Artist imaging of protein spike on the surface of cells exposed to the vaccine. Image Credit: University of Southampton
The spike protein of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), which protrudes from the virus envelope, is the key structure responsible for infecting host cells. The S1 subunit helps bind the virus to the angiotensin-converting enzyme 2 (ACE2), and the S2 subunit helps with membrane fusion with the host cell.
The virus spike protein is the main target of vaccines, but vaccines use different methods to target the spike protein. The Moderna and the Pfizer vaccines encode the full-length spike protein with two mutations for stability. Sinovac's vaccine uses an inactivated virus that presents the wild-type spike protein.
Most vaccines aim to elicit a robust immune response, mainly against the receptor-binding domain (RBD) of the spike protein that has several neutralizing epitopes. To enable this, many vaccines include mutations that ensure the spike protein is in the conformation before it fuses with the host cell.
The AstraZeneca ChAdOx1 vaccine uses a chimpanzee adenovirus and encodes the full-length spike protein. It has been shown to elicit a robust immune response as well as a T-cell response. In a recent study, researchers report the characteristics of the spike protein expressed by the vaccine.
Graphic depicting how the protein spikes form on the surface of cells presented with the vaccine. Image Credit: University of Southampton
Characterizing spike protein produced by vaccination
The researchers used HeLa S3 cells infected with the ChAdOx1 vaccine to detect the presence of spike protein on the cell surface. Vaccinated mice sera showed about 60-70% of the cells expressed the spike protein.
How the spike protein is processed after vaccination depends both on the receptors and the enzymes produced by the spike-producing cells. The presence of receptors such as ACE2, mainly in the nose, gastrointestinal tract, and lungs, can result in a change in the structure of the spike protein.
However, these receptors are generally not present in the types of cells targeted by the ChAdOx1 vaccine, which is administered intramuscularly. The cells selected for this study also do not express these receptors and so can act as a suitable substitute for in vitro studies.
Testing using recombinant angiotensin-converting enzyme 2 (ACE2) and human monoclonal antibodies confirmed that the spike protein is present in the correct conformation, just like that on the wild-type virus.
The team then used cryo-electron tomography on cells infected with the vaccine to understand the spike's structure on the cells. They saw the surface of the cells were covered with protrusions, similar to the spike protein on SARS-CoV-2. Further image analysis of the protrusions revealed their structure is very similar to the spike protein conformation before membrane fusion.
The spike protein's presence on vaccinated cells provides good evidence that the vaccine can generate a strong immune response. Unlike some other vaccines, the ChAdOx1 vaccine does not use any mutations on the spike protein to ensure its stability.
Spike protein produced mimics natural infection
The virus spike protein also has glycans, a type of sugar, coating it, which disguise the virus so that the host immune response does not detect the virus. This is a common strategy used by many viruses to elude the host immune system. So it is crucial that the spike proteins produced by vaccination also produce these glycans to ensure complete mimicking of the virus for the production of suitable neutralizing antibodies.
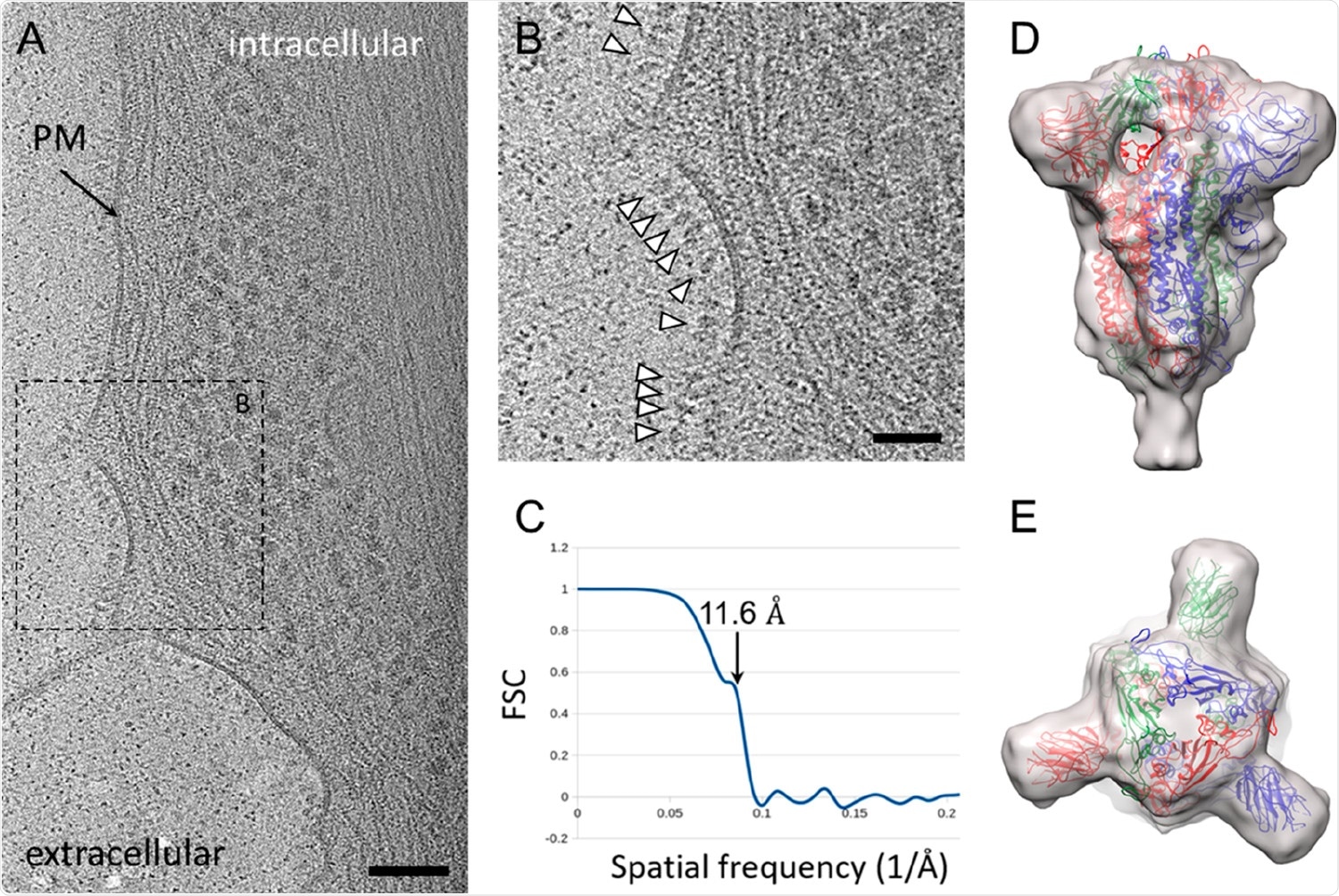
CryoET and subtomogram average of ChAdOx1 nCoV-19 derived spike. (A) Tomographic slice of U2OS cell transduced with ChAdOx1 nCoV-19. The slice is 6.4 Å thick; PM = plasma membrane, scale bar = 100 nm. (B) Detailed view of the boxed area marked in (A). White arrowheads indicate spike proteins on the cell surface; scale bar = 50 nm. (C–E) Subtomogram average of ChAdOx1 nCoV-19 spikes at 11.6 Å resolution as indicated by Fourier-Shell correlation at 0.5 cutoff (C), shown from side view (D) and top view (E). SARS-CoV-2 atomic model (PDB 6ZB5)(29) is fitted for reference.
To test for the presence of glycans, the team used HEK293 cells infected with the vaccine and used enzymes to generate glycopeptides. Tests revealed a high level of glycans present on the spike, providing evidence that the spike proteins produced by ChAdOx1 vaccination were similar to the spike protein expressed by natural infection, triggering an immune response that can protect against COVID-19.
The team used cryo-electron microscopy to study the glycosylation further. They found the glycosylation to be a mixture of oligomannose/hybrid and complex-type sites. However, they did not detect O-linked glycans, perhaps because of their low abundance.
Thus, the results reveal that vaccination by ChAdOx1 produces spike protein that is very similar to that produced by the SARS-CoV-2 virus upon natural infection, providing more evidence that the vaccine is triggering the immune system to fight COVID-19.