Australia is an acknowledged world leader in the medical technology, biotechnology and pharmaceutical (MTP) sector due to its vibrant ecosystem of start-ups and established companies and thriving research environment.
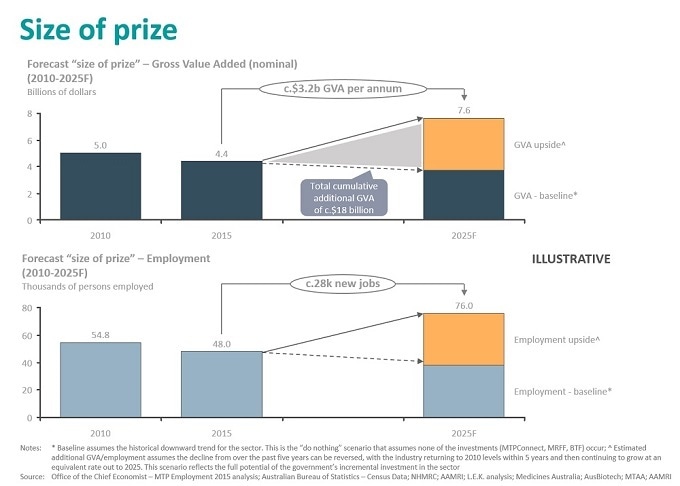
The MTP sector is a significant contributor to Australia’s economy, generating $4.4 billion gross added value (GVA), $4.6 billion in annual exports from manufacturing and employs more than 48,000 people across medtech, biotechnology and pharmaceuticals, and health and medical research.
MTPConnect | Medtech and Pharma Growth Centre
The MTP sector is on the cusp of enormous growth globally, due to a range of factors including changing demographics, ageing populations, the growth of emerging markets seeking better healthcare solutions, and the rapid development of new technologies and treatments that target individuals and the wider health system.
In order to remain sustainable and increase its economic and health contributions to the Australian economy, the sector must capitalize on existing strengths, as well as address issues of collaboration and commercialization and well-known barriers including funding, skills, policy and regulatory impediments.

How has the MTP sector in Australia changed over the last decade?
Historically Australia’s well-structured health sector, strong research competence and diverse demographics meant that we were an important part of the innovation ecosystem. The MTP sector has matured over the last 10 years and built on this strong base.
We are in an exciting time in Australia, with support for innovation and science providing an excellent environment for nurturing and development of pioneering and impactful new advances in the MTP sector.
We have sophisticated and now scaled research environments with world class infrastructure, leading scientists and clinicians and high standards of care. We have an internationally recognized regulatory system that includes Clinical Trial Notification (CTN) for rapid clinical trials approvals and strong intellectual properties (IP) frameworks. We also have strong ties with Asia supported by Free Trade Agreements (FTA’s) and an ethnically diverse population.
We are cost competitive and have some compelling Research & Development (R&D) tax incentive schemes and investment from the Biomedical Translation Fund (BTF) and Medical Research Future Fund (MRFF).
What opportunities exist for Australia’s MTP sector in Europe?
Europe is currently one of the most significant global MTP markets, however this is changing with emerging BRICS (Brazil, Russia, India, China and South Africa) and ASEAN (Association of Southeast Asian Nations) markets rapidly increasing in health expenditure.
Europe currently represents 23 per cent of the global MTP market, with an average of 10% of gross domestic product (GDP) spent in healthcare and 7.3 per cent of this attributed to medical technologies.
Australia’s MTP sector has an opportunity to become a leading player in the research, development and manufacturing of smart and connected medical devices. Our medtech sector comprises over 500 companies and has a strong international reputation for quality and a track record for being inventive in the areas of digital health, bionics and implants.
Australia can leverage its high‑quality production advantage in the short term, and collaborate over the longer term to develop solutions to deliver high quality and sophisticated technologies, products and healthcare to international investors and global companies.
Can you please give examples of Australian companies that are thriving in Europe? What can be learnt from these organizations?
The Australian MTP sector has produced numerous success stories, but two great examples of companies with international operating models and major manufacturing centers in Australia is Nanosonics and Cochlear Ltd.
Nanosonics is among the largest medical technology companies in Australia by market capitalization. Founded in 2001, Nanosonics was primarily a local medtech company, but now their focus has shifted to international markets where 90 per cent of the company’s revenues are generated, with offices in Europe and the US.
The company’s flagship device, the Trophon EPR, is a high-level disinfection system which has seen global adoption in recent years. Nanosonics financed its development through government grants, capital raisings and an eventual listing on the ASX in 2007. The company had its first sales of the Trophon EPR in Australia and New Zealand in early 2009 following TGA approval, with a European launch following later in the same year.
Cochlear Ltd is another success story of an Australian company with global operations. The company’s global headquarters is located on campus at Macquarie University in Sydney, however, Cochlear has direct operations in 20 countries and exports to over 100 countries.
For over three decades, Cochlear has been developing and manufacturing a range of implants, investing more than $100 million each year in research and development, with the majority of these R&D activities taking place in Australia.
Most of Cochlear’s manufacturing takes place in Australia and Sweden, with smaller sites in Belgium and the USA. In 2016, Cochlear’s Asia Pacific (APAC) sales totaled over $210 million (18% of total sales) while the major markets of Europe, the Middle East and Africa (EMEA) and the Americas totaled over $427 and $519 million respectively.
How does Europe view Australia’s MTP sector?
