Jun 18 2018
Researchers demonstrate new modeling framework and use it to predict efficacy of different prophylaxis strategies
A new mathematical simulation approach predicts the efficacy of pre- and post-exposure prophylaxis (PrEP) medications, which help prevent HIV infection. The framework, presented in PLOS Computational Biology by Sulav Duwal and Max von Kleist of Freie Universität Berlin and colleagues, could help streamline development of new PrEP treatments.
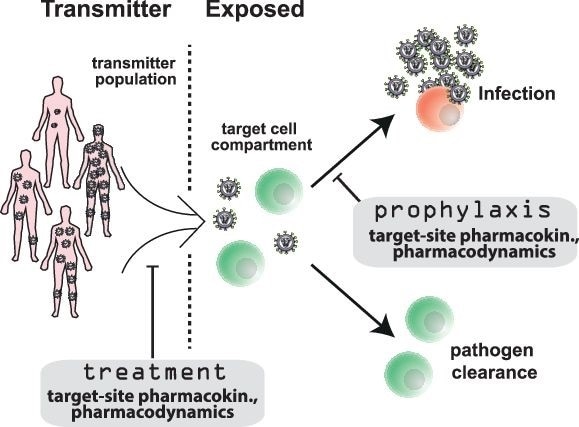
Image Credit: Max von Kleist, Sulav Duwal.
PrEP is an innovative HIV prevention method by which HIV exposed individuals take anti-HIV medication to prevent infection. PrEP in the form of a two-drug combination called Truvada can drastically decrease risk of HIV infection. However, Truvada is not perfectly effective, it is costly, and it must be taken once daily, raising adherence concerns. HIV continues to spread globally, so a hunt for new PrEP strategies—especially long-acting drug formulations that may only need to be administered monthly—is well underway.
Despite its vigor, the drive to develop new PrEP approaches is hampered by the inefficiency of clinical testing in patients. To address this challenge, Duwal and colleagues set out to build a new mathematical framework that could help researchers predict how well new PrEP drugs will work, so that the most promising drugs can be identified and optimized for clinical trials.
The new tool mathematically integrates a suite of molecular factors that affect drug efficacy, including drug activity itself and the response of the patient and virus to the treatment. Notably, it draws on recent developments in the simulation of stochastic processes to calculate drug efficacy according to drug dosing and drug activity in the body.
“Using the mathematical framework we developed, it is now possible to evaluate the clinical efficacy of any PrEP regimen prior to conducting any confirmatory trial,” says study senior author von Kleist.
The researchers used the new tool to predict that a 50mg oral dose of the drug dolutegravir is non-inferior to Truvada in pre-exposure prophylaxis, and that it may be superior to Truvada when taken after HIV exposure to prevent infection. They also used the tool to calculate the blood concentration of dolutegravir required for it to be 90 percent effective against sexual transmission of HIV; this could aid current efforts to develop long-acting dolutegravir formulations.
“The cost of PrEP is a major burden to its broad implementation, particularly in sub-Saharan Africa, which is hit hardest by the epidemic,” von Kleist says. “The next step for this research is to evaluate the potential of extremely cost-efficient regimens that are currently neglected in drug re-purposing programs for PrEP.”
Source: https://www.plos.org/