The ongoing coronavirus disease (COVID-19), caused by SARS-CoV-2, has caused more than seven million infections and 400 thousand deaths worldwide as of June 10, 2020. Nonetheless, there are still tremendous gaps in our understanding of how the virus causes a spectrum of disease presentations, ranging from asymptomatic cases to lethal respiratory failures.
Another problem is that there is still no vaccine in sight, while the treatment is currently limited to supportive solutions aimed at alleviating symptoms. Accordingly, there is a pressing need to discover effective antiviral agents.
.jpg)
Novel Coronavirus SARS-CoV-2 Colorized scanning electron micrograph of an apoptotic cell (pink) heavily infected with SARS-COV-2 virus particles (green), isolated from a patient sample. Image captured at the NIAID Integrated Research Facility (IRF) in Fort Detrick, Maryland. Credit: NIAID

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Interferons and cholesterol
Interferons are a group of small, secreted proteins that can potently suppress the replication of many viruses through the concerted action of hundreds of interferon-stimulated genes. These genes are also upregulated in SARS-CoV-2 infected cells and lung tissues.
In any case, the identification of interferon-stimulated genes against different virus families has provided us with critical insights into both virus entry pathways and the evolution of host innate immune responses.
Cholesterol 25-hydroxylase is an interferon-stimulated gene that exhibits broad antiviral activity against a plethora of enveloped viruses. It encodes a cholesterol 25-hydroxylase that catalyzes the formation of 25-hydroxycholesterol from cholesterol.
And while the direct administration of interferons often results in potentially dangerous adverse effects in humans, a precisely targeted approach of activating the antiviral effects of specific interferon-stimulated genes holds substantial promise.
This is why researchers from the Washington University School of Medicine, Harvard Medical School, and Autonomous Therapeutics in the U.S., as well as from the Ocean University of China, decided to appraise cholesterol 25-hydroxylase and its enzymatic products as potent inhibitors of viral replication.
Cholesterol 25-hydroxylase and 25-hydroxycholesterol can inhibit SARS-CoV-2
To identify possible anti-coronavirus interferon-stimulated gene effector proteins that can act at the entry or exit points of the viral replication cycle, this research group utilized chimeric vesicular stomatitis virus (VSV) decorated with either full-length SARS-CoV or SARS-CoV-2 spike protein (S-protein) in place of their native glycoprotein.
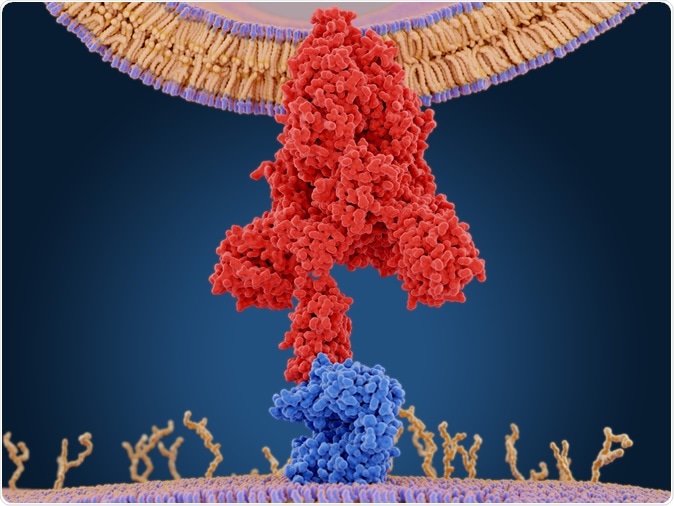
The coronavirus spike protein (red) mediates the virus entry into host cells. It binds to the angiotensin converting enzyme 2 (blue) and fuses viral and host membranes. 3d rendering. Image Credit: Juan Gaertner / Shutterstock
Such chimeric SARS-CoV-2 with an infectious molecular clone of VSV that expresses enhanced green fluorescent protein (eGFP) as a marker of infection have already been used in previous research endeavors and was touted as an optimal model for viral cycle research.
Collectively, the results of this study suggest a potent antiviral activity of cholesterol 25-hydroxylase and its natural product 25-hydroxycholesterol in suppressing SARS-CoV-2 virus infection. No cytotoxicity has been observed even at the highest concentrations tested.
When plaque assays (i.e., a common method for quantifying infectious viruses) were performed in the presence of 25-hydroxycholesterol, a notable reduction in both plaque numbers and sizes has been observed. Furthermore, wild-type SARS-CoV-2 virus replication was also inhibited by 25-hydroxycholesterol, but not by 7α, 25-dihydroxycholesterol treatment.
Mechanistically speaking, internalized 25-hydroxycholesterol is hoarded in the late endosomes and can successfully block cholesterol export – restricting, in turn, SARS-CoV-2 spike protein catalyzed membrane fusion.
"Our results highlight a unique antiviral mechanism of 25HC and provide the molecular basis for its possible therapeutic development", emphasize study authors in their tour de force research paper available on the bioRxiv preprint server.
Cholesterol redistribution impedes membrane fusion
Cell treatment with 25-hydroxycholesterol results in the reduced cell surface, but enhanced intracellular cholesterol levels. Therefore, it is convincing that 25-hydroxycholesterol acts at more than one subcellular compartment and that cholesterol redistribution leads to the inhibition of membrane fusion.
"Our data also instruct potential drug combinations of 25-hydroxycholesterol in conjunction with those targeting the cytoplasmic steps of the SARS-CoV-2 replication cycle such as its main protease or polymerase", accentuate study authors.
In any case, additional studies in animal models of SARS-CoV-2 infection and pathogenesis are definitely needed in order to establish the exact physiological impact of 25-hydroxycholesterol-based drugs or chemicals that modulate antiviral activities.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources