Alison Edelman, MD, MPH, Professor of Obstetrics and Gynecology in the Oregon Health & Science University School of Medicine. I am a clinician-researcher at an academic hospital and medical school – which means I see patients, am involved in research and teach/educate.
This research is the direct result of public interest in the relationship between COVID vaccines and menstrual health, their interest in menstrual cycle tracking, and our interest in providing high-quality evidence to help answer a really important question.
The menstrual cycle typically varies month to month for women. Why is this?
Correct, menstrual cycles fluctuate from month to month and throughout an individual’s lifetime. Our bodies are not perfect timekeepers but they do a pretty good job considering the complex system the human body is! Small changes to the length of a person’s menstrual cycle are common and should not be cause for concern. There are multiple reasons why the length of a person’s cycle may change, including stress, the use of contraception, changes to diet or exercise, age, medical conditions, etc. However, if a person experiences noticeable changes over the course of three or more months or a change in overall cycle length that exceeds eight days, they should consider consulting their care provider.
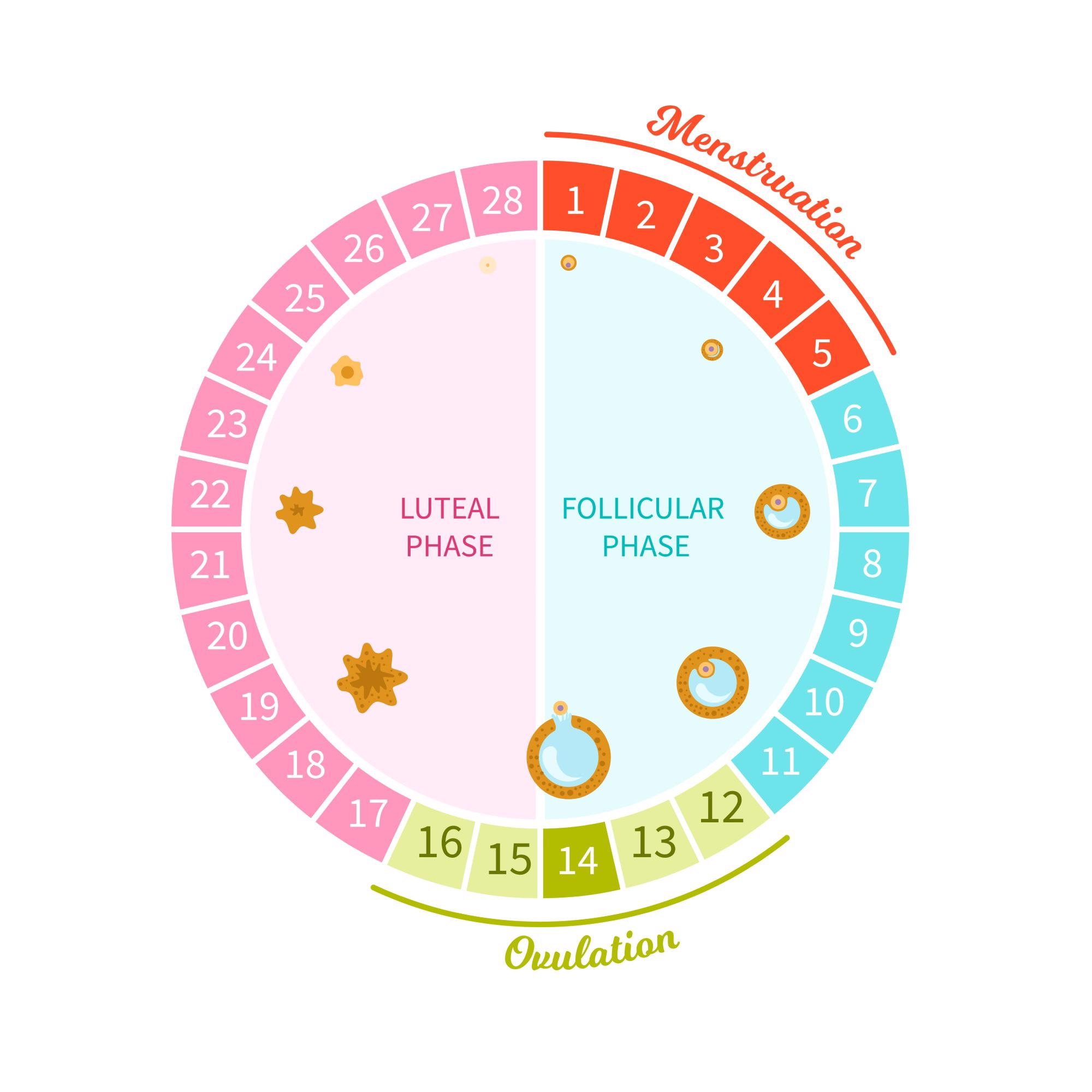
Image Credit: Igdeeva Alena/Shutterstock.com
Despite millions of people worldwide having their COVID-19 vaccinations, little research has been carried out into the effects the vaccinations have on the menstrual cycle. Why is this and why is it important to investigate all potential side effects?
Menstrual health has been both understudied and under-recognized as an outcome of importance in scientific research overall, and particularly in terms of vaccine trials. Menstrual health is clearly an important patient-reported outcome as evidenced by the number of individuals who closely track their periods but menstruation is also considered a ‘vital sign’ or an overt sign of health, well-being, and also future fertility. As an important sex- and gender-specific issue, I believe it should be included in future research and vaccine trials.
