Global public health continues to be threatened by zoonotic and pandemic influenza. The virus has a high mutation rate, owing to its interspecies transmissibility and adaptability. The influenza A virus has diverse avian and animal origins with several viral subtypes; those in aquatic birds differ in neuraminidase (N1-N9) and hemagglutinin (H1 – H16).
Subtypes H6 and H9 of the influenza A virus infect game birds like pheasants, quails, and domestic chickens. H9N2 viruses containing acquired neuraminidase (NA) and hemagglutinin (HA) genes generate H5N1, H5N6, H10N8, and H7N9 viruses that may infect poultry and thereby threaten another zoonotic outbreak and onset of a pandemic.
The study
A recent virological study published in the Emerging Infectious Diseases journal identified the novel influenza A virus (H3N8) in live poultry in Hong Kong, genetically similar to the zoonotic H3N8 viruses reported in mainland China with its origin akin to H5N1, H5N6, H10N8, and H7N9 viruses. In addition, a risk assessment approach has been described to estimate the effects of human age stratified immunity in a population.
 Study: Novel Zoonotic Avian Influenza Virus A(H3N8) Virus in Chicken, Hong Kong, China. Image Credit: krugloff / Shutterstock
Study: Novel Zoonotic Avian Influenza Virus A(H3N8) Virus in Chicken, Hong Kong, China. Image Credit: krugloff / Shutterstock
Results
The study entailed virologic surveillance between December 2021 and March 2022. Influenza A(H3N8) virus was identified in samples collected from four farms (A-D) in the study duration. All four farms were serologically positive for past H3N8 infections. Consequently, the caretakers were advised to disinfect their farms thoroughly and strengthen their biosecurity measures. Follow-up testing was conducted in May 2022 and again at the end of June 2022; all four farms tested negative for the influenza A(H3N8) virus.
Between January and June 2022, many swab samples of drinking water from poultry cages, defeathering machines, fecal droppings, and chopping boards were sampled from stalls and markets. One swab specimen from a defeathering machine and one chopping board from two different live poultry markets were H3N8 positive. DNA barcoding determined that the virus detected in the swab specimens originated from domestic chickens.
Full-genome phylogenetic analysis of the isolated influenza A (H3N8) viruses revealed a close relationship between H3N8 viruses isolated from farms and poultry markets and an H3N8 viral strain implicated in the zoonotic outbreak in mainland China. The polymerase acidic, nonstructural protein, NA, polymerase basic 1 and 2, and M gene segments were identical to the G57 sublineage of the H9N2 virus found in mainland China. The sequences of the HA gene were derived from the Eurasian avian H3 lineage, which is found in wild birds and ducks.
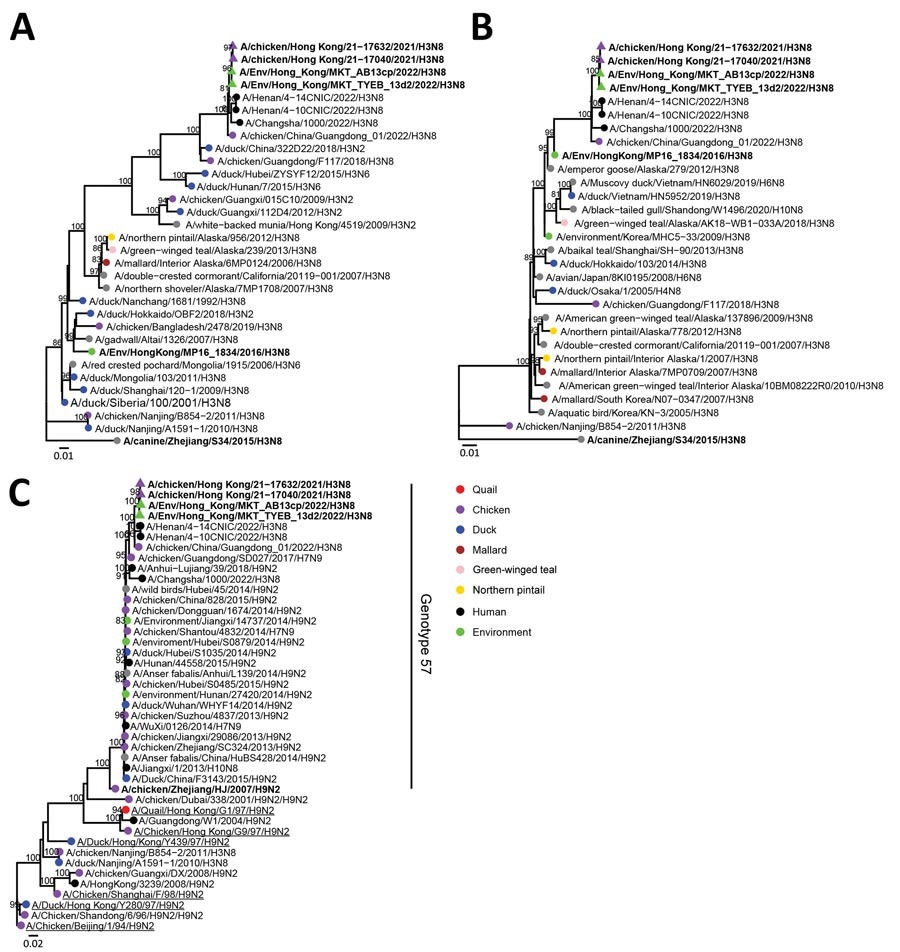
Phylogenetic analysis of influenza A(H3N8) viruses isolated from chicken farms, live poultry markets, and the Mai Po Wetlands, Hong Kong, China (bold). A) Hemagglutinin gene segment; B) neuraminidase gene segment; C) polymerase basic 2 gene segments. Strains were analyzed with other relevant virus sequence data available in public databases (accession numbers in Appendix Table). Trees were generated by using IQ-tree with the general time reversible plus gamma model. Bootstrap values >80% are shown. Scale bars indicate estimated genetic distances.
NA gene sequence of H3N8 in poultry A was from the North American lineage. On the other hand, the N8 NA sequence detected by DNA barcoding of a virus from a fecal specimen in Hong Kong in 2018 was derived from Northern pintail duck or Anas acuta.
Two H3N8 viruses from fecal dropping samples from Hong Kong were related to the Northern shoveler duck or Anus clypeata; these strains were unrelated to chicken H3N8 in all gene segments. A similarity was found between the N8 gene segment sequence and the H3N8 virus from other aquatic birds in mainland China. Except for the N8 NA gene segment, no other gene segments detected in poultry Influenza A(H3N8) virus were from the wild bird H3N8 virus from Hong Kong.
Referencing was carried out of HI titers with the World Health Organization (WHO) antiserum to A/Switzerland/8060/2017 against A/chicken/MKT-AB13cp/2020 H3N8 virus and on comparing with the A/Switzerland/8060/2017 homologous virus. Limited antigenic cross-reactivity was noted among the novel avian influenza A(H3N8) virus and the seasonal human H3N2 viruses.
The overall seroprevalence was insignificant in human serum samples stratified by age. In contrast, high seroprevalence in the same serum samples was observed in a recent seasonal influenza A (H3N8) virus in humans.
Herd immunity is an important parameter when assessing any pandemic threat of zoonotic origin.
Conclusion
According to the study, the influenza (AH3N8) virus has been detected among poultry in Hong Kong. This virus might pose a serious zoonotic and pandemic threat. Therefore, it is recommended that poultry surveillance for this virus be enhanced, a comprehensive risk assessment be conducted, and pandemic seed vaccine strains be prepared if necessary.