Vaccines are among the most economical, successful, and safe public-health strategies for infectious disease prevention. However, increased vaccine apprehension has resulted in lower vaccination coverage in the general public, particularly among children.
While coronavirus disease 2019 (COVID-19) can have serious health repercussions for children, the public still has reservations regarding pediatric vaccination. The SARS-CoV-2 pediatric vaccination effort faces a critical challenge from parental hesitation. As a result, while developing health communication methods, it is vital to consider their viewpoints. Furthermore, the opinions of other adults on this subject are worth contemplating since social media now has a massive impact on vaccination decisions. Appeals to the individual and social advantages of COVID-19 vaccination have positively influenced the attitude of grownups, whereas their efficacy in pediatric vaccination is not known.
About the study
In the present study, the researchers assessed whether the perceptions of the adults on COVID-19 childhood vaccination could be altered by conducting surveys involving two sets of information on 3,524 Italian subjects and 3,066 volunteers from the United Kingdom (UK).
Respondents were randomly allocated to one of three messages: 1) a risk message emphasizing the hazards of SARS-CoV-2 infection in children, 2) a herd immunity message underscoring the community advantages of child vaccination, or 3) a control message with a brief notice on the vaccination program. On a 0 to 100 scale, the likelihood of subjects endorsing COVID-19 childhood vaccination was rated.
The comparison of the UK and Italy was fascinating since they began their childhood vaccination programs on different dates. The ramifications may be especially significant given that the UK just started distributing vaccines to children aged five to 11 in April 2022. Gender, parental status, age, occupational status, and educational qualification were among the socio-demographic factors obtained in the surveys. In addition, details about SARS-CoV-2 vaccination history, most regarded data sources about COVID-19 vaccines, and reasons for pediatric vaccine hesitation of respondents were also collected.
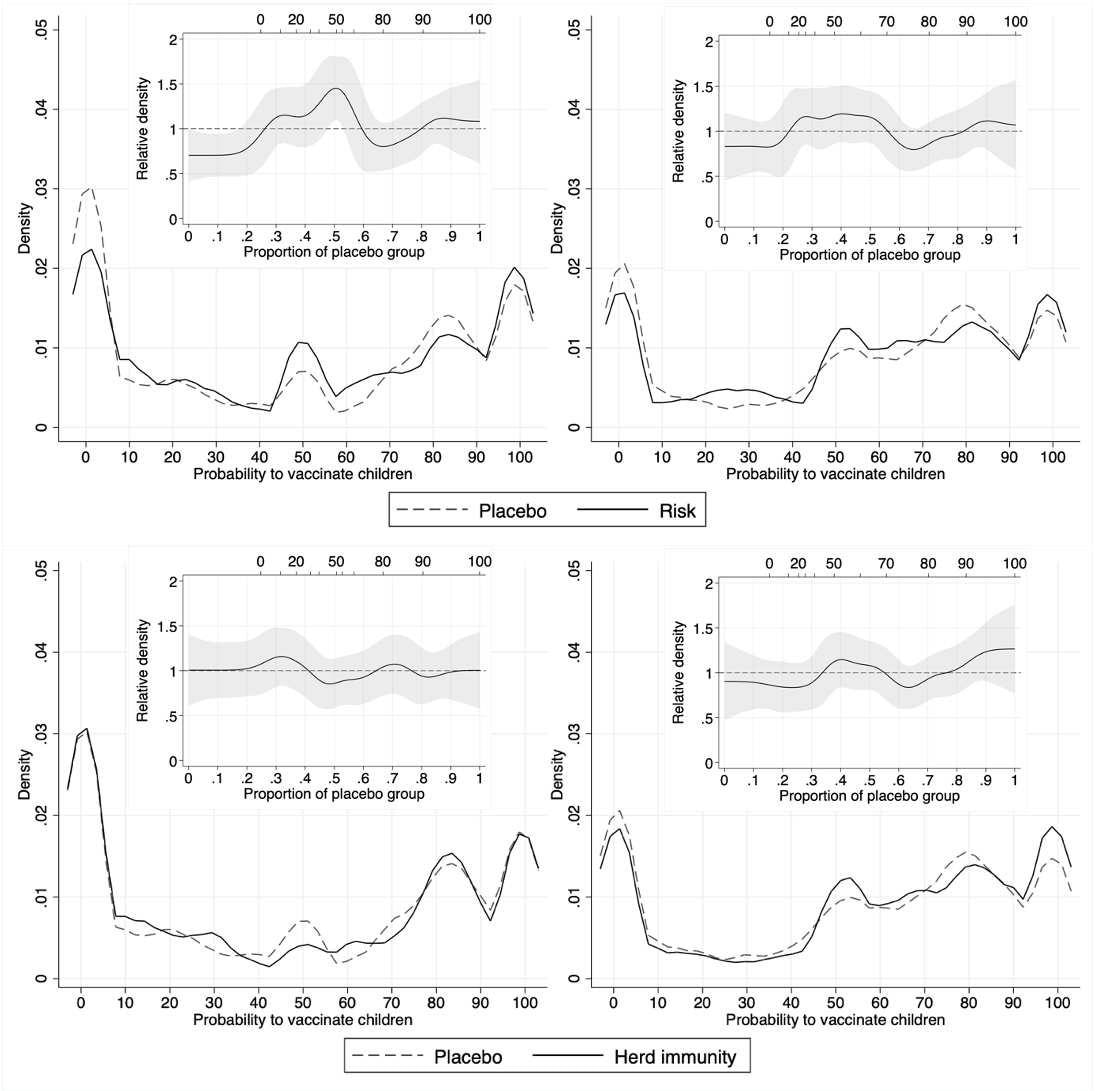
Kernel empirical and nested RD plots of vaccination intentions among parents in Italy (left) and UK (right), comparing risk-treated and controls (top), herd-immunity-treated and controls (bottom). Estimates of RD are presented with 95% CI.
Results and discussions
The study results showed that both interventions, i.e., a risk message and herd immunity message on COVID-19 childhood vaccination, affected the opinions of the participants generally. Nevertheless, the transition was only toward neutral attitudes for parents, whereas non-parents adopted vaccine-positive viewpoints. This finding implies that parents' beliefs were firmer and less impacted by a one-time information delivery, suggesting that parents may have established a stronger opinion on the issue than those without children under the age of 18. Furthermore, the results were consistent across Italy and the UK.
The authors demonstrated that the risk treatment message lowered the number of highly anti-vaccination Italian parents by around 29.6% while boosting the percentage of neutral parents to approximately 45%. Instead, the herd immunity therapy message was only successful among non-parents, culminating in lower fractions of people opposed to pediatric vaccination and higher percentages of those who support it by about 20%.
As noted by the researchers, the timing of vaccination policies did not affect acceptance of COVID-19 childhood vaccination as it was just beginning in Italy, while in the UK, it was just being discussed.
On COVID-19 vaccine-associated information, 83% of British and 70% of Italian parents mostly trusted health specialists, such as local pharmacies, general practitioners, public health experts, and local health care institutions. Thus, health practitioners should be enlisted to advise parents about the dangers of their children developing SARS-CoV-2 infection to maximize the impact of a risk treatment message.
Childhood vaccine apprehension was motivated mainly by concerns about vaccine adverse effects. According to the scientists, this was more prominent among parents than non-parents. Indeed, the worry of unfavorable health implications motivated vaccination hesitation among 37% of Italian parents and 39% of UK parents compared to 30% and 29% of non-parents in the UK and Italy, respectively.
Conclusions
To summarize, the study findings demonstrated that distinct worries might arise when considering the possibility of SARS-CoV-2 vaccination for own child versus children in general. All responders in the UK and Italy responded favorably to messages concerning the hazards of COVID-19 to children. Additionally, non-parents were influenced by the prosocial appeal, while views of parents were unaffected. Moreover, the authors stated that more studies were required to investigate the impact of information treatments clarifying misconceptions regarding the risks of SARS-CoV-2 vaccination in children.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Alessia Melegaro, Chiara Chiavenna, Laura Pasqua et al. Personal risk or societal benefit? Investigating adults' support for COVID-19 childhood vaccination, 18 May 2022, PREPRINT (Version 1) available at Research Square [https://doi.org/10.21203/rs.3.rs-1609643/v1], https://www.researchsquare.com/article/rs-1609643/v1
- Peer reviewed and published scientific report.
Chiavenna, Chiara, Laura P. Leone, Alessia Melegaro, Tiziano Rotesi, Scott E. Bokemper, Elliott E. Paintsil, Amyn A. Malik, et al. 2023. “Personal Risk or Societal Benefit? Investigating Adults’ Support for COVID-19 Childhood Vaccination.” Vaccine, May. https://doi.org/10.1016/j.vaccine.2023.05.017. https://www.sciencedirect.com/science/article/pii/S0264410X23005443.