From our recent interactions at Australia’s Innovation Delegation to Europe, visiting France, Germany and Switzerland, it’s evident that there was a positive level of awareness of what Australia’s MTP sector can offer, given some of the relationships that exist with Max Planck, Fraunhofer and LyonBioPole and our companies, research organizations and CSIRO.
However, there wasn’t an in-depth understanding of the full product offering right across our MTP sector and the high caliber early research that Australia was undertaking in cutting edge areas including genomics, regenerative medicine, digital health and 3D printing.
The European market is an important market for our technologies and companies, in particular Germany is an acknowledged high quality advanced manufacturing country. Therefore, it is important for Australia’s MTP sector to share our capability in these areas to encourage further collaboration.
We already have an experienced Austrade team in Europe, with an established Landing Pad in Berlin, and we also have good contacts and relationships in Europe and can tap the sector into that global value chain.
What needs to be done to drive innovation further in this sector?
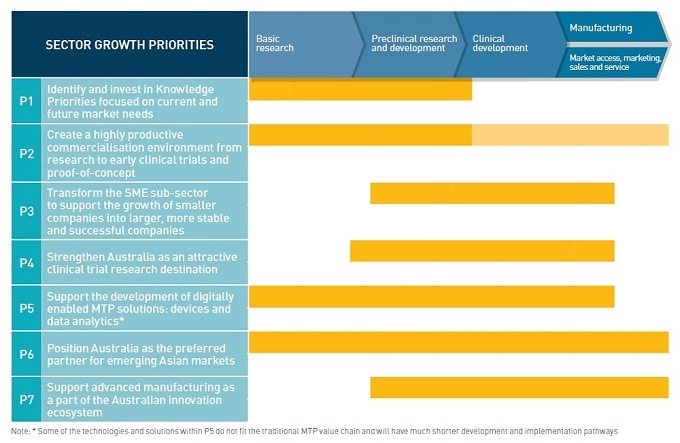
While Australia has a vibrant research sector, its commercialization productivity is behind that of other leading research countries and this is a focus for the sector as a whole. A recent report from Scientific America Worldview put us in 5th position down from 4th. The recent Pugatch Consortium “Race for Biopharmaceutical innovation” report places Australia 9th out of 10 mature markets.
Removing key barriers to growth and innovation, including lack of collaboration, policy instability, lack of commercialization skills, investment focus and the complexity of accessing global markets, is the key to growing our MTP sector and seizing global export opportunities.
Australia has created some successful MTP companies, such as Cochlear, CSL, Mesoblast, Nanosonics, ResMed, Bionomics and Starpharma. However, they are too few and the vision is to create more success stories.
Australia has a pool of start-ups with an inventive and entrepreneurial mindset. It is important to not only foster this culture to continue to drive innovation, but to also provide more support to translate start-ups and SMEs into bigger companies.
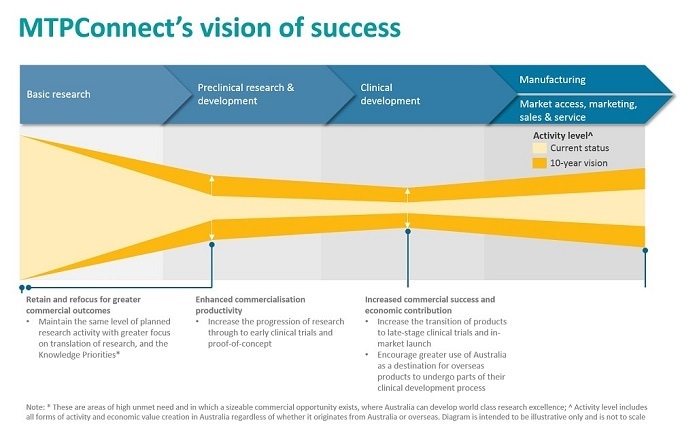
What is MTPConnect’s vision?
The MTP sector is in a significant position of having the ability to provide up to an additional 28,000 jobs and additional cumulative GVA of $18 billion over a 10-year period to the Australian economy, as well as translate its inventive MTP ideas into innovative solutions, to create a high-skilled and innovation-based economy for the future.
Our long-term vision is to achieve greater commercialization success, create more products that reach proof-of-concept and early stage commercialization, increase the number of medium to large companies with late-stage product successes and maximize the value of any intellectual-property, while retaining all current and planned levels of Research and Development (R&D). The overall effect would be greater employment and wealth creation for Australia.
Where can readers find more information?
Our Sector Competitiveness Plan (SCP) outlines a vision and strategy to build on our internationally acclaimed sector to maximize competitiveness and productivity, address key barriers, and support rapid and sustained growth through the yearly, dollar-for-dollar matched MTPConnect Project Fund Program. You can find more out more: https://www.mtpconnect.org.au/SCP
About Sue MacLeman
 Sue MacLeman is Managing Director and Chief Executive officer of MTPConnect. Sue has more than 25 years’ experience as a pharmaceutical, biotechnology and medical technology executive with roles in corporate, medical, commercial and business development at Schering-Plough Corporation (now Merck), Amgen, Bristol-Myers Squibb and Mesoblast Ltd.
Sue MacLeman is Managing Director and Chief Executive officer of MTPConnect. Sue has more than 25 years’ experience as a pharmaceutical, biotechnology and medical technology executive with roles in corporate, medical, commercial and business development at Schering-Plough Corporation (now Merck), Amgen, Bristol-Myers Squibb and Mesoblast Ltd.
Sue has also served as CEO and Board member of several ASX and NASDAQ listed companies in the sector and is currently a non-executive director at RHS Ltd and Oventus Medical Ltd.