Natural severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection or COVID-19 vaccination both result in the production of antibodies in the body. However, the amount of antibodies present in the population is still in question since the virus continues to spread rapidly and vaccine uptake is decreasing.
A collaborative team, led by principal investigator Dr. Eric Boerwinkle, at The University of Texas Health Science Center at Houston, School of Public Health is conducting a survey designed to assess the frequency of antibody-positive individuals across Texas. The TEXAS Coronavirus Antibody REsponse Survey (CARES) aims to look for antibodies in the blood made in response to natural SARS-CoV-2 infection or vaccination.
A preprint version of the ongoing research is available on the medRxiv* preprint server.
Why test for SARS-CoV-2 antibodies?
SARS-CoV-2 antibodies are induced by the COVID-19 vaccine or by SARS-CoV-2 infection or both. The presence of antibodies is not a direct measure of protection. An immune response includes a cellular response and a humoral or antibody response with complex dynamics. So why measure the antibodies induced? These tests are essential to detect previous infections in people who were asymptomatic or had mild COVID-19 disease. They also provide population data regarding the segments of populations producing antibodies.
This latest survey data provides pertinent information regarding:
- The number of people infected with SARS-CoV-2 in Texas.
- The number of people vaccinated and developed antibodies.
- How long do SARS-CoV-2 antibodies last in the body.
- The characteristics of people who develop SARS-CoV-2 antibodies.
- The characteristics of people who never develop SARS-CoV-2 antibodies.
How will this data help?
The scientists have undertaken this survey to provide answers to the following questions. First, do people with SARS-CoV-2 antibodies have immunity against future infection? Second, if the antibodies provide immunity, then what amount of antibodies are required for protection? Third, how long does the protection last? Finally, can the people with antibodies still infect others?
The scientists propose that this data will be useful to inform local and state policies. In addition, it will be helpful for clinical and public health recommendations and practice.
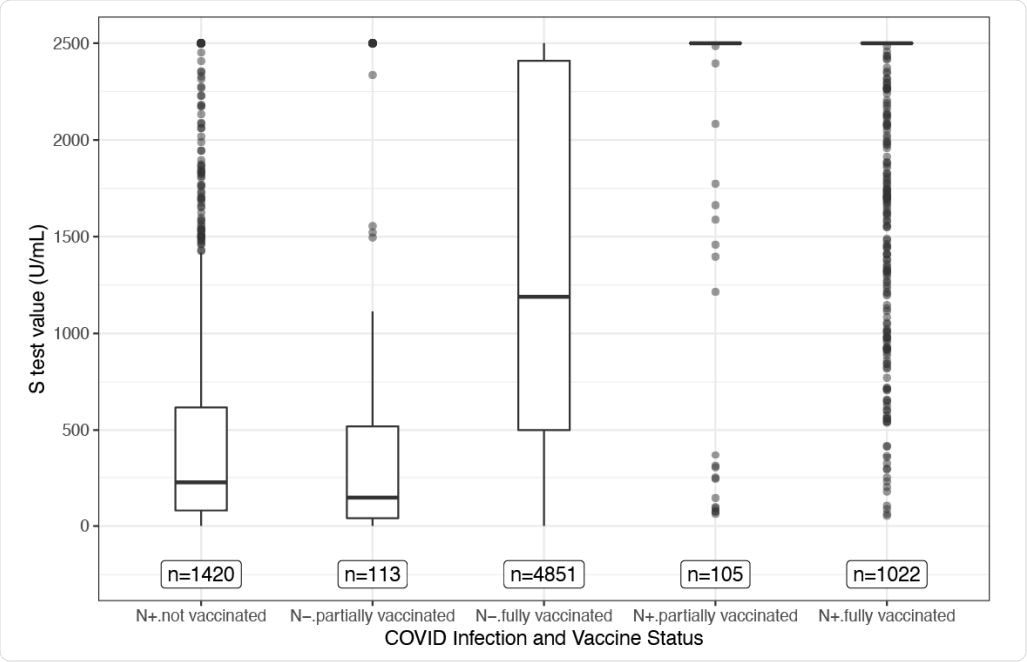
Boxplots of S-test values for each group. This figure shows a traditional boxplot for the S-test values for each group. The N-test status (+ or -) denotes prior infection (N+) or no prior infection (N-). Each box represents the data falling between the 25th and 75th percentiles. The horizontal bar within the box represents the median, and the whiskers extend 1.5 times the interquartile range below the 25th and above the 75th percentiles. The points that lie beyond the whiskers can be considered extreme values. Note that for the groups who had a prior infection and either partial or full vaccination, over 80% of the values are greater than or equal to 2,500 U/mL, so the box part of the boxplot collapsed to the bar at the top of the graph.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
How to test for SARS-CoV-2 antibodies?
The TEXAS CARES is a statewide program designed to estimate the frequency of antibody-positive individuals across the state of Texas. It is a large and diverse population consisting of employees of various retail and industrial sectors, K-12 and university students and educators, and patients and staff at Federally Qualified Health Centers.
Two different immunoassays or antibody tests are being used in this survey: Roche S-test and Roche N-test. The Roche S-test identifies antibodies against the SARS-CoV-2 spike protein - a result of natural infection or COVID-19 vaccination.
The Roche N-test identifies antibodies against SARS-CoV-2 nucleocapsid protein - a result of natural infection. Two different tests aid in monitoring the combined impact of prior infection and the COVID-19 vaccine.
Whole blood samples are used for the tests.
Currently, the survey reports data from 8,846 individuals. The individuals answered a questionnaire to inform the vaccination status.
SARS-CoV-2 antibody data
The Roche N-test identified participants that either had a prior infection (N+) or no prior infection (N-). According to the infection and vaccination status, participants were grouped into five categories:
- N+/not vaccinated
- N-/partially vaccinated
- N-/fully vaccinated
- N+/partially vaccinated
- N+/fully vaccinated
Of the participants, 80.95% of N+/partially vaccinated, and 83.07% of N+/fully vaccinated individuals had the highest S-test value indicating a high amount of antibodies. The S-test values were not significantly different between the individuals with prior infection and partial vaccination and individuals with prior infection and full vaccination. Conversely, unvaccinated participants with prior infection had low S-test values indicating less protection.
Conclusions
According to the data obtained so far through this survey, individuals with prior SARS-CoV-2 infection and partial or fully vaccinated status have a high amount of antibodies in their blood.
On the other hand, individuals infected and not vaccinated have a low amount of antibodies in their blood. Thus, there is a combined impact of prior infection and vaccination on antibody presence.
The TEXAS CARES project is still underway and is continuously updating. There is a possibility of over or underestimation of antibody data because the participation in this survey is volunteer-based. Therefore, currently, it is a convenience-based sample size. The data will reflect accurate values when more people from the entire population of Texas participate in the study.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
Source:
Journal reference: