As scientists continue to grapple with the coronavirus disease 2019 (COVID-19), caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), special attention has been paid to population groups at high risk. This includes healthcare workers (HCWs) who are on the frontline of the pandemic and are frequently exposed to the pathogen for long periods of time.
A new study by US-based researchers describes the results of a study on the immune response among HCWs, in particular, their T cell responses predating the pandemic.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
The team’s findings are available on the medRxiv* preprint server.
Endemic coronaviruses
There are a number of endemic coronaviruses (CoVs), such as the human CoVs (HCoVs), especially the one responsible for the common cold – known as the “common cold” coronavirus (CCC).
The researchers cite alphacoronaviruses 223E and NL63, and betacoronaviruses OC43 and HKU1 as also belonging to this category. They are both seasonal and cause endemic mild infections of the upper and lower respiratory tract. The genomes of OC43 and HKU1 share some degree of commonality with SARS-CoV-2, since they share a common family tree (betacoronavirus).
Earlier studies have established the presence of cross-reactive memory CD4 T cells that react to SARS-CoV-2 in naïve patients, along with cross-neutralizing antibodies. The impact of such pre-existing immunity remains uncertain, especially the degree of protection or, conversely, harm, caused by it. The current study thus aims to understand T cell immunity to CCC in HCWs.
Study details
The subjects of two cohorts came from Miami, and included HCWs in high-risk professions, classified as seronegative or positive (either seropositive, or positive by polymerase chain reaction (PCR) testing). A third (“shelter in place”, SIP) cohort comprised seronegative volunteers from the community, without exposure to infected persons. Finally, there was a control cohort of asymptomatic unexposed and seronegative subjects from San Diego, and another of seropositive individuals from the same area. Each cohort contained 10-32 subjects, with the median age being 41 years.
The other four cohorts used the enzyme-linked immunosorbent assay (ELISA) to test for antibodies to SARS-CoV-2’s spike protein and receptor-binding domain (RBD), with both required to be positive. The SIP cohort contained an IgG and IgM assay for antibodies targeting the N antigen. Significant titers were present in all HCWs from the two positive cohorts, with the lowest titers being correlated with asymptomatic disease. Seronegative cohort subjects had undetectable titers.
Interestingly, all three Miami cohorts were seropositive for the four endemic CCCs, with the highest reactivity to NL63. Titers were higher for HKU1 in the positive Miami cohort, a finding that agrees with the back-boosting effect observed in COVID-19, whereby reactivity to endemic CCC HKU1 and OC43 spike proteins is enhanced. These results confirm that most people are reactive to the CCC viruses.
Higher CD4 T cell reactivity in negative HCWs
The researchers also examined activation-induced markers OX40 (CD134) and 4-1BB (CD137) to differentiate recent T cell activation from prior CCC-related activation. They used sets of dominant class II-restricted T cell peptides (225 to 294 each) for the four CCCs, in order to examine the hypothesis that CCC CD4 T cell reactivity would be higher in the negative HCWs.
The cross-reactive CCC-SARS-CoV-2 T cell epitopes were homologous for up to 67% or more residues. When these were removed, the researchers found specific CD4 T cell reactivity to the four CCCs, highest in the negative HCW cohort. Compared to the naïve SIP cohort, the greatest difference was for NL63 and the lowest for HKU1. Relative to the positive Miami cohort, however, the highest reactivity was seen against 229E, NL63 and OC43. The same patterns were replicated in the two control San Diego cohorts.
Thus, CD4 T cell reactivity against CCC was higher in healthy unexposed donors vs COVID-19 donors.
Higher SARS-CoV-2 CD4 T cell reactivity in positive HCWs
The CD4 T cell responses to the full-length SARS-CoV-2 spike and other peptides were highest in the positive cohort, compared to the other two (negative and SIP), and were found to be due to memory T cell activation. This is due to SARS-CoV-2 infection in the positive cohorts.
In the other two, however, the observed reactivity may be, again, due to the reactivation of cross-reactive CD4 T cells from prior CCC exposure, and is unlikely to be due to resolved SARS-CoV-2 infection, as shown by the CCC-specific responses.
CD8 T cell reactivity to SARS-CoV-2 epitopes
The CD8 T cell reactivity to the SARS-CoV-2 spike protein was highest in the positive cohort, and at lower comparable levels for the other two Miami cohorts. This confirms that the higher total reactivity in the negative cohorts to CCC is due to CD4 T cell activation, rather than prior SARS-CoV-2 infection.
What are the implications?
For the first time, the researchers used pooled epitopes to distinguish T cell reactivity to CCC and SARS-CoV-2 peptides. The findings show that CCC reactivity in T cells is higher in HCWs who have been exposed to respiratory viruses than in a community cohort without similarly high levels of exposure. However, CCC antibodies were similar in all cohorts.
They concluded that this was associated with higher T cell reactivity against SARS-CoV-2 peptides in the absence of actual infection with this virus. They also found that in the vast majority of seronegative individuals have not been infected. Activation markers indicating recent SARS-CoV-2 infection were found only in positive HCWs, in both CD4 and CD8 T cells.
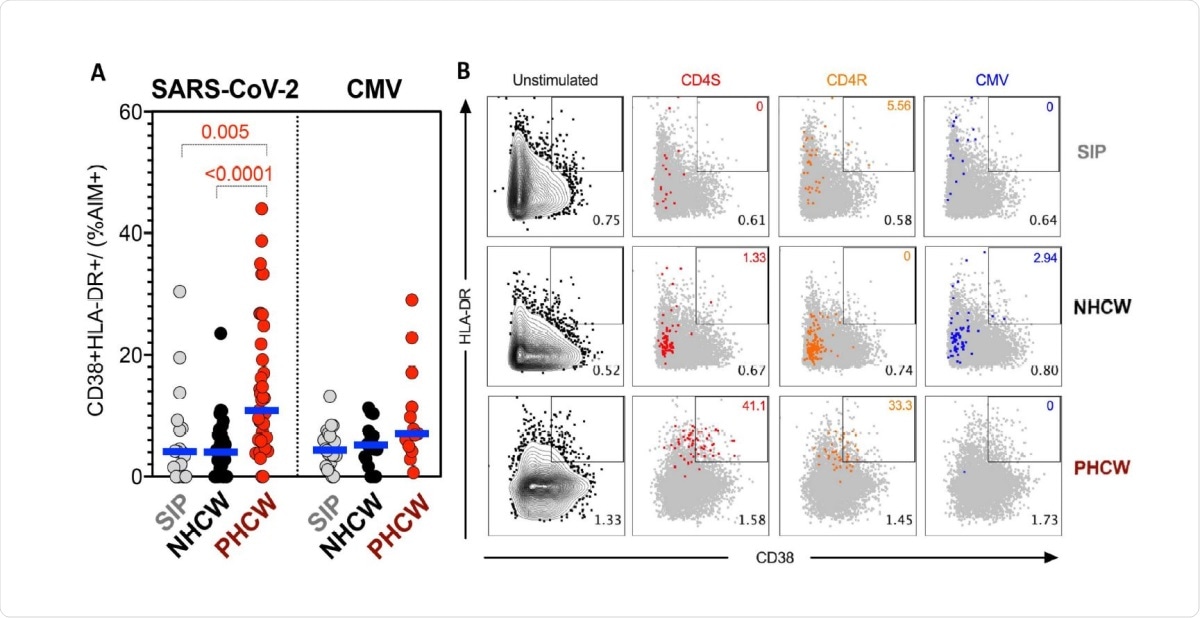
Highest PHCW reactivity in CD4+ T cell responses associated with recent infection. (A) Recently activated SARS-CoV-2-specific CD4+ T cells were measured as percentage of CD38+/HLADR+ cells in AIM+ (OX40+CD137+) CD4+ T cells after stimulation of PBMCs with peptide pools encompassing a spike only (“S”) MP and MP representing all the proteome without spike (“CD4R”). Graphs show data for specific responses against SARS-CoV-2 (both “S” and “CD4R”) the ubiquitous pathogen CMV of responses with SI>2. Each dot represents the response of an individual subject to an individual pool. Geometric mean for the 3 different groups is shown. Non-parametric Kruskal-Wallis multiple comparison test was applied. P values are shown for the statistical significant comparisons. “SIP” = Shelter In Place community volunteers (n=20). “NHCW” = SeroNegative Health Care Workers (n=33). “PHCW” = Antibody or PCR Positive Health Care Workers (n=39). (B) Representative FACS plots of HLA-DR/CD38+ cells in AIM+ (OX40+CD137+) CD4+ T cells (colored) overlapped with total HLA-DR/CD38 expression (grey) for all the cohorts in the different unstimulated or stimulated conditions. Cell frequency of HLADR/ CD38+ in AIM+ cells or total CD4+ T cells is indicated on the top and bottom right corner respectively.
CCC reactivity was lower in the positive cohorts, however. They offer several explanations, such as a global downregulation of CD4 T cell responses to CCC following SARS-CoV-2 infection, or as the result of COVID-19-associated type I interferon responses, indicative of a potential impairment of the innate immune response. Alternatively, it may be due to the expansion of SARS-CoV-2 specific T cells, that competitively reduce CCC-specific T cells.
CCC reactivity may thus contribute to, or even mediate, the cutting short of SARS-CoV-2 infection. Some studies have suggested that the presence of CD4 or CD8 T cells that cross-react with CCC might be able to terminate the infection rapidly without the development of severe disease.
As further precision is achieved in differentiating CCC vs SARS-CoV-2 reactivity, it may be possible to better understand protective and specific T cell responses to the novel coronavirus.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Antunes, R. da S. et al. (2021). Differential T cell reactivity to seasonal coronaviruses and SARS-CoV-2 in community and health care workers. medRxiv preprint. doi: https://doi.org/10.1101/2021.01.12.21249683,https://www.medrxiv.org/content/10.1101/2021.01.12.21249683v1
- Peer reviewed and published scientific report.
Silva Antunes, Ricardo da, Suresh Pallikkuth, Erin Williams, Esther Dawen Yu, Jose Mateus, Lorenzo Quiambao, Eric Wang, et al. 2021. “Differential T-Cell Reactivity to Endemic Coronaviruses and SARS-CoV-2 in Community and Health Care Workers.” The Journal of Infectious Diseases 224 (1): 70–80. https://doi.org/10.1093/infdis/jiab176. https://academic.oup.com/jid/article/224/1/70/6209393.