchers examined the HLA genotype of 111 patients with fatal COVID-19 and ~430 controls by scoring each allele based on how well it could present over 6,500 unique SARS-CoV-2 peptides. None of the participants had severe chronic disease. However, only a fifth were free of chronic underlying disease.
The risk score differentiated the deceased below-60 group from the controls and the over-60 deceased patient group. A low risk score was associated with higher HLA-A*02:01 and HLA-A*03:01 frequencies, while HLA-A*01:01 was overrepresented in the high-risk group. This agrees with existing research.
They then compared the genotypes in three groups: adults who died at or before the age of 60, adults older than this, and controls. About 4% of the first group had cerebrovascular disease compared to ~34% in the second. Chronic kidney disease stages 4 or 5 were higher in both groups relative to the population frequency of 0.05%, at ~23% and ~17% for those below and above 60 years, respectively.
Notably, the median age of death among COVID-19 patients is 73 years, agreeing with earlier studies and emphasizing the primary risk associated with advanced age. Over 60% of the deceased at or below 60 years in this study were men, while less than half the population in this age group are men. This conforms to earlier research findings that COVID-19 prevalence is higher in males. In the older deceased patient group, only ~38% of the population are male, but almost 50% of this group were composed of males.
The researchers also looked at homozygous HLA alleles in these groups and how each of the viral proteins was linked to COVID-19 risk. They found that the distribution of homozygous HLA-A*01:01 alleles was disproportionately increased in the below-60 years group compared with either those above 60 years or the control group. While 4/26 in the first group of deceased patients were homozygous for this allele, only 1/85 older deceased patients and 8/428 controls were homozygous. Other alleles did not show this pattern.
However, overall, patients who were homozygous for any allele had a lower age at death relative to those with heterozygous alleles. The proportion of homozygosity was higher, at ~42%, in the below-60 group, compared to 15% to 19% in the other two groups.
Interestingly, homozygosity for HLA-A*02:01 and HLA-A*03:01 was associated with low risk, and later age at death.
The researchers comment, “Homozygosity by HLA class I genes is generally associated with poor prognosis except some alleles like HLA-A*02:01 and HLA-A*03:01.”
The reason attributed for this is the double intensity with which important or relevant viral peptides are presented in these patients, causing a hyper-immune response. In fact, they say, “HLA class I homozygosity can act like a double-edged sword.”
These important peptides were dispersed across multiple viral proteins. The additional implication is that a vaccine based on the spike protein should act at least as well as inactivated whole-virus vaccines since they contain the same antigens that bind to HLA class I receptors.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources