A team of scientists from India has recently investigated the antiviral potency of a traditional medicinal plant Cissampelos pareira L. in preventing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. The findings reveal that both whole-plant extracts and bioactive compounds can inhibit SARS-CoV-2 replication by modulating estrogen receptor 1 (ESR1) activity. The study is currently available on the bioRxiv* preprint server.
Since its emergence in late December 2019 in Wuhan, China, SARS-CoV-2, the causative pathogen of coronavirus disease 2019 (COVID-19), has infected more than 111 million people and claimed 2.46 million lives globally.
As an attempt to curb the COVID-19 trajectory, several potential vaccines targeting the viral spike protein have been developed at record speed. Some of these vaccines have already received emergency use approval in many countries. In addition, a substantial number of human clinical trials have been conducted to determine the efficacy of repurposed antiviral medicines, therapeutic monoclonal antibodies, and small molecule inhibitors in treating moderate to severe COVID-19 patients. Besides pharmaceutical interventions, the anti-SARS-CoV-2 effectiveness of many alternative medications, such as phytochemicals, has been studied extensively to develop cost-effective COVID-19 therapeutics with minimal side-effects.
Cissampelos pareira L. (Cipa) is one such medicinal plant traditionally used to treat hormonal imbalance, fever, cough, skin ulcer, jaundice, and asthma. There is evidence suggesting that aqueous and ethanolic extracts of Cipa have antiviral and antimalarial properties. In the current study, the scientists have conducted genome-wide expression profiling of Cipa-treated human breast cancer cell lines to evaluate the mode of action and antiviral potency of Cipa.
Study design
Specifically, the scientists treated the cells with different concentrations of whole plant aqueous/ethanolic extracts of Cipa and isolated RNA samples to conduct whole transcriptome analysis. Next, they conducted Gene Set Enrichment Analysis to identify specific sets of Cipa-modulated genes. By analyzing the differential expression profiles of genes, they aimed to decipher the involvement of Cipa in different cellular pathways, as well as to investigate its association with substances that are able to modify intracellular processes.
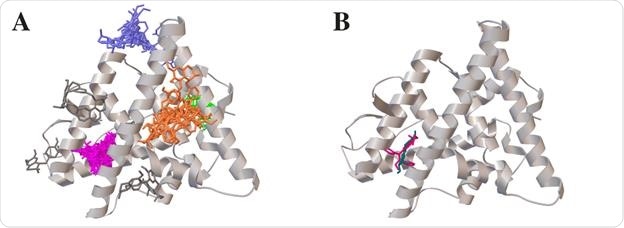
Plot for virtual docking sites of 61 Cipa constituents (four clusters, pink, green, orange and blue) and binding site of tamoxifen (pink) and 17 beta estradiol (blue) on estrogen receptor alpha.
Important observations
Using gene ontology analysis, the scientists observed that Cipa treatment downregulated a specific set of genes involved in lipid metabolism pathways. Similarly, by conducting Gene Set Enrichment Analysis, they noticed that Cipa treatment caused both upregulation and downregulation of specific genes involved in the estrogen signaling pathway. Specifically, they observed a reduction in mRNA expression of estrogen receptor 1 (ESR1), which they further confirmed by real-time quantitative analysis. To obtain information about possible binding sites, they conducted a virtual screening of bioactive compounds present in Cipa against ESR1 and identified 4 preferential binding sites for a total of 61 compounds.
Furthermore, they performed connectivity map analysis using the dataset obtained from genome-wide transcriptome analysis and observed a high connectivity score between Cipa and protein synthesis inhibitors (emetine, cycloheximide, verrucarin-a, and cephaeline) that are known to have antiviral activities in vitro. With further analysis, they observed that Cipa treatment significantly downregulated the mRNA expression of ribosomal protein L7 (RPL7), which is a coactivator of ESR1.
To investigate whether the antimicrobial activities of Cipa are modulated via ESR1, they administered Cipa to two Dengue virus-infected cell lines that differentially express ESR1. The findings revealed that Cipa-mediated inhibition of the Dengue virus was higher in cells expressing a high level of ESR1. Moreover, they observed that the virus inhibitory effect of Cipa was diminished by the experimental knockdown of ESR1 in these cells.
To explore the association between Cipa and SARS-CoV-2, they analyzed transcriptome data of COVID-19 patients and observed high expression levels of both ESR1 and RPL7. Given these observations, they investigated the effectiveness of Cipa in modulating SARS-CoV-2 infection. Using SARS-CoV-2-infected cells, they observed that both aqueous and ethanolic extracts of Cipa caused a significant reduction in viral RNA level, indicating potent antiviral efficacy of Cipa against the virus. With further analysis, they observed that specific bioactive compounds present in Cipa extracts could also significantly reduce SARS-CoV-2 RNA levels in infected cells.
Study significance
The study identifies the potential antiviral efficacy of Cipa extracts, which are traditionally used as a potent hormone balancing Ayurvedic medicines. The findings reveal that Cipa exerts anti-SARS-CoV-2 activities by modulating the estrogen signaling pathway. In addition, the study highlights that estrogen receptors can serve as potential therapeutic targets against COVID-19.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources