The ongoing COVID-19 pandemic has prompted intensive research into new vaccines and therapies, as well as into the immunological basis of protection. Now a new study by researchers at SUNY Upstate Medical University and published recently on the preprint server medRxiv* in September 2020 shows that memory B cells are upregulated in convalescent COVID-19 patients, correlating with a better immune response and shorter symptom duration.
The Role of Memory Cells
Immunological memory is a vital part of durable specific immunity to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). Based on earlier research, which shows COVID-19 patients to have altered immune profiles that can be grouped into one of several clusters, the current study focuses on finding cell-based immune markers for better clinical outcomes in convalescent plasma samples.
B cells respond to viral antigens by first secreting germline or near-germline antibodies from plasmablasts outside the follicles. Once the T cells bind to the CD40 surface markers, leading to the stimulation of specific cytokines, B cells enter a process called class switching. As a result, they are now found within the germinal centers inside several lymphoid organs and mature functionally. This leads to the production of both long-lived plasma cells and memory B cells that can respond to a repeated challenge with the same or another antigen.
Plasmablast expansion in the early B cell response has been suggested to lead to poor clinical outcomes. In contrast, memory B cells formed after SARS-CoV-2 infection offer durable specific immunity. They occur both in the classical CD27+ class-switched form as well as activated CD24- form and the natural CD27+ form, which resembles an innate immune cell, with both IgD and IgM markers.
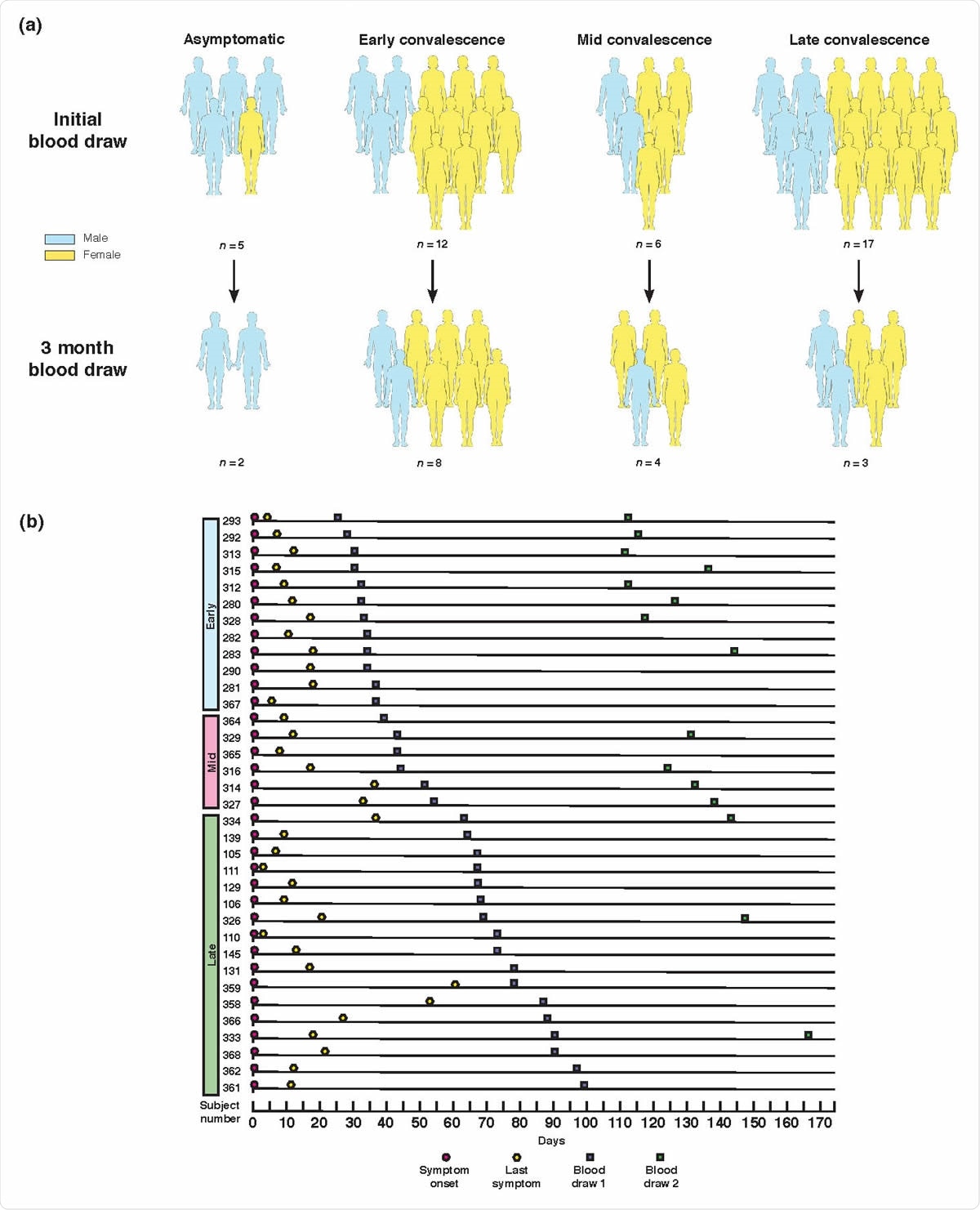
Study design and clinical data. (a) Graphical representation of convalescent subject groupings and gender. All grouping and subset designations were done retrospectively. (b) Symptom and sampling timeline for symptomatic subjects, ordered by length of convalescence.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Memory B cells Associated with Rapid Recovery
The researchers found that increased frequencies of memory B cells may mark a successful response to the acute infection and help understand the T cell response as well. Both switched and unswitched memory B cells correlated with a shorter duration of symptoms.
The extent of IgM+ memory was a strong correlate of the anti-RBD IgG1 antibody response. As a result, it appears that some COVID-19 patients do develop a protective memory response, either before or after the production of these IgM+ memory cells.
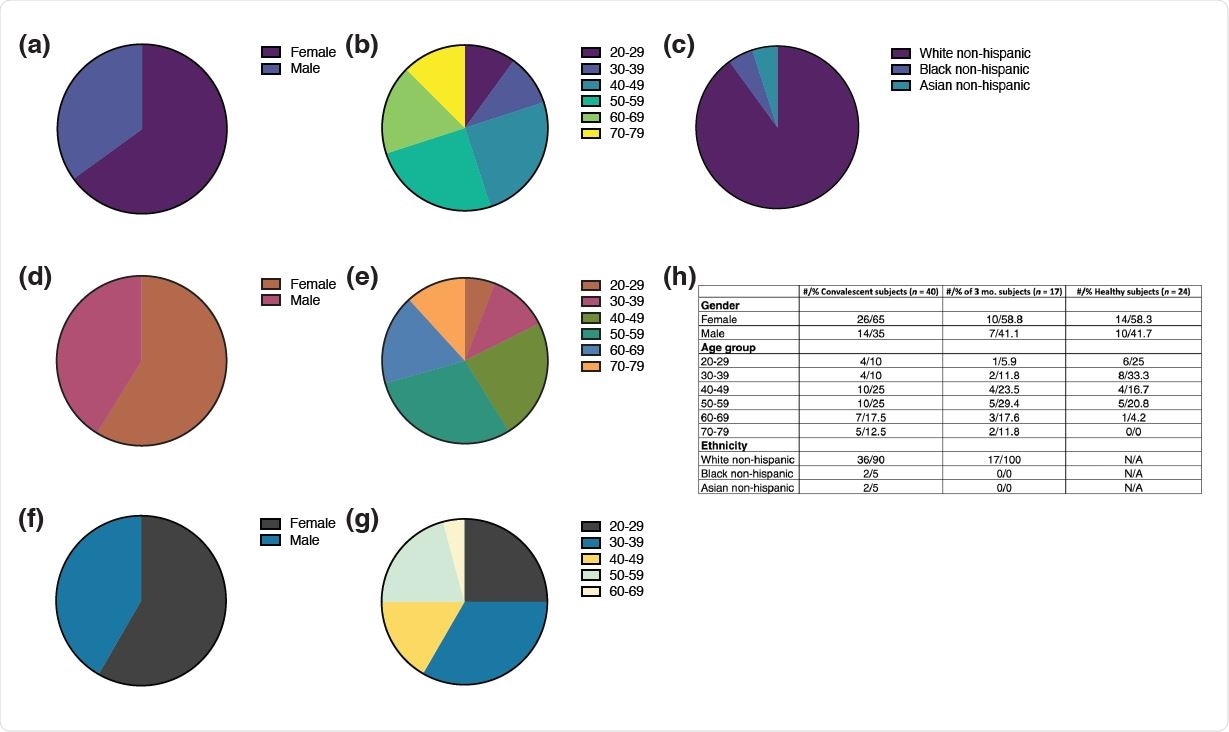
Demographic data. (a-c) Distribution of (a) gender, (b) age, and (c) ethnicity among the convalescent plasma donor cohort (n = 40). (d-e) Distribution of (d) gender and (e) age among healthy donors (n = 24). (f-g) Distribution of (f) gender and (g) age among the subset convalescent plasma donor cohort (n = 15) analyzed in figure 6 three months after initial visit. (h) Cumulative table of demographic data.
Mechanisms of Protection
The reasons suggested by the researchers are:
- Some of the B cell memory is cross-reactive against past coronavirus infections. Strangely, however, the anti-RBD IgG1 response is proportional to the IgM+ memory cells, since the latter do not secrete switched immunoglobulins. The researchers say, therefore, that perhaps coronavirus infections produce abundant IgM+ cells, of which some enter germinal centers and switch to IgG1 production.
- This ability of IgM+ memory cells to migrate to germinal centers after activation is well known, and may be of great benefit to induce immunity to agents like the coronaviruses with many highly similar strains.
- A seemingly paradoxical finding was that T bet+ B cell frequency did not correspond with that of resting memory B cells, since the former is key to forming B cell immunity. One explanation may be that they are not a vital part of convalescence, but are confined to acute and chronic viral infection. This aspect will need more research.
- Another reason for the higher recovery rates with increased memory B cells is these cells form part of a bigger pool, as a result of better T cell facilitation of B cell germinal center responses in naïve individuals during primary infection. This explanation is plausible since memory production is closely linked to the production of specific antibodies against the pathogen responsible.
This may throw light on the role played by the local immune environment, by spotlighting the importance of the germinal centers in providing the right milieu for T cell assistance. Where lymphoid tissue is destroyed or inefficient, severe COVID-19 has been observed. Also, in murine experiments involving severe bacterial infection within the cell, IgM+ memory played a protective role. This could indicate that disruption of the germinal centers due to SARS-CoV-2 actually enhances IgM+ B cell memory production. Thus, the study suggests that IgM+ memory B cells boost the plasma cells from germinal centers to provide long-term protection against repeat infection.
- Another hypothesis is that the expansion of B cell memory could merely mean that these individuals did not develop acute inflammation or hyperactive cytokine responses to the primary infection, instead veering towards an activated extrafollicular B cell profile, as has been seen to occur in systemic lupus erythematosus (SLE). Animal studies may help to clear up this point.
Implications
These findings show that “both switched and unswitched memory B cells in the maintenance of stable, durable SARS-CoV-2 immunity.” Notably, the current studies support earlier data showing that SARS-CoV-2 infection does produce long-term immunity and that some individuals have superior immunity due to their natural characteristics. In light of these findings, both IgM+ and switched memory B cells are essential to long-lasting immunity.
They are also potential markers for the effectiveness of vaccine strategies and for individual immunity status when faced with reinfection. Further prospective studies will show if increased numbers of memory B cells indicate pre-existing or immunologically elicited antiviral immunity.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Newell, K. L. et al. (2020). Switched and Unswitched Memory B Cells Detected During SARS-Cov-2 Convalescence Correlate with Limited Symptom Duration. medRxiv preprint. medRxiv preprint doi: https://doi.org/10.1101/2020.09.04.20187724. https://www.medrxiv.org/content/10.1101/2020.09.04.20187724v1
- Peer reviewed and published scientific report.
Newell, Krista L., Deanna C. Clemmer, Justin B. Cox, Yetunde I. Kayode, Victoria Zoccoli-Rodriguez, Harry E. Taylor, Timothy P. Endy, Joel R. Wilmore, and Gary M. Winslow. 2021. “Switched and Unswitched Memory B Cells Detected during SARS-CoV-2 Convalescence Correlate with Limited Symptom Duration.” Edited by Yolande Richard. PLOS ONE 16 (1): e0244855. https://doi.org/10.1371/journal.pone.0244855. https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0244855.