A recent Brazilian study demonstrates that hyperimmune globulin formulations generated against the recombinant spike protein of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) provide significantly high antibody titer with highly neutralizing capacity. The study is currently available on the bioRxiv* preprint server.
The entire scientific community is currently aiming at finding solutions to stop the worldwide pandemic of coronavirus disease 2019 (COVID-19) caused by SARS-CoV-2. Although several vaccines are already undergoing human clinical trials, there is still a long way to go before it is actually feasible to vaccinate the entire world population. Besides routine antiviral treatments, medical professionals are relying on passive immunization strategies using plasma samples (convalescent plasma) of people who have recovered from COVID-19. However, this alternative intervention is not always effective in producing high neutralizing antibody titers in COVID-19 patients.
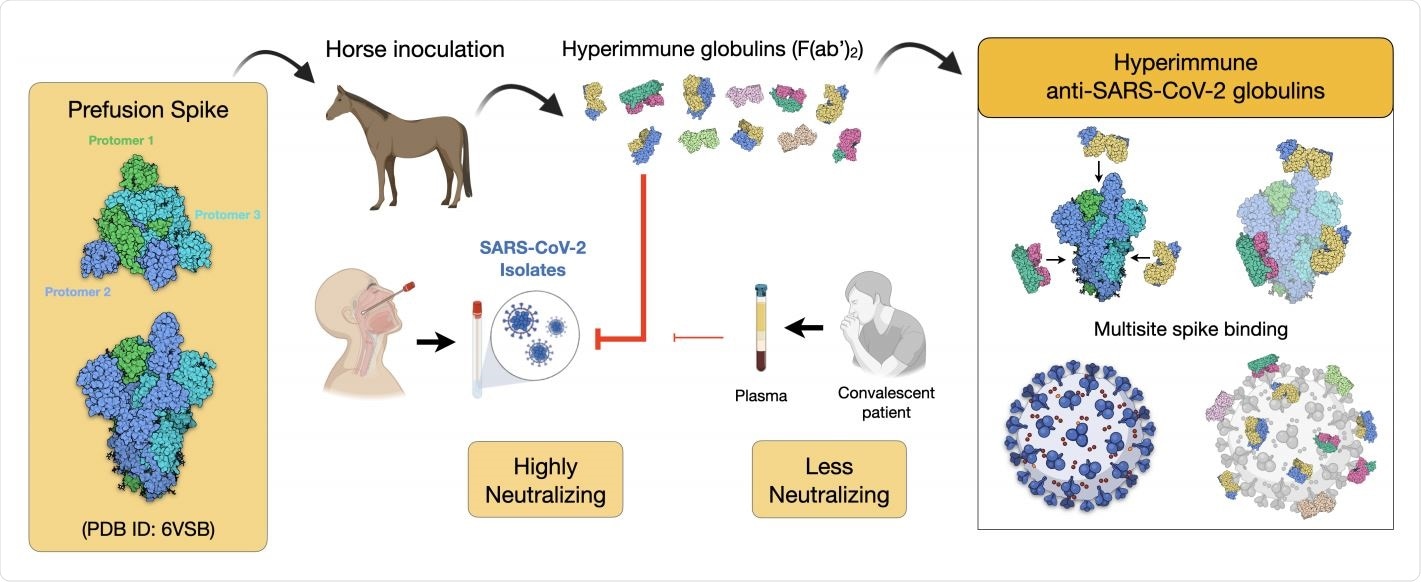
Scheme of the immunization strategy and anti-SARS-CoV-2 hyperimmune globulin production. Recombinant prefusion trimeric S protein is used to inoculate horses and to produce hyperimmune F(ab`)2 concentrate. The equine antibody preparation presented a much higher capacity to neutralize a SARS-CoV-2 isolate than human convalescent plasma. One advantage of using the full-length recombinant spike trimer is the production of antibodies against different antigenic segments of the viral protein. This strategy may result in more efficient neutralizing capacity than antibodies produced against isolated fragments of the spike protein, such as the receptor binding domain (RBD).

 *Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
Reasons for choosing hyperimmune globulins
Previous studies searching for interventions to treat pathogen-induced diseases like rabies and tetanus have found that hyperimmune serum developed in horses can serve as an effective alternative to treat these conditions. Similarly, horse hyperimmune serum samples have been shown to produce neutralizing antibodies against beta coronaviruses, such as SARS-CoV-1 and Middle East Respiratory Syndrome coronavirus.
Current study design
In the current study, the researchers used a recombinant protein, which is the trimeric form of the complete spike protein of SARS-CoV-2 composed of both S1 and S2 subunits. For getting a large quantity of high-quality neutralizing antibodies, the recombinant protein was produced in a horse in its stabilized perfusion conformation. As mentioned by the researchers, this recombinant protein is safe to use and rapidly induces adaptive immunity in horses. The fully folded trimeric spike protein with perfusion conformation has higher immunogenicity than smaller protein fragments, such as the receptor-binding domain of the spike protein. Moreover, it is very safe to use recombinant proteins rather than the whole virus for inoculation.
For the production of neutralizing antibodies against SARS-CoV-2, the researchers subcutaneously inoculated 5 horses with the recombinant protein for 6 weeks (six injections given at an interval of one week). Of these horses, 4 showed strong immune responses. By making an average of all five horses, the researchers observed antibody (immunoglobin G; IgG) titers of more than 1: 1,000,000 and neutralizing titers (PRNT90) of 1: 14,604. Next, the researchers compared these titers with convalescent plasma antibody titers of three previously SARS-CoV-2 infected individuals and observed that serum samples of immunized horses have almost 140-fold higher neutralizing titers.
Overall, the researchers used only 1.2 mg of the recombinant protein per horse and observed that for 90% neutralization, the average titer was 1: 14,604 if the horse with low immune response is included; however, the titer became 1: 18,000 if the low-responding horse is not included.
Given the fact that horse antisera can induce potential side-effects (antibody-dependent enhancement of infection), the researchers removed the Fc fragments of antibodies by digesting IgG molecules with pepsin and subsequently purifying them by fractional salt precipitation. The neutralizing titers of purified F(ab')2 fragments were found to be 150-fold higher than that of the convalescent plasma samples.
Overall, the researchers obtained a high ratio of neutralizing to binding antibodies, which might be because of the high antigenic efficacy of trimeric spike protein in its perfusion conformation. Moreover, Montanide ISA 50V used as an adjuvant in the current study is known to increase immune responses by causing a slower release of antigens into the blood, which is a crucial step to maintain activities of immune cells for a prolonged period of time.
Study significance
According to the researchers, hyperimmune F(ab’)2 formulations prepared against SARS-CoV-2 can be an effective treatment strategy to tackle the pandemic until the vaccines come. Moreover, these formulations can be prepared easily in existing facilities available in both high- and low-income countries. Taking the reference of Vital Brazil Institute that produces many registered F(ab’)2 formulations, the researchers mentioned that about 10 liters of plasma could be obtained from one horse, leading to the production of 200 doses of F(ab’)2 concentrate per bleeding.

 *Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: bioRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.