As the world faces unprecedented challenges posed by the COVID-19 pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus, it is evident that there is an unmet need for effective diagnostic methods, therapeutic intervention, and vaccination.
Neutralizing molecules such as antibodies or their derivatives have become crucial tools for the treatment of COVID-19. Such binding molecules also offer a unique way to monitor neutralizing immune response in people infected or vaccinated with SARS-CoV-2.
Ever since the global outbreak of COVID-19, an increasing number of neutralizing antibodies that target the RBD of SARS-CoV-2 has been detected in COVID-19 patients, which underlines the significance of RBD-specific antibodies that can block the RBD: ACE2 interaction site and thus help develop a protective immune response.
A promising alternative to traditional antibodies or IgGs are single-domain antibodies (nanobodies, Nbs) that are derived from the heavy-chain antibodies in camelids. Thanks to their compact folding and small size, Nbs show good chemical stability, fast tissue penetration, and solubility. Nbs can be quickly produced at high yields in bacteria, and, in their monovalent form, they bind to their target with very high affinities.
Researchers from the University of Tuebingen, Eberhard-Karls University, Centre for Experimental and Clinical Infection Research, University Hospital Tuebingen, Helmholtz Centre for Infection Research, Germany, recently described a set of 11 Nbs derived from an immunized alpaca which showed high-affinity binding to the glycosylated SARS-CoV-2 spike receptor domain (RBD) in a study published in the preprint server bioRxiv.*
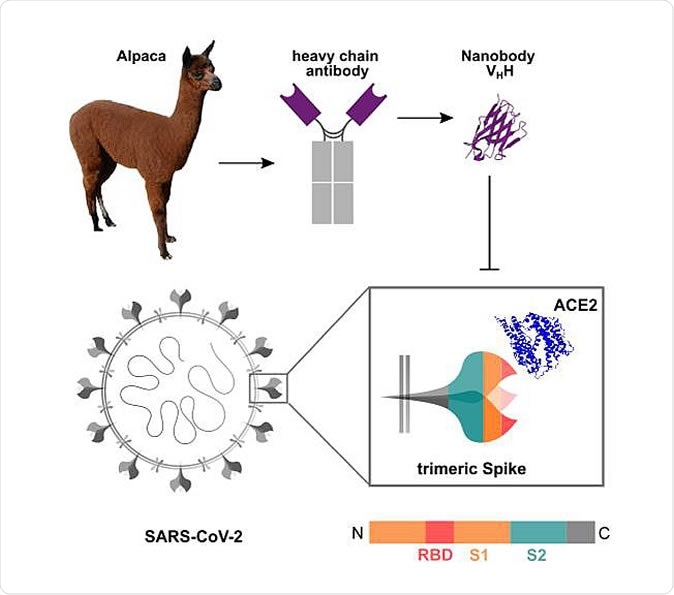
Schematic depiction of the generation of Nbs blocking the SARS-CoV-2 RBD:ACE2 interaction site Nanobodies (Nbs) are genetically engineered from heavy chain only antibodies of alpacas. The interaction between the SARS-CoV-2 homotrimeric Spike protein and ACE2 can be blocked by RBD-specific Nbs. Protein structures adapted from PDB 3OGO (Nb) and 6CS2 (ACE2).

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Nanobodies block interaction between virus proteins and human receptors
With the help of a multiplex in vitro binding assay, the team showed that 8 of the Nbs were effective in blocking the interaction of the S1-domain, RBD, and homotrimeric spike protein with the angiotensin-converting enzyme 2 (ACE2), which is the docking site of the virus in human cells.
Using detailed epitope mapping and competitive binding analysis, the researchers grouped all the Nbs blocking the RBD: ACE2 interaction in three distinct sets. They demonstrated the neutralizing effect of the Nbs with low nanomolar range IC50 values in a cell-based SARS-CoV-2 neutralization assay.
When tested, the Nbs combinations from different sets had significantly lower IC50 values in both functional assays, which indicated an intense synergistic effect of Nbs targeting different epitopes in the RBD simultaneously.
“Interestingly, IC50 values obtained for inhibitory Nbs on RBD and homotrimeric Spike show a higher correlation compared to IC50 values obtained for the S1 domain”
The team applied the most potent Nbs combination (NeutrobodyPlex) in a competitive multiplex binding assay and were able to detect a neutralizing immune response in COVID-19-infected individuals’ plasma samples.
NeutrobodyPlex can help monitor immune status of SARS-CoV-2 patients
Based on the findings of the study, the team designed a novel diagnostic test called NeutrobodyPlex to monitor the emergence and presence of neutralizing antibodies in the serum of individuals infected with SARS-CoV-2. With the help of combinations of Nbs with high affinity covering the RBD: ACE2 interface, the team succeeded in directly and precisely displacing IgGs in the serum samples from these RBD epitopes.
“To our knowledge, the NeutrobodyPlex employing Nbs blocking the RBD: ACE2 interaction site shows for the first time an antigen-resolved analysis of the presence of human IgGs in convalescent individuals suffering from SARS-CoV-2 infection.”
All the monovalent Nbs identified (except NM1225) showed high affinities in the low nanomolar range. Thus, there is no need to reformat these Nbs into bivalent forms by fusing them to an Fc domain or by combining binding sites. By employing better screening strategies, Nbs that can bind to predefined domains in larger antigens can be developed.
According to the team, the NeutrobodyPlex Nbs that block the RBD: ACE2 interaction site is the first to show an antigen-resolved analysis of the human IgG presence in convalescent individuals with SARS-CoV-2 infection. The research team believes that these Nbs can be very promising in both prophylactic and therapeutic approaches in the fight against COVID-19.
The Nbs also offer a novel approach for screening neutralizing immune responses in individuals infected or vaccinated with the virus, thus aiding in monitoring the immune status of patients and in the development of effective vaccines. Compared with other neutralizing antibody detection tests, this novel assay enables automatable high throughput analysis. It can be performed with non-infectious and non-living viral material, which enhances safety while reducing costs.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
NeutrobodyPlex - Nanobodies to monitor a SARS-CoV-2 neutralizing immune response Teresa Wagner, Philipp D Kaiser, Marius Gramlich, Matthias Becker, Bjoern Traenkle, Daniel Junker, Julia Haering, Helen Schweizer, Stefan Nueske, Armin Scholz, Anne Zeck, Katja Schenke-Layland, Annika Nelde, Monika Strengert, Gerard Krause, Juliane S Walz, Natalia Ruetalo, Michael Schindler, Nicole Schneiderhan-Marra, Ulrich Rothbauer bioRxiv 2020.09.22.308338; https://www.biorxiv.org/content/10.1101/2020.09.22.308338v2
- Peer reviewed and published scientific report.
Wagner, Teresa R, Elena Ostertag, Philipp D Kaiser, Marius Gramlich, Natalia Ruetalo, Daniel Junker, Julia Haering, et al. 2021. “NeutrobodyPlex—Monitoring SARS‐CoV‐2 Neutralizing Immune Responses Using Nanobodies.” EMBO Reports 22 (5). https://doi.org/10.15252/embr.202052325. https://www.embopress.org/doi/full/10.15252/embr.202052325.