The unchecked advance of the coronavirus disease 2019 (COVID-19) has made it a top public health priority to produce a vaccine capable of inducing a safe and powerful protective response against its causative pathogen: severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). An encouraging new preprint, that appeared on the bioRxiv* server shows the immunogenicity, safety, and protective efficacy, of a Newcastle disease virus (NDV)-based intranasal vector vaccine in animal studies.
A host of candidate vaccines being developed against SARS-CoV-2 use viral vector platforms to present the SARS-CoV-2 spike antigen to the host immune system. Many have used the intramuscular route (i.e., ‘jabs’ or ‘shots’), but the intranasal route allows the development of local or mucosal immunity as well as systemic immunity against the virus. This will allow protection against not just symptomatic disease, but against the establishment of infection in the upper respiratory tract as well. This will help to break the chain of viral transmission effectively.
NDV platform
The NDV vector has multiple advantages for intranasal use. Firstly, it is an avian virus, and is highly attenuated in human cell lines. This means it will not cause any clinical symptoms, except a few flu-like short-lived conjunctivitis in humans. Several NDV strains are being used as oncolytic viruses in humans, due to their track record on safety. It is also genetically stable, and foreign transgenes have been inserted into the NDV genome without disrupting its replication.
Humans do not