Beyond the morbidity and mortality associated with the pandemic, the coronavirus disease 2019 (COVID-19) has paralyzed the global economy, causing suffering to education and psychological health. At an unprecedented rate, research into numerous strategies to mitigate the disease is underway.
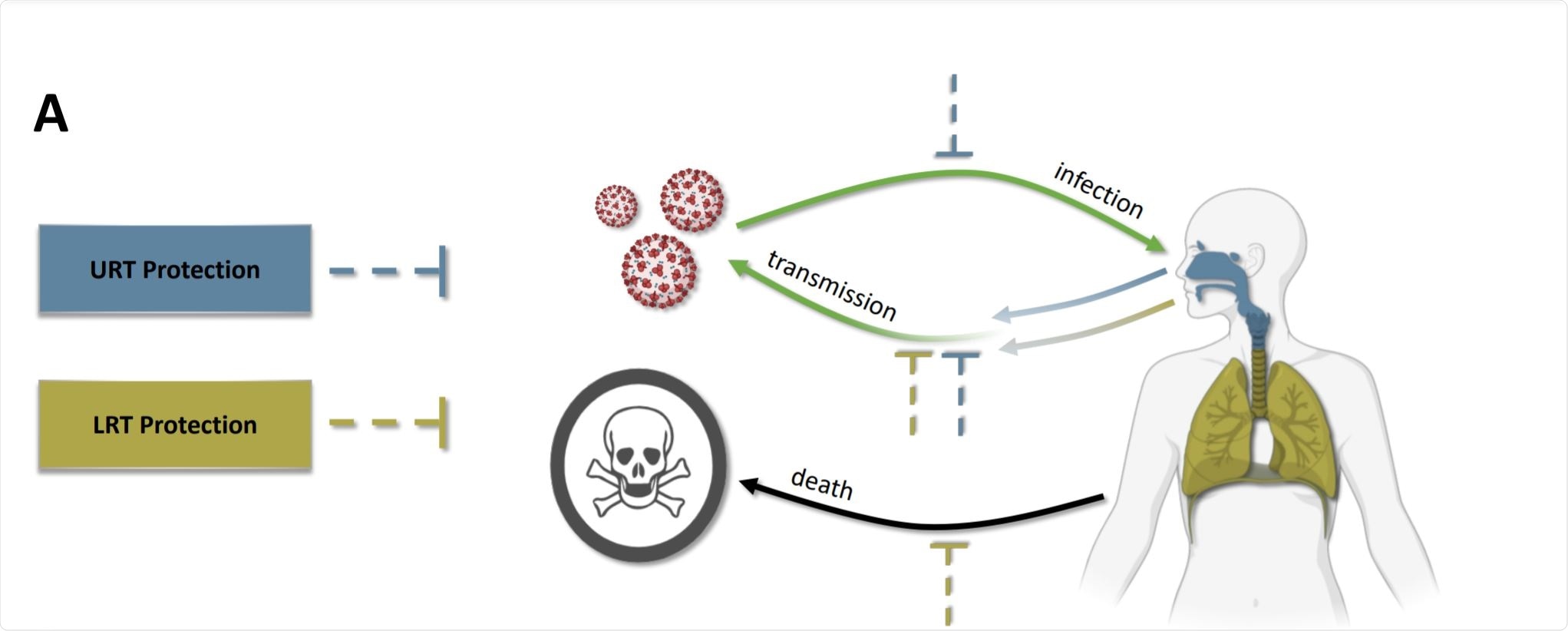
Image Credit: https://www.medrxiv.org/content/10.1101/2020.12.01.20241836v1.full.pdf

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
The United Kingdom is the first country to approve a COVID-19 vaccine that has been tested in a large clinical trial involving only 170 infections. The vaccine tested on 43,000 people showed 95% effectiveness at preventing disease after its second dose. The real-world efficacy of the vaccines when rolled-out to millions of people across the world is, however, currently unknown.
The efficacy of vaccines is critical in determining what path the infection takes. Vaccines that only reduce the clinical severity but not the transmission could drive the evolution of virulence.
An in-depth study considering the viral evolution under vaccine introduction is needed. In this context, Ian F. Miller, from Department of Ecology and Evolutionary Biology, Princeton University, and C. Jessica E. Metcalf, from Princeton School of Public and International Affairs, review the evidence surrounding the evolution of SARS-CoV-2 transmission and the existence of a trade-off between this trait and disease severity.
The negative outcomes of viral evolution include: 1) vaccine-escape via antigenic evolution, resulting in a decrease or loss of vaccine efficacy, and 2) the evolution of increased virulence, which could result in more severe health outcomes and a higher infection fatality ratio (IFR). The duo team focuses on the potential for vaccines to drive the evolution of SARS-CoV-2 virulence.
In this study, they present an evo-epidemiological model that incorporates the differential effects of vaccinal and natural immunity in the upper respiratory tract (URT) and the lower respiratory tract (LRT). They identify desirable features of vaccines that would minimize selection for increased virulence while simultaneously providing personal and population-level protection. They have presented their observations in a recent medRxiv* preprint paper.
Pathogens evolve towards greater transmissibility, reduced host damage, and also less mortality. However, many pathogens can damage the host to enhance transmission or as an unavoidable consequence of transmission. This association results in a positive saturating relationship between transmission rate and mortality rate - the canonical ‘virulence transmission trade-off’ - and limits the evolution of virulence.
This phenomenon is important in the context of vaccine deployment. COVID-19 vaccines show evidence of differential effects, such as conferring greater protection in the LRT. The viral growth in the LRT causes the most impact on the disease severity, whereas protection in the LRT is associated with reduced disease severity, potentially reducing the costs of increased transmission.
Intramuscular delivery of the vaccine stimulates the production of IgG antibodies. The LRT and other tissues infected by SARS-CoV-2 are protected by these IgG antibodies, while the URT is primarily protected by IgA antibodies involved in mucosal immunity. Vaccines that protect both the LRT and URT could achieve reducing disease severity while minimizing the evolution of increased virulence.
An immunity which reduces disease to a greater extent than transmission drives the evolution of virulence. Vaccinal immunity to SARS-CoV-2 could have this combination of effects.
If the impact of vaccination on COVID-19 infection in the upper respiratory tract (URT) and lower respiratory tract (LRT) are not equal, then virulence evolution occurs.
While vaccines will be a critical factor shaping the landscape of immunity that drives SARS-CoV-2 evolution, naturally acquired immunity could also contribute to selective pressures, the authors write.
This study synthesizes important information on the factors necessary for virulence evolution to occur. They find the evolution of transmission occurring but no definitive support for links between transmission and virulence or a trade-off between the two traits.
Our evo-epidemiological model identified URT protection as a desirable characteristic of ‘second wave’ vaccines expected to be administered to large portions of the population, as it would 10 mitigate selection for increased virulence should the transmission be subject to an evolutionary tradeoff with disease severity.”
The authors discuss in detail the various factors and limitations in studying the virulence evolution. In conclusion, the authors point out that based on current evidence, there is no risk of vaccine driven virulence evolution. However, they warn that despite these low risks, SARS-CoV-2 virulence should be closely monitored. The transmission limiting characteristics should be prioritized for ‘second-wave’ vaccines, the authors advise.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
No current evidence for risk of vaccine-driven virulence evolution in SARS-CoV-2 Ian F. Miller, C. Jessica E. Metcalf medRxiv 2020.12.01.20241836; doi: https://doi.org/10.1101/2020.12.01.20241836
- Peer reviewed and published scientific report.
Miller, Ian F., and C. Jessica E. Metcalf. 2022. “Assessing the Risk of Vaccine-Driven Virulence Evolution in SARS-CoV-2.” Royal Society Open Science 9 (1). https://doi.org/10.1098/rsos.211021. https://royalsocietypublishing.org/doi/10.1098/rsos.211021.