Can you describe how you carried out your latest research into COVID-19 vaccinations and the menstrual cycle? What did you discover?
Our study analyzed the menstrual tracking data of nearly 4,000 U.S.-based individuals who use an FDA-cleared birth control app called Natural Cycles. All participants consented to share their de-identified data with the study. For this analysis, we analyzed data from those who at baseline had clinically normal menstrual cycles (pre-vaccine or if unvaccinated, had cycles over a similar time). They were either unvaccinated or vaccinated with Pfizer, Moderna, or Johnson & Johnson COVID-19 vaccine.
The analysis found that, at a population level, people who menstruate may experience a small, but temporary, change to the length of their menstrual cycle after getting a COVID-19 vaccine.
The average change in length is less than one day during the menstrual cycle when a shot was given. People who receive two doses of a vaccine during one menstrual cycle, may have a two-day change.
This means that the time between a person’s first day of menses, or bleeding, until their next first day of menses may be slightly longer than what they consider “normal”. For instance, a person used to a 28-day cycle may experience a 29-day cycle after getting the vaccine, meaning their ‘period’ (menses) may begin a day later.
It is important to note that we did not find any changes to the length of menses ( bleeding period). Additionally, the cycle length variances appear to resolve quickly – as soon as the next cycle when the vaccine is not given.
Why are your study results reassuring for women? How can we use this study to help educate women on what to expect from their COVID-19 vaccination?
While the identified changes to menstrual cycle length, overall, do not rise to the level of ‘clinical significance’ defined as 8 days or greater by professional societies; on a personal level, any noticeable change to a person’s cycle – whether it be related to vaccination or other environmental stressors – can indeed feel significant especially if unexpected. While reassuring, the study findings may also be validating to individuals who experienced menstrual disruption following vaccination. As a clinician, I now have information that I can provide to patients about what to expect and have them prepared for this possibility so that they do not need to worry.
If menstrual disruption is the only reason an individual is not considering COVID-19 vaccination, this research should be reassuring that potential changes are minimal, appear to be temporary – and in alignment with other research specific to fertility and pregnancy – do not appear to cause long-term health or reproductive impacts.
Image Credit: Studio Romantic/Shutterstock.com
Were there any limitations to your latest research? If so, what were they?
Users of the menstrual cycle tracking applications, like Natural Cycles, are not necessarily reflective of a general population. We know in this dataset that this population is more college-educated, white, and has a lower BMI than the national distribution. BMI is a factor that can influence menstrual cycle variability; however, that may have been offset by the inclusion criteria for our dataset (needing 3 cycles pre-vaccine that met the criteria for normal cycle length).
We also know that many more questions exist regarding menstrual cycles and vaccines. Statistically, in order to maintain the integrity of the analyses for big population studies like this one, we have to limit the number of questions we ask at one time. We are working on that next set of questions in hopes of getting more answers out there.
About Professor Alison Edelman
Alison Edelman, M.D., M.P.H. is a Professor of Obstetrics and Gynecology and the Director of the OHSU Complex Family Planning Section and Fellowship. She is Lead Editor of the Cochrane Fertility Regulation Group, newly housed at OHSU. She is a ‘lifer’ at OHSU having completed all of her medical training at OHSU including a fellowship in family planning (2003). As part of her fellowship, she completed a Master’s in Public Health at UW, Seattle (2003)..jpg)
Dr. Edelman is an active clinician-scientist with independent research funding. She also serves as a global technical consultant, adapting global evidence to local context and resource availability, supporting sexual and reproductive health training and programming and the development of government guidelines and their implementation as well as designing and performing research.
She has several additional leadership positions in the field including the Principal Investigator of OHSU’s NIH/NICHD-funded Contraceptive Clinical Trials Network site, Associate Editor of the journal, Contraception, member of the expert working group for the Centers for Disease Control Medical Eligibility Criteria (MEC) for Contraceptive Use as well as the Selective Practice Guidelines for Contraceptive Use and a member of the World Health Organization’s Global Guidance Group (GSG) for the MEC. She is currently the Chair of American College of Obstetrics and Gynecology (ACOG) Committee on Gynecology Practice Bulletins and part of the ACOG’s COVID-19 Gynecology Guidance group. Dr. Edelman provides Obgyn care at the Center for Women’s Health, with a focus on family planning.
Dr. Edelman’s research has focused on several significant research gaps in family planning with the ultimate goal of improving the contraceptive experience for women and users of contraception. She has investigated the interaction between body weight and contraception, the development of new contraceptive methods, improving pain control with office-based gynecologic procedures and performing and supporting the generation of Cochrane systematic reviews. Her research has been nationally recognized with the American Society for Emergency Contraception ‘The Charlotte Ellertson Innovation Award’ (2017) and Contraception’s 2019 Daniel R. Mishell, Jr MD Outstanding Article Award.
Her current research includes the NIH/NICHD Contraceptive Clinical Trials Network Site. OHSU as well as 12 other sites across the country are part of NICHD’s mission to develop protocols and conduct clinical trials that will assist in development of new contraceptive methods in order to provide safe and effective contraception.
Another project is Improving emergency contraceptive effectiveness. Individuals of higher body mass index (BMI) are more likely than their peers with a lower BMI to experience failure of their emergency contraceptive. We are performing detailed drug level and ovarian monitoring in women to determine why this is occurring and how to fix it.