By Jeyashree Sundaram (MBA)
Barrett’s esophagus (BE) which is a condition defined by the unusual growth of esophagus cells, in rare occasions, might grow into a cancer known as esophageal adenocarcinoma. The major cause of BE is GERD and acid reflux. Risk of arising adenocarcinoma is greater by 30-125 times in BE affected humans than in humans who doesn’t have BE.
In case of BE, the diagnosis and treatment methods available are efficient enough; and the condition itself is not considered a harmful disease. But, if it converts into cancerous stage, it attains complexity. Therefore, researches in this field targets for preventing the progression of BE, for controlling the symptoms, and for providing better survival to the patient.
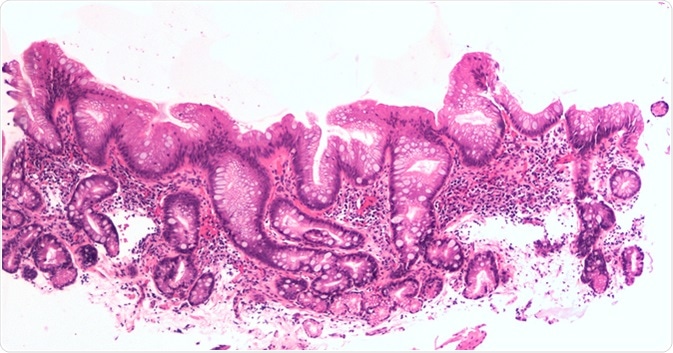
Intestinal metaplasia of the esophagus, aka Barrett's, is a response to injury due to acid reflux. Image Credit: David Litman / Shutterstock
Research on Genes
In 2012, a study was conducted to examine the DNA methylation of vimentin gene in the neoplasm of the upper part of the gastrointestinal (GI) tract. Using a real-time quantitative methylation particular polymerase chain reaction (PCR) assay, researchers tested the archival samples of gastric neoplasia and esophagus for the methylation of vimentin. They found that the abnormal DNA methylation of vimentin gene is high in this type of neoplasm.
Finally, they proved by cytology brushings test that vimentin DNA methylation noticeable in the case of BE is 100%, but it was beyond control. These results recommend that the biomarker of the upper part of GI tract is vimentin methylation. These findings are expected to be helpful in future studies of BE.
Another study was carried out for finding the specific genes that methylate abnormally in BE. For this, a group of researchers conducted a genome-wide approach for DNA methylation during 2016. They analyzed the specimens of BE and stomach and then compared each specimen’s level of methylation at around 485,000CpG sites inside the DNA samples.
Pyrosequencing assays are used for the approval of results and develop methylight assays for identifying the alleles of DNA methylated in endoscopic brushings. Thus, the result showed that the gene ZNF793 and B3GAT2 are unusually methylated highly in BE and these genes methylated grade were used to identify BE in sample tissues.
Mayo Clinic Researchers FISH for Answers about Barrett's Esophagus
Research on Cellular Process
A study was carried out in 2015 for identifying the role of autophagy in a BE patient. At present, this research is poorly studied. In this, the level of autophagy in the cell lines of a BE patient, a transgenic BE mouse model, and biopsy of esophageal adenocarcinoma (EAC) are defined.
The researchers found that the level of autophagic vesicles (AVs) is high in the non-dysplastic BE person, whereas it is decreased in the usual BE cells of dysplastic and squamous cells, and is not present in EAC.
They also found that the level of AVs is highly increased in BE humans than EAC or usual squamous and recommended that functions of autophagy after damage by acid reflux improves the survival of cell. Thus, autophagy can play an important role in the progression and pathogenesis of BE.
Research on Treatment Methods
In May 2016, an article was published by ten authors on the development of treatment for BE. In this article, they state that BE can develop into cancer due to many reasons; this is identified by new testing methods (pH monitoring and specifically multichannel within lumen impedance) that aid to get better knowledge of the pathophysiology of GERD along with its complications.
Such findings are more helpful in predicting the progression of BE neoplasm. Finally, they show a new treatment choice for BE and EAC termed endoscopic elimination, which is more effective for patients. This information gives more knowledge on the mechanical connection between GERD and BE.
Research on Hereditary Factors
In 2017, research is ongoing for predicting BE within the family; authors have come together to examine a clinical model from 92 multiplex pedigrees of BE and 787 individually ascertained pedigrees of BE, fitting a model to a multivariate logistics that includes medical risk factors and family history.
The risk factors included in the model are education level, smoking, age, parental status, sex, regurgitation frequency, heartburn frequency, and using acid suppressant. Using the training dataset and separate validating dataset of the 643 multiplex pedigrees with BE, the exact prediction was determined. Eventually, the result showed that BE risk can be found with the help of family information; the possibility of predicting risk for individuals who are not a member of the family without the suggestion from any family relation was also identified.
Research Related to Pathogenesis
During 2007, a few authors combined to predict the risk of high-grade dysplasia and esophageal adenocarcinoma from BE. These were prospectively evaluated by the cases of nearly 325 BE patients; they were subjected to endoscopic biopsies. Among them, 269 patients were continuously followed up with one or many endoscopies by a strong platform for the analysis of heterozygosity loss (LOH) with baseline 17p (p53). Finally, it was found that LOH is the predictor of BE progression and the outcome is an increase in esophageal adenocarcinoma and high-grade dysplasia.
Further Reading
Last Updated: Feb 26, 2019