Sponsored Content by LonzaReviewed by Ify IsiborApr 30 2026
Novel molecular formats - Fc-fusion proteins, bispecific antibodies, and Fab fragments - are driving therapeutic breakthroughs in an ever-evolving field of biologics. Yet their increased complexity presents considerable analytical development challenges.
It is essential to develop fit-for-purpose analytical methods early in development to ensure manufacturing consistency, product quality, and regulatory compliance.
Much has been discovered surrounding disease pathologies since the introduction of monoclonal antibody (mAb) therapies. For example, it is now known that attacking a single target is often insufficient when treating complex diseases. Many new complex protein therapeutics have been developed based on this knowledge, with novel molecular formats capable of targeting two or more targets.
Trials with Fc-fusion proteins, bispecific non-Fc proteins such as blood factors and Fab fragments, and others are now demonstrating increased therapeutic efficacy, with several candidates already approved for marketing.
The analytical challenges of complex molecules
Increased molecular complexity poses additional analytical challenges, and standard methods used for mAbs do not apply in many cases (Figure 1).
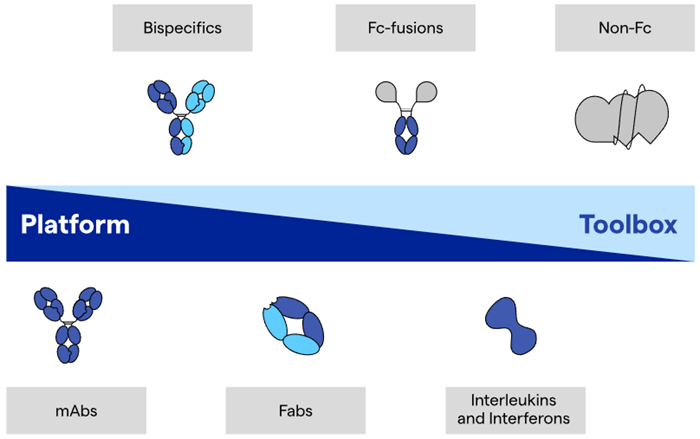
Figure 1. Platform and advanced analytical toolbox approaches to meet the specific needs of the product. Image Credit: Lonza
As a result, it can be challenging to establish fit-for-purpose analytical methods for complex molecules within the typically accelerated development timelines. It is important to employ the right methods to ensure that products’ critical quality attributes are accurately measured with the appropriate level of sensitivity.
It is equally important to develop the right analytical methods for biologics from the earliest possible stages of development. This provides increased confidence that the manufacturing process is consistent and that the candidate is the right molecule, with the right quality and purity, as it moves into the clinic, on to BLA filing, and eventually to commercial production.
Supporting process development with analytical expertise
Jennifer Newman is a member of the mammalian analytical development team at Lonza’s Mammalian Visp site. This role involves supporting the company’s process development (PD) teams with analytical method development, process analytics, and troubleshooting.
Jennifer boasts almost 20 years of experience in analytical method development, leveraging her wide-ranging knowledge of all aspects of PD and an understanding of method requirements as they transition into the quality control (QC) environment for product release.
This expertise is especially valuable when developing the phase-appropriate methods required to support Lonza’s reduced project timelines.
Every molecule tells a different story, and Lonza’s teams must listen and translate this into the most appropriate analytical strategy for that molecule. This mindset guides how the company approaches each project, ensuring that its methods are tailored to the distinct challenges and characteristics of each molecule and are scientifically sound.
Jennifer and her team bring this philosophy to life by leveraging Lonza’s extensive experience and its toolbox of advanced analytical methods to develop flexible phase-appropriate analytical strategies for process development and manufacturing. These strategies ensure that analytical method development readiness is specifically tailored to the needs of a customer project as the project progresses.
For instance, methods must deliver accurate estimates of product and impurity levels at the PD stage and provide sufficient information for decision-making. These methods do not need to be super precise at this stage, however.
Methods are then refined at later stages when more purified material is available, thereby better ensuring the appropriate levels of sensitivity, precision, accuracy, and overall suitability for use in a QC environment.
Lonza’s analytical toolbox for accelerated development
Lonza’s analytical toolbox accelerates biologics method development. This toolbox includes every type of method previously developed by experts across the global Lonza network for mAbs and a range of complex formats.
This suite of methods has been achieved through a comprehensive array of techniques and method-development approaches, so Lonza’s teams do not need to start analytical method development from scratch. Rather, it is possible to choose an existing method chosen as a starting point and begin there, based on an in-depth understanding of the molecule’s design and structure (Figure 2).
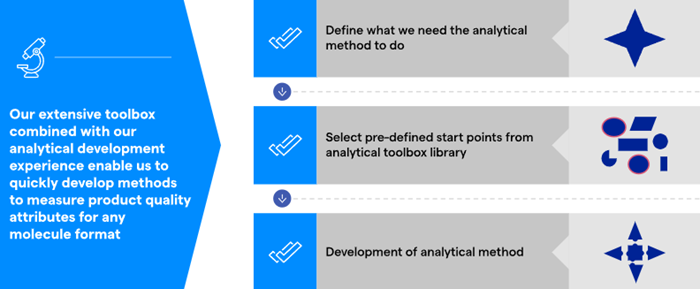
Figure 2. Our analytical strategy for Method Development. Image Credit: Lonza
A recent example demonstrated low-pH-induced aggregation using a standard small-scale purification method. Using Lonza’s prior experience and Jennifer’s experience in method troubleshooting provided the team with a useful starting condition, allowing them to focus on tailoring the elution buffer pH.
This approach significantly reduced the time required to develop the product-specific method, ensuring the customer received reliable, process- and product-specific results without compromising accelerated development timelines to IND filings.
Tackling novel analytical challenges with a proactive approach
There will always be cases where a plug-and-play approach may not work, and it will be necessary to develop a method from scratch. Lonza’s team is well-positioned to tackle these challenges thanks to the company’s proactive, highly flexible, and data-driven approach to analytical method development.
Another example saw the team evaluating an existing capillary electrophoresis (CE SDS) method using spiking studies to confidently quantify glycan occupancy for a product with more than 95 % glycosylation.
The spiking approach in this instance was developed using a glycosylated antibody on a fully deglycosylated sample in a reduced CE SDS analysis (Figure 3). Lonza’s team tailored the analytical strategy for a customer molecule, developing a fit-for-purpose method within an accelerated timeline for lead clone selection.
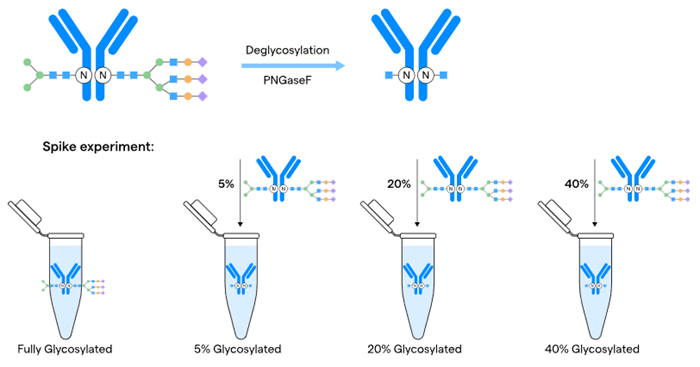
Figure 3. Method evaluation strategy using spiking for quantitative glycosylation. Image Credit: Lonza
Collaboration and innovation in analytical development
Jennifer considers these types of projects to be the most exciting because they offer an opportunity to collaborate with her talented and enthusiastic colleagues.
She highlights how rewarding it is to support members of the mammalian analytical development team in developing their expertise and skills while also helping to identify effective analytical strategies that allow Lonza’s customers to rapidly bring their complex molecules into the clinic with reduced risk.
Acknowledgments
Produced from materials originally authored by Jennifer Newman from Lonza.
About Lonza
Lonza is one of the world’s largest contract development and manufacturing organizations (CDMOs) dedicated to serving the healthcare industry. Working across five continents, our global team of approximately 20,000 colleagues works alongside pharma and biotech companies to turn their breakthrough innovations into viable therapies. We support our customers in bringing life-saving and life-enhancing treatments to patients worldwide with a combination of cutting-edge science, smart technology, and lean manufacturing.
Our company generated sales of CHF 6.5 billion with a CORE EBITDA of CHF 2.1 billion in Full-Year 2025.
Find out more at www.lonza.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.