A group of Harvard researchers recently expanded the use of cryo-electron microscopy (cryo-EM) to analyze small proteins at high resolution. In their study, published on the preprint server bioRxiv*, the researchers describe the method that allows high-resolution structure determination of small proteins by single-particle cryo-EM using a target-binding nanobody ensemble called a Legobody.
Single-particle electron cryo-EM is an ideal technique for determining the structure of small proteins. Despite its utility, cryo-EM has been limited in its ability to analyze particles smaller than 100 kilodaltons (kDa). This poses a challenge during the analysis of proteins that are smaller than 100 kDa, as approximately 50% of membrane and other medically significant proteins are smaller than 50 kDa.
Among the several different approaches that have been employed to image small proteins using cryo-EM, one method involves the fusion of the target protein with different scaffolds or the use a binding partner to increase the size of the protein. Some of the different scaffolds that have been employed for this purpose include modified ankyrin repeat proteins (DARPins), Fab fragments of antibodies, or nanobodies.
What are nanobodies?
Nanobodies, which are derived from single-chain antibodies of camelids, are an appealing option for the fusion approach. Importantly, nanobodies can form rigid structures that bind to diverse shapes of the target proteins such as loops, convex surfaces, and cavities.
Additionally, these scaffolds can be selected from immunized camelids or from large in vitro libraries displayed by phages, yeast cells, or ribosomes.
Nanobodies can also be quickly produced in large quantities. Additional advantages of this approach include their ability to lock a protein in a fixed conformation, and their existing extensive use in X-ray structure analysis.
Despite these advantages, the small size of nanobodies, which is between 12 and 15 kDa, is a limiting factor for its direct use in cryo-EM. In an effort to overcome this problem, the researchers of the current study increased the nanobody size to about 120 kDa by attaching two rigid scaffolds to this binding partner.
The utility of the Legobody
In their work, the researchers began by determining the structures of two small proteins including the KDEL receptor (KDELR), which is about 23 kDa in size. The second protein was the receptor-binding domain (RBD) of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) spike protein, which is about 22 kDa. Both of these proteins are asymmetric monomers and are smaller than 40 kDa, which is well below the estimated limit for direct cryo-EM single-particle analysis.
The KDELR is a small and non-symmetric protein with no domains outside the membrane and a high tendency to aggregate during sample prep. In the current study, the researchers used supplemental ‘tricks’ to reduce aggregation, enhance stability, and ensure preferred particle orientation. KDELR is representative of membrane proteins, many of which are of great interest for drug development.
Likewise, the RBD of the SARS-CoV-2 spike protein posed similar challenges for cryo-EM analysis, as it consists of b-strands and extended polypeptide segments. In addition to its small size, these characteristics cause the RBD to be a more difficult model as compared to a protein that consists of primarily a-helices.
Here, the researchers obtained a good quality map, especially at the RBD/nanobody interface, with side-chain density for all interacting amino acids.
Study findings
The researchers showed a 3.2Å and 3.6 Å overall resolution with a density sufficient for de novo model building for the map of KDELR and SARS-CoV-2 RBD. These resolutions confirmed the interactions between the two scaffolds and the nanobody.
In addition to the two scaffolds, both a Fab-fragment of an antibody directed against the nanobody and a nanobody-binding protein A fragment fused to maltose-binding protein (MBP). Fab-binding domains were also incorporated into the Legobody. All interactions between these structures were rigid that maintained the distinct shape of the Legobody.
To this end, the Legobody consisted of two lateral arms that were formed by the two scaffolds, as well as a central lobe that was contributed by the nanobody. Taken together, the Legobody had an overall size of approximately 120 kDa. Additionally, the center of alignment at the position of the nanobody facilitated all steps of cryo-EM analysis, from particle picking and classifications to final refinement.
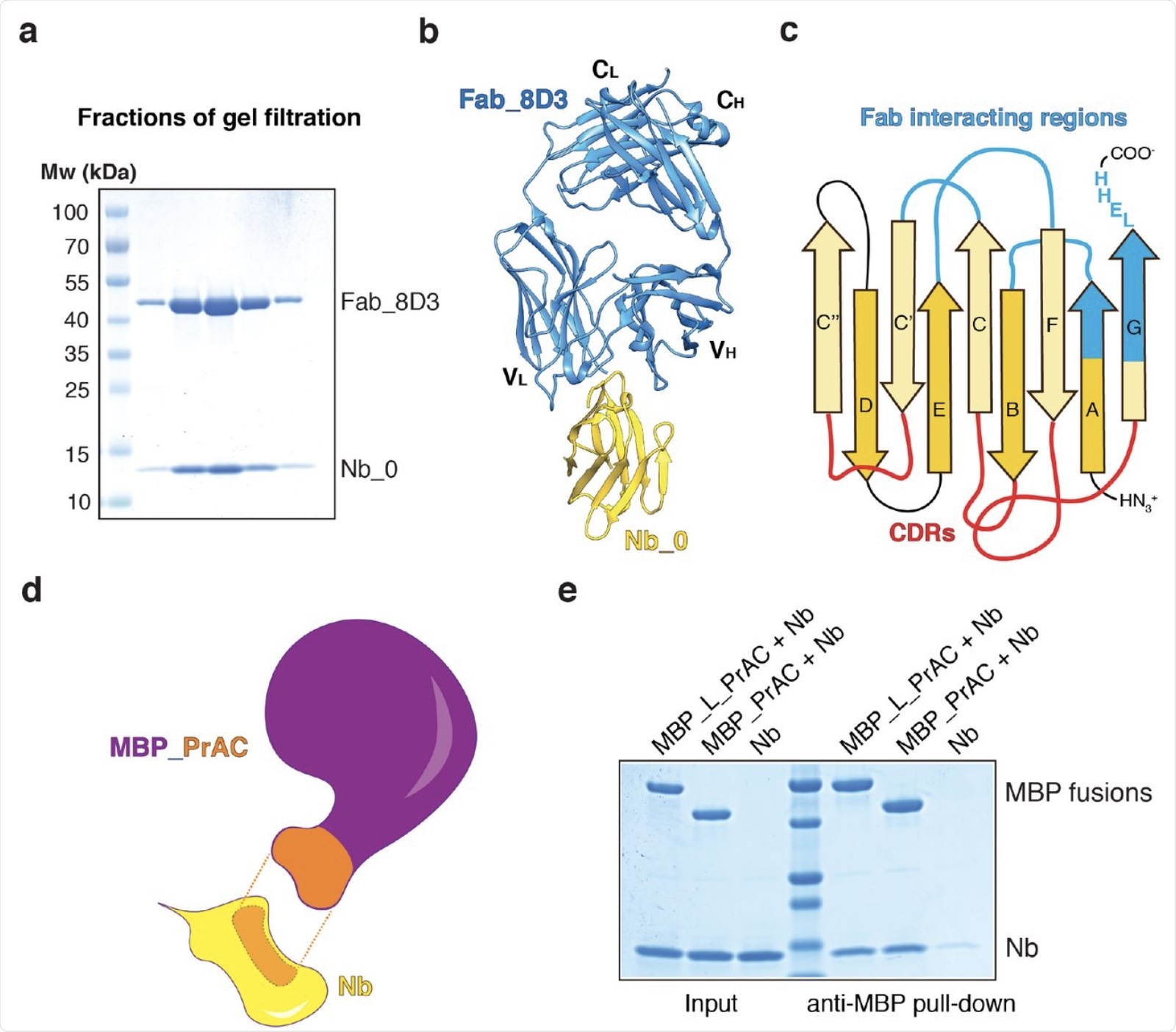 Generation of two nanobody-interacting scaffolds. a. Nanobody Nb_0 was mixed with Fab_8D3 and the complex was subjected to gel filtration. Fractions were analyzed by SDS-PAGE and Coomassie-blue staining. b, Crystal structure of the Nb_0/Fab_8D3 complex shown in cartoon representation. Nb_0 is colored in yellow and Fab_8D3 in blue. c, Scheme of the nanobody, with β-strands labeled according to convention. CDR loops are colored in red and regions interacting with the Fab in blue. d, Scheme of the interaction of nanobody (Nb; yellow) with the domain C of protein A (PrAC; orange) grafted onto maltose-binding protein (MBP; purple). The interacting regions between Nb and MBP_PrAC are shown in orange with dashed lines in between. e, PrAC fused to MBP through a flexible linker (MBP_L_PrAC) or grafted onto MBP (MBP_PrAC) was mixed with Nb. The mixture was incubated with MBP-interacting amylose resin, and the bound material analyzed by SDS-PAGE and Coomassie-blue staining. The input corresponds to 50% of the material used for the pull-down.
Generation of two nanobody-interacting scaffolds. a. Nanobody Nb_0 was mixed with Fab_8D3 and the complex was subjected to gel filtration. Fractions were analyzed by SDS-PAGE and Coomassie-blue staining. b, Crystal structure of the Nb_0/Fab_8D3 complex shown in cartoon representation. Nb_0 is colored in yellow and Fab_8D3 in blue. c, Scheme of the nanobody, with β-strands labeled according to convention. CDR loops are colored in red and regions interacting with the Fab in blue. d, Scheme of the interaction of nanobody (Nb; yellow) with the domain C of protein A (PrAC; orange) grafted onto maltose-binding protein (MBP; purple). The interacting regions between Nb and MBP_PrAC are shown in orange with dashed lines in between. e, PrAC fused to MBP through a flexible linker (MBP_L_PrAC) or grafted onto MBP (MBP_PrAC) was mixed with Nb. The mixture was incubated with MBP-interacting amylose resin, and the bound material analyzed by SDS-PAGE and Coomassie-blue staining. The input corresponds to 50% of the material used for the pull-down.
Conclusions
Overall, the Legobody approach discussed here is a sufficient method that allows cryo-EM to be used for the structural determination of small proteins. The researchers indicate that this novel method could be used for any target binding following its tight binding to an available nanobody.
Notably, several limitations are associated with the current method. Additionally, it may be important to modify the Legobody in the future to increase its molecular mass, stability, and rigidity to ultimately improve the resolution of the cryo-EM maps.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources