Novel Coronavirus SARS-CoV-2 Colorized scanning electron micrograph of an apoptotic cell (green) heavily infected with SARS-COV-2 virus particles (orange), isolated from a patient sample. Image at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID
SARS-CoV-2, a causative agent of the disruptive coronavirus disease (COVID-19) pandemic, is primarily seen as a respiratory pathogen; however, the evidence suggests that the virus can independently replicate in the gut, raising the likelihood of fecal-oral transmission.
This could indeed be an important issue with fecal microbiota transplantation, which is the process of transferring the stool from a healthy donor into a patient's gut in order to restore the intestinal microbiome. Currently, this approach is recommended for frequent hospital-acquired infections with Clostridioides difficile and is currently explored as an experimental approach for many other conditions.
However, despite the consensus that fetal microbiota donors should be screened for SARS-CoV-2, the optimal strategy for detecting asymptomatic carriage among donors is still elusive, while the combination of tests has not yet been appraised or compared.
Hence, the researchers from the OpenBiome (i.e., a nonprofit organization in Massachusetts, which operates a public stool bank and supports human microbiome developed research) developed a mathematical model of SARS-CoV-2 infection among fecal transplant donors that simulates the effect of diverse testing strategies.
Quantifying testing effect on the reduction of SARS-CoV-2 transmission
In short, these scientists built an abstract model of fecal microbiota transplantation donors, simulating their donation schedule, the infection incidence with SARS-CoV-2, as well as COVID-19 disease course.
Various screening strategies were layered on top of these simulations – taking into account the suboptimal sensitivity and specificity of each test – in order to estimate how many virus-negative and virus-positive donations would be appropriately or undesirably released, respectively.
Accordingly, the devised model presented two outcomes: the number of "true negative," virus-negative donations released, and the number of "false negative," virus-positive donations released.
Consequently, a desirable screening approach would release many virus-negative donations and only a handful or no virus-positive donations; on the other hand, a poor strategy would needlessly destroy a wide array of virus-negative donations or release a plethora of virus-positive donations.
"The model quantifies the effect of more stringent testing on the desirable reduction in potentially infectious, virus-positive donations processed into fecal microbiota transplantation material and released for use, as well as the undesirable reduction in virus-negative donations released", study authors summarize their methodological approach.
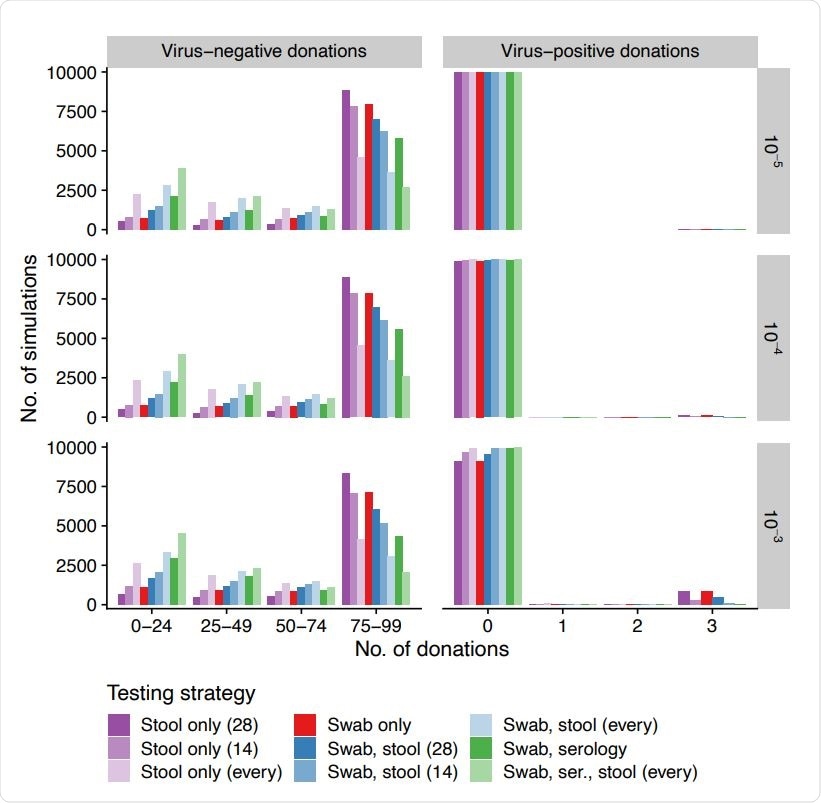
Number of virus-negative and -positive donations released (columns, x-axis) across simulations (y-axis) for different daily incidences (rows, infections per person per day) when using different testing strategies (colors). Swabs are always at 14-day intervals and serology is always at 60-day intervals. Stool tests are performed at 14-day intervals, 28-day intervals, or for every donation.
Key parameters: donation interval, test specifics and viral incidence
The number of virus-positive and virus-negative donations released differed over simulations and were contingent on testing strategy and infection incidence. In short, the most sensitive strategies were also the least specific ones.
"In general, the more sensitive strategies released fewer virus-positive donations but also removed donors early due to false positives and therefore released fewer virus-negative donations per donor," further clarified study authors.
Moreover, the risk difference of released virus-positive material when the most and least stringent strategies were compared was equivalent to the effect of a 100-fold change in daily SARS-CoV-2 incidence.
When sensitivity analysis was pursued, the parameters that were strongly associated with the two outcomes mentioned above were donation interval (i.e., longer interval yielded fewer virus-negative donations), the characteristics of the three tests (i.e., more virus-negative donations released with the use of specific tests), and SARS-CoV-2 incidence (i.e., higher incidence correlated with more virus-positive donations released).
Ascertaining the value of the model
The strength of this analysis lies in its quantitative treatment of a burning clinical question. Still, all quantitative predictions made by the model should be viewed as guidelines to clinical reasoning rather than clear-cut and unambiguous forecasts.
Furthermore, verifying this model may prove rather challenging, since the possibility of SARS-CoV-2 fecal-oral transmission has not been confirmed, and there is no "gold standard" for detecting this viral agent in stool specimens. Also, many assumptions in the model may no longer be applicable.
"Although these results are encouraging, we again caution that they depend on a number of assumptions about testing quality and SARS-CoV-2 epidemiology that will be refined in the coming months", say study authors.
Nevertheless, the method is indeed valuable in appraising the transmission risks in this ongoing pandemic. Besides, this approach may be utilized as a template to evaluate testing strategies for other pathogenic microorganisms or human-derived therapies beyond fecal microbiota transplantation.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources