The ongoing coronavirus disease 2019 (COVID-19) pandemic which is caused by a novel coronavirus, namely, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has claimed more than 4.6 million lives worldwide. This disease causes a broad range of symptoms related to respiratory, neurological, and digestive disorders. Some severely infected COVID-19 patients develop acute pneumonia, while others suffer from gastrointestinal (GI) symptoms, such as diarrhea, vomiting, etc.
SARS-CoV-2 Infection in Humans
SARS-CoV-2 is an RNA virus that belongs to the family Coronaviridae of genus Betacoronavirus. The Spike (S) protein of this virus primarily mediates interaction with the human host cells. The receptor-binding domain (RBD) present on the surface of the S protein binds with the angiotensin-converting enzyme 2 (ACE2) receptor of the host. ACE2 is present on several host tissues, such as GI epithelial cells and liver cells.
Researchers have detected the presence of this virus in several tissues, such as the esophagus, stomach, duodenum, and rectum. Also, endoscopy of hospitalized patients with severe COVID-19 infection has shown esophageal bleeding with erosions and ulcers.
People with comorbidities generally suffer from severe COVID-19 infection. These patients exhibit higher levels of ACE2 in the lungs than those without comorbidities. A recent study has shown that ACE2 was significantly expressed in the lungs of people suffering from chronic obstructive pulmonary diseases and pulmonary arterial hypertension. As the expression of the ACE2 depends on certain conditions such as elevated glucose levels, hypoxia, and cell stress, researchers believe that other comorbidities related to the GI could be associated with different forms of COVID-19.
A New Study
A new study published in the journal Frontiers of Medicine indicated that Barrett's esophagus (BE) and gastroesophageal reflux disease (GERD) may represent novel comorbidities associated with SARS-CoV-2 disease. A previous study had estimated that in the United States, 5.6% of adults have BE, a disease where esophageal squamous mucosa gets damaged by GERD.
In this study, researchers revealed that ACE2 is highly expressed in the esophagus of patients with BE. In these patients, the acid pH induces the expression of ACE2. The team cultured human primary monocytes in reduced pH, which revealed enhancement in the expression of ACE2 and more significant viral load upon SARS-CoV-2 infection.
The researchers suggest that acid pH enhances COVID-19 infection by up-regulating the ACE2 receptor, and this, in turn, might have clinical implications for patients suffering from GERD or BE. Although there is no evidence of mechanisms that connects pH alterations and ACE2 expression, previous studies have shown that hypoxic conditions can increase the expression of ACE2. This study revealed that two of the known regulators of ACE2, namely, HNF1B and FOXA2, were reported to be up-regulated in 6 out of 8 BE transcriptomic studies. This indicates that these regulators could be associated with the pH-induced ACE2 expression in BE.
One of the main characteristic features of COVID-19 disease is pulmonary damage, which may lead to acute hypoxia and another respiratory acidosis. In addition, researchers believe that in some of the severely infected COVID-19 patients, blood acidosis occurs. This condition deteriorates the patient's condition by increasing ACE2 expression, which enhances the entry of SARS-CoV-2 into human cells. Also, an increased level of lactate dehydrogenase enzyme, which converts lactate from pyruvate, has been linked with severe COVID-19 disease. This increased level of lactate may directly alter the extracellular and intracellular pH, which alters ACE2 expression. More research is required to evaluate the extent to which acute systemic acidosis influences COVID-19 severity. In this study, researchers have indicated that pH alone might be able to modify ACE2 expression and subsequently inhibit SARS-CoV-2 infection in patients.
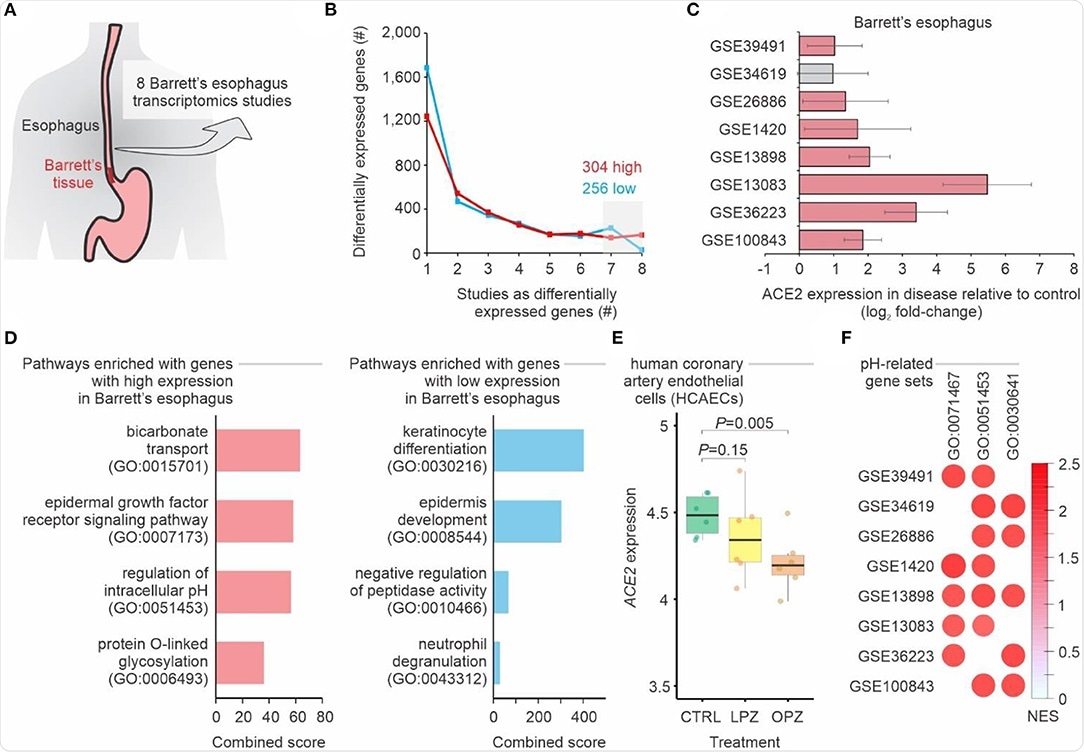
Meta-analysis of gastroesophageal junction transcriptomes of patients with Barrett's esophagus. (A) Meta-analysis of 8 studies of Barrett's esophagus transcriptomes. (B) Number of differentially expressed genes in Barrett's esophagus compared with non-Barrett's esophagus. The lines show the number of genes (y-axis) considered up-regulated (red lines) or down-regulated (blue lines) in Barrett's esophagus (P-value < 0.05; log2 fold-change > 1; combined FDR <0.01) in one or more datasets (x-axis). The numbers of up-regulated and down-regulated genes in at least seven studies are indicated. (C) ACE2 is upregulated in patients with Barrett's esophagus. Each bar represents the log2 expression fold-change between patients and control individuals. The error bars indicate the 95% confidence interval. Bars in red represent a P-value < 0.05 and in gray a non-significant P-value. (D) Pathway enrichment analysis using the up-regulated and down-regulated genes in at least seven studies. The bars represent the combined score (x axis) calculated by Enrichr tool for selected Gene Ontology gene sets (y axis). (E) ACE2 expression in cells treated with proton pump inhibitors. Each boxplot represents the log2 expression of untreated (CTRL) cells and cells treated with either omeprazole (OPZ) or lansoprazole (LPZ). (F) Gene Set Enrichment Analysis (GSEA) of the 8 studies of Barrett's esophagus transcriptomes using pH-related gene sets. The size and color of the circles are proportional to the normalized enrichment score (NES) of the gene sets (columns) on each study (rows). The Gene Ontology IDs are indicated at the top.
This study included two independent cohorts comprising 1,357 SARS-CoV-2 infected patients. One group had a history of using proton pump inhibitors (PPI) and another group contained individuals who did not use the drug. This study revealed patients treated with PPI (used for GERD treatment) were at a greater risk of developing severe SARS-CoV-2 infection. This was assessed by observing increased intensive care admission.
Further, the scientists also reported that the rate of mortality increased 2- to 3-fold in the group treated with PPI, compared to those not using this drug. This result aligns with previous studies that conducted a meta-analysis revealing that PPIs elevated the risk of progression to severe COVID-19. A prior study also reported that individuals treated with PPI were at a greater risk of getting COVID-19 infection, perhaps because PPI reduced the gastric barrier to SARS-CoV-2.
Conclusion
The authors of this study believe that pH may greatly influence SARS-CoV-2 infection and the severity of the disease. However, more studies using larger cohorts are required to validate this finding and also determine the molecular mechanism related to pH-induced ACE2 expression.