Sponsored Content by LonzaReviewed by Ify IsiborApr 30 2026
Purifying novel therapeutics remains a major challenge as increasingly complex molecules move towards the clinic. With Lonza, you can ensure optimal product quality and bring your molecules to the clinic more efficiently.
By leveraging advanced downstream processing techniques, we empower our customers to navigate the complexities of novel therapeutic purification, ensuring their innovative treatments reach patients swiftly and effectively.
Cintia Carreria, Associate Principal Scientist, Purification Development at Lonza.
Addressing the purification needs of increasingly complex novel therapeutics
In recent decades, the biopharmaceutical industry has implemented established platform purification processes for monoclonal antibodies (mAbs). Improved knowledge frameworks in biology and disease pathways – combined with creativity in biomolecular design – have enabled innovative process development for many novel, protein-based formats that often form complexes with unfamiliar downstream processing challenges.
Cintia Carreria, Associate Principal Scientist, Purification Development at Lonza, has spent six years at Lonza, applying her experience in biochemistry and chromatography techniques to the design of custom purification solutions for:
- New molecular formats (e.g., non-Fc-containing molecules, enzymes, etc.)
- Bispecific antibodies
- mAb-like molecules
- Fusion proteins
Lonza’s highly skilled team capitalizes on its access to sophisticated high-throughput screening and analytical technologies, along with a global network of subject-matter experts. The team’s collaborative approach helps connect the different parts of downstream processing.
Challenges in process development
One of the greatest challenges during purification process development for novel molecular formats is their abstract, out-of-the-ordinary nature. As a result, there is often limited information available in downstream processing literature.
Moreover, for many complex non-mAb molecules, no “off-the-shelf” commercial affinity resins are available. While these formats can also undergo post-transcriptional modifications (PTMs), they may exhibit extreme acidity or basic isoelectric points (pI), and may be unstable or unstable under certain pH conditions, with elevated levels of impurities, including host-cell proteins (HCP).
As a result, tailored processes must be readily available. These bespoke approaches often take an unconventional, data-driven approach to align with the process requirements. With an acceleration in going from development to clinic, there is a growing need for swift timelines, and therefore, toolbox solutions are generally needed during process development.
The team at Lonza has the capability to conduct high-throughput chromatography, supported by the integration of high-throughput analytical methods into its toolbox. By focusing on screening chromatography resins, Lonza can determine product chromatography behavior and select the most appropriate operating conditions.
Rigorous downstream process development to achieve client goals
At Lonza, each project begins with a discussion about the molecule to build a clear understanding of its key characteristics. These include, where available, the impurity profile, the risk of aggregation, and any other attributes likely to affect downstream processing. That information serves as the basis for an initial development plan.
A recent process development case that Lonza worked on involved a unique non-Fc fusion protein. This molecule demonstrated novel characteristics, including a low acidic pI and exotic post-transcriptional modifications, which were critical to maintaining the product's potency and efficacy.
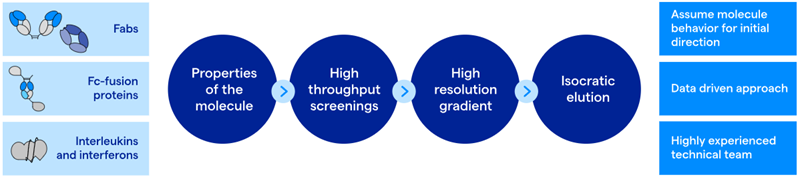
Figure 1. Downstream processing strategy for new molecular formats. Image Credit: Lonza
The capture step was subjected to stringent process development protocols, including high-throughput screening of various anion-exchange resins, which allowed Lonza to identify viable candidates. This is crucial when an affinity resin is not feasible. Upon resin selection, a high-resolution gradient on an ÄKTA FPLC was performed alongside fractionation to provide comprehensive details on product quality and impurities.
The gradient was then converted to isocratic elution to enable scalable operations. Here, the rigor of the step was examined to boost yield and enhance product quality, including optimizing wash conditions to eliminate additional HCP.
To first assess polishing steps, an examination screen of various resin types, including hydrophobic interaction (HIC), mixed-mode, and ion-exchange resins, was conducted. To support the resin screen, high-throughput analytical methods were employed, enabling the analysis of a large number of samples while maintaining rapid turnaround.
To piece the puzzle together, chromatography performance, impurity profile (e.g., HCP), and product quality data, informed by intelligent process design and optimal flow, were leveraged to design the potential final purification process.
The polishing resins with the most promise were further evaluated using high-resolution gradients and, subsequently, converted to isocratic elutions, enabling easier implementation and increased step reproducibility at scale. In particular, the HIC step demonstrated remarkable results that benefited the process; it showed a 3 LRV in HCP levels and reduced HMWS to ≤ 1 %. The final polishing step introduced enhanced durability during impurity reduction (DNA, HCP, and viral clearance), while refining molecule-specific product quality: the refinement of the correct PTM characteristics was supported by a high-resolution IEX resin with a thoughtfully designed separation scheme.
The toolboxes offered by Lonza, in combination with data-driven and high-throughput approaches, facilitate the development of bespoke purification solutions optimized for new molecular formats within tight turnarounds. All process development work is carried out in close collaboration with the Manufacturing Science & Technology team, ensuring development solutions can be easily rolled out in the production plant.
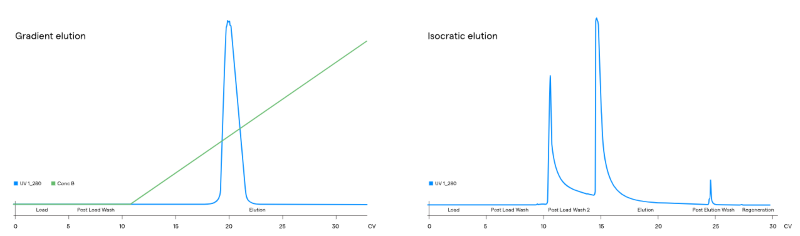
Figure 2. Example of a high-resolution gradient converted into an isocratic elution. Image Credit: Lonza
Achieving optimal downstream processing to help our customers bring their molecules to the clinic
Lonza toolboxes and data-driven approaches allow the company to swiftly develop new downstream processes for its customers. Integration of high-resolution gradients and novel design of experiments (DoE) into tailored development processes delivers durable functionality and optimization to the purification process.
Developing new processes that can be successfully transferred to Lonza’s manufacturing facilities is one of the company’s main drivers. The reward comes when it achieves key milestones such as successful GMP batch release and IND approval. Lonza is known for helping its customers bring new and important medicines to those who need them.
About Lonza
Lonza is one of the world’s largest contract development and manufacturing organizations (CDMOs) dedicated to serving the healthcare industry. Working across five continents, our global team of approximately 20,000 colleagues works alongside pharma and biotech companies to turn their breakthrough innovations into viable therapies. We support our customers in bringing life-saving and life-enhancing treatments to patients worldwide with a combination of cutting-edge science, smart technology, and lean manufacturing.
Our company generated sales of CHF 6.5 billion with a CORE EBITDA of CHF 2.1 billion in Full-Year 2025.
Find out more at www.lonza.com.
Sponsored Content Policy: News-Medical.net publishes articles and related content that may be derived from sources where we have existing commercial relationships, provided such content adds value to the core editorial ethos of News-Medical.net, which is to educate and inform site visitors interested in medical research, science, medical devices, and treatments.