Scientists are working to develop a new drug that will regenerate a critical enzyme in the human body that “ages” after a person is exposed to deadly chemical warfare agents.
Christopher Hadad, Ph.D., professor of chemistry at The Ohio State University (OSU), is leveraging Ohio Supercomputer Center (OSC) resources to help develop a more effective antidote to lethal chemicals called organophosphorus (OP) nerve agents.
“This project is a combination of synthetic and computational organic chemistry conducted through OSC at Ohio State, and biochemical studies conducted by colleagues at the U.S. Army Medical Research Institute of Chemical Defense at Aberdeen Proving Ground in Maryland,” said Hadad.
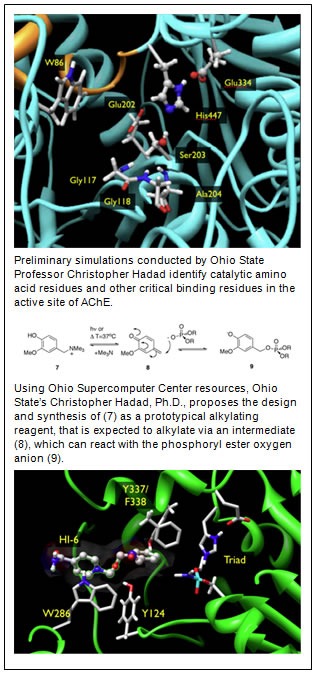 OP nerve agents inhibit the ability of an enzyme called acetylcholinesterase (AChE) to turn off the messages being delivered by acetylcholine (ACh), a neurotransmitter, to activate various muscles, glands and organs throughout the body. After exposure to OP agents, AChE undergoes a series of reactions, culminating in an “aging” process that inhibits AChE from performing its critical biological function. Without the application of an effective antidote, neurosynaptic communication continues unabated, resulting in uncontrolled secretions from the mouth, eyes and nose, as well as severe muscle spasms, which, if untreated, result in death.
OP nerve agents inhibit the ability of an enzyme called acetylcholinesterase (AChE) to turn off the messages being delivered by acetylcholine (ACh), a neurotransmitter, to activate various muscles, glands and organs throughout the body. After exposure to OP agents, AChE undergoes a series of reactions, culminating in an “aging” process that inhibits AChE from performing its critical biological function. Without the application of an effective antidote, neurosynaptic communication continues unabated, resulting in uncontrolled secretions from the mouth, eyes and nose, as well as severe muscle spasms, which, if untreated, result in death.
Conventional antidotes to OP nerve agents block the activity of the nerve agent by introducing oxime compounds, which have been the focus of a number of studies. These compounds attach to the phosphorus atom of the nerve agent, after the OP is bound to AChE, and then split it away from the AChE enzyme, allowing the AChE to engage with receptors and finally relax the tissues.
However, in some cases, the combined nerve agent/AChE molecule undergo a process called aging, in which groups of single-bonded carbon and hydrogen atoms called alkyl groups are removed from the molecule and a phosphonate residue is left behind in the AChE active site. Relatively unstudied in nerve agents, this process, called dealkylation, makes the nerve agent/AChE molecule unreceptive to oximes – an unfortunate situation, considering that certain nerve agents (e.g., soman) can undergo aging within minutes of exposure to AChE.
Hadad’s study is focused on the identification of compounds that would return an appropriate alkyl group to the aged nerve agent/AChE molecule, thus allowing treatment with oximes to provide for complete recovery. The project is investigating common OP nerve agents Tabun, VX, VR, Sarin, Soman, Cyclosarin and Paraoxon, all of which take on a similar molecular structure upon aging.
“Computational studies of the interaction of the alkylating compounds with AChE were used to provide insight for the design of selective reagents,” Hadad explained. “Ligand-receptor docking, followed by molecular dynamics simulations of the interactions of alkylating compounds with aged OP-AChE, was carried out in conjunction with experimental studies to investigate the binding of alkylating compounds to AChE. These results were then used to suggest interactions that aided in the orientation of alkylating compounds for maximal efficacy.”
Throughout the project, Hadad employed computational studies to guide the progress of each objective, as well as to rationalize the observed experimental results.
“Dr. Hadad’s work on this project has made use of a range of the tools of electronic structure theory, molecular docking, molecular dynamics and hybrid quantum mechanical/molecular mechanical methods,” said Ashok Krishnamurthy, interim co-executive director of OSC. “It was by design that OSC’s flagship system, the Glenn IBM 1350 Opteron cluster, was developed to meet the needs of the bioscience research investigators, such as Dr. Hadad.”
Hadad’s investigations of nerve agent antidotes are funded by the Defense Threat Reduction Agency (W81XWH-10-2-0044) and supported by the award of an OSC Discovery Account.