Introduction
Overview of sirtuin biology
Role in metabolism
Role in aging and longevity
Mitochondrial function and oxidative stress
Therapeutic potential and limitations
Clinical and research significance
References
Further reading
Sirtuins are NAD+-dependent enzymes that regulate metabolism, oxidative stress, mitochondrial function, and genomic stability to influence aging and healthspan. Their tissue-specific activities have made them promising but complex therapeutic targets for metabolic, cardiovascular, neurodegenerative, and age-related diseases.
 Image Credit: Ficta Stock / Shutterstock.com
Image Credit: Ficta Stock / Shutterstock.com
Introduction
The biology of aging is largely influenced by cellular responses to metabolic and environmental stress. Sirtuins contribute to this adaptability through their involvement in metabolic regulation, stress signaling, and genome maintenance. Because sirtuin activity depends on NAD+, these enzymes help couple the cell’s energetic state to transcription, mitochondrial function, DNA repair, inflammation, and stress resistance.1,4
Overview of sirtuin biology
Sirtuins, or SIRT, are a family of highly conserved class III histone deacetylases that use nicotinamide adenine dinucleotide (NAD+) to participate in diverse biological processes.2 Their dependence on NAD+ implicates sirtuins in cellular responses to nutrient availability, environmental stress, metabolic regulation, and circadian rhythms, as well as in the pathophysiology of geriatric diseases.1,5
Mammalian sirtuins are distributed across different cellular compartments to manage localized signaling and metabolic demands. Sirtuin homologs SIRT1, SIRT6, and SIRT7 are predominantly distributed in the nucleus, where they regulate gene expression and maintain genomic stability.6,7
Although SIRT2 is localized in the cytoplasm, it can translocate to the nucleus during specific phases of the cell cycle to influence mitosis.2,7 SIRT3, SIRT4, and SIRT5 are located in the mitochondria, wherein they influence oxidative metabolism, energy production, and the detoxification of reactive oxygen species (ROS).1
The primary biochemical function of sirtuins involves deacetylation of lysine residues on target proteins, a process that requires NAD+ as a co-substrate and releases nicotinamide and 2'-O-acetyl-ADP-ribose.6
Beyond deacetylation, certain isoforms exhibit distinct acyl-modifying activities. For example, SIRT5 can remove malonate, glutaryl, and succinate groups from lysine residues in proteins.1 Other sirtuins like SIRT4 also possess mono-ADP-ribosyl transferase, lipoamidase, or additional deacylation activity that regulate metabolic enzymes and transcriptional regulators.7
The structural architecture of sirtuins includes an NAD+-binding pocket and zinc ribbon motif that support enzymatic stability and catalytic efficiency. Despite structural similarities, some sirtuins interact with diverse protein targets to influence transcriptional regulation, mitochondrial activity, and responses to nutrient fluctuations and environmental stress.1,5
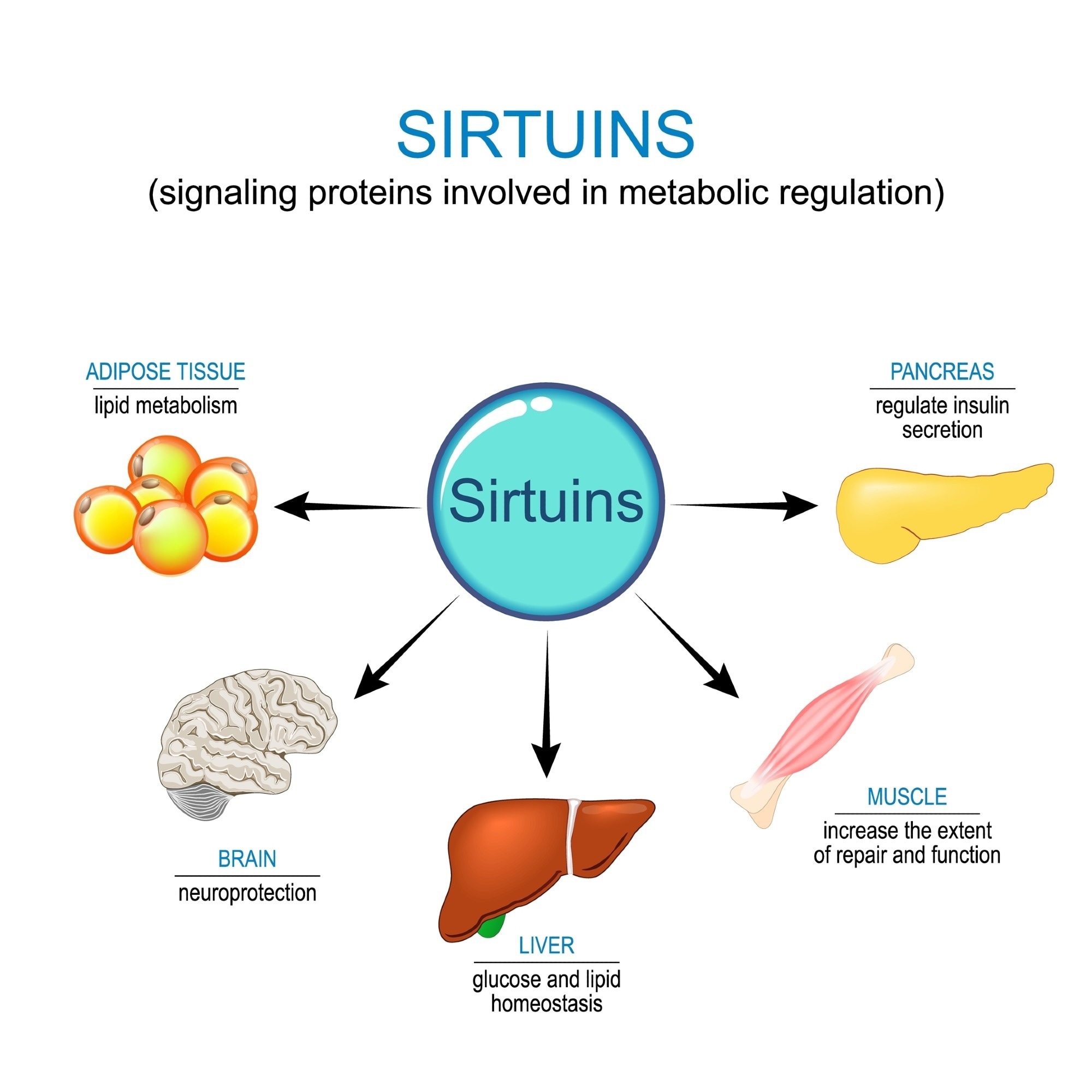 Image Credit: Designua / Shutterstock.com
Image Credit: Designua / Shutterstock.com
Sirtuins facilitate adaptive metabolic responses during periods of fasting, caloric restriction, and exercise. For example, SIRT1 acts as an autophagy modulator through the deacetylation of major transcription factors like peroxisome-activated receptor coactivator 1-α (PGC-1α) and forkhead box O3 (FoxO3) proteins, thereby enhancing fatty acid oxidation and glucose production in the liver.7,8 In skeletal muscles, SIRT1 has been associated with mitochondrial biogenesis and improved insulin sensitivity to support systemic metabolic health.7 Skeletal-muscle sirtuins also regulate glucose uptake, fatty acid oxidation, mitochondrial dynamics, autophagy, myogenesis, fiber-type switching, regeneration, and exercise adaptation, although these effects remain isoform- and context-dependent.7
The mitochondrial sirtuins SIRT3, SIRT4, and SIRT5 influence fatty acid oxidation in skeletal muscles. SIRT3 is the major mitochondrial deacetylase, as it regulates many enzymes involved in the electron transport chain and tricarboxylic acid (TCA) cycle to optimize adenosine triphosphate (ATP) production.1,8
Conversely, SIRT4 can repress certain metabolic pathways, such as amino acid-stimulated insulin secretion in pancreatic beta cells through its ADP-ribosyltransferase activity.1 SIRT5 also influences the urea cycle and glycolysis through its specific demalonylation and desuccinylation activities.3 Together, these enzymes coordinate nutrient utilization and energy expenditure across various tissues.
Role in aging and longevity
The association between sirtuins and longevity was first established in yeast. Herein, an additional copy of the Sir2 gene significantly extended yeast survival, whereas the loss of a copy was associated with shortened replicative lifespan.1
In mammals, sirtuins preserve genomic integrity and delay the onset of cellular senescence to support healthspan, although their direct role in natural lifespan extension remains debated and may vary by isoform, tissue, and experimental context.3,4 SIRT6, for example, is essential for efficient DNA double-strand break repair and telomere maintenance, with its deficiency in mice inducing a premature aging phenotype.1,8
Sirtuins also increase cellular stress resistance by activating antioxidant defense mechanisms. SIRT1 and SIRT3 regulate FoxO-dependent antioxidant pathways, while SIRT3 more directly deacetylates and activates mitochondrial superoxide dismutase 2.5,8 Rather than simply lowering oxidative damage, sirtuins may help tune ROS signaling so that moderate mitochondrial stress can trigger protective hormetic responses.8
Mitochondrial function and oxidative stress
SIRT3 directly enhances manganese superoxide dismutase (MnSOD/SOD2), the primary antioxidant enzyme within mitochondria, while SIRT1 and SIRT6 influence oxidative-stress control more indirectly through transcriptional, inflammatory, and DNA-repair pathways.1,8 SIRT4 and SIRT5 also participate in mitochondrial control by regulating metabolic enzymes involved in fatty acid oxidation and amino acid metabolism.1,7
Dysfunction in mitochondrial sirtuin pathways is closely linked to the progression of age-related metabolic and degenerative disorders. Low SIRT3 activity often leads to mitochondrial hyperacetylation, resulting in decreased metabolic efficiency and increased ROS production.7
This state of mitochondrial distress increases the risk of cardiac fibrosis, neurodegeneration, and skeletal muscle atrophy in aging populations.1,5 Conversely, maintaining robust mitochondrial sirtuin activity supports the metabolic flexibility required to adapt to various physiological stressors.7 In the heart, NAD+-dependent activation of SIRT1 and SIRT3 has been linked to reduced fibroblast activation, improved mitochondrial homeostasis, AMPK signaling, and protection against oxidative and inflammatory damage; these effects intersect with TGF-β/Smad, NF-κB, ROS, and EndoMT pathways, whereas SIRT4 may have context-dependent pro-fibrotic effects.5
 Image Credit: nobeastsofierce / Shutterstock.com
Image Credit: nobeastsofierce / Shutterstock.com
Therapeutic potential and limitations
The potential to modulate sirtuin activity for therapeutic purposes has led to the development of sirtuin-activating compounds and NAD+ precursors. Resveratrol, a natural polyphenol, has been reported to activate SIRT1 in experimental systems and to reproduce some calorie-restriction-like effects in preclinical models, although its specificity and clinical translatability remain limited.2,6
Sirtuin activity can also be modulated using NAD+ precursors, which restore declining NAD+ levels and improve mitochondrial function and metabolic regulation during aging.1,7 However, evidence in humans remains disease-specific and less definitive than preclinical findings.2,6 Nevertheless, additional studies are needed to establish the long-term safety of synthetic activators, as sirtuins like SIRT1 may exhibit context-dependent effects in cancer biology, functioning as either tumor suppressors or tumor promoters depending on cellular setting and disease stage.2,3
Clinical and research significance
In the cardiovascular system, sirtuins are being investigated for their ability to inhibit the transdifferentiation of fibroblasts into myofibroblasts, a key step in the development of myocardial fibrosis.5 Selective SIRT2 inhibition is also being explored for its potential to reduce toxic protein aggregation and neuronal injury in models of Parkinson's and Huntington's diseases.3
The emerging field of sirtuin biology highlights a fundamental link between energy metabolism and the molecular pathways of aging. Understanding how sirtuin activity can be precisely targeted in specific tissues may lead to novel interventions for chronic geriatric diseases and extension of human healthspan.1,3,5 However, because sirtuins can have isoform-specific and sometimes opposing effects, future therapies will need to define the target tissue, disease stage, dose, and duration of modulation rather than treating sirtuin activation as uniformly beneficial.1,2,4
References
- Wu, Q. J., Zhang, T. N., Chen, H. H., et al. (2022). The sirtuin family in health and disease. Signal Transduction and Targeted Therapy 7(1); 402. DOI: 10.1038/s41392-022-01257-8. https://www.nature.com/articles/s41392-022-01257-8
- Carafa, V., Rotili, D., Forgione, M., et al. (2016). Sirtuin functions and modulation: from chemistry to the clinic. Clinical Epigenetics 8(61). DOI: 10.1186/s13148-016-0224-3. https://link.springer.com/article/10.1186/s13148-016-0224-3
- Zhao, L., Cao, J., Hu, K., et al. (2020). Sirtuins and their Biological Relevance in Aging and Age-Related Diseases. Aging and Disease 11(4); 927-945. DOI: 10.14336/AD.2019.0820. https://www.aginganddisease.org/EN/10.14336/AD.2019.0820
- Houtkooper, R. H., Pirinen, E., & Auwerx, J. (2012). Sirtuins as regulators of metabolism and healthspan. Nature Reviews Molecular Cell Biology 13(4); 225-238. DOI:10.1038/nrm3293. https://www.nature.com/articles/nrm3293
- Grzeczka, A., Graczyk, S., Gong, X., et al. (2025). Aging hearts, fibrotic fears: The sirtuin connection. Biomedicine & Pharmacotherapy 193; 118882. DOI: 10.1016/j.biopha.2025.118882. https://www.sciencedirect.com/science/article/pii/S0753332225010765
- Lavu, S., Boss, O., Elliott, P. J., & Lambert, P. D. (2008). Sirtuins--novel therapeutic targets to treat age-associated diseases. Nature Reviews Drug Discovery 7(10); 841-853. DOI: 10.1038/nrd2665. https://www.nature.com/articles/nrd2665
- Gibril, B. A. A., Xiong, X., Chai, X., et al. (2024). Unlocking the Nexus of Sirtuins: A Comprehensive Review of Their Role in Skeletal Muscle Metabolism, Development, and Disorders. International Journal of Biological Sciences 20(8); 3219-3235. DOI: 10.7150/ijbs.96885. https://www.ijbs.com/v20p3219.htm
- Merksamer, P. I., Liu, Y., He, W., et al. (2013). The sirtuins, oxidative stress and aging: an emerging link. Aging 5(3); 144-150. DOI: 10.18632/aging.100544, https://www.aging-us.com/article/100544/text
Last Updated: May 12, 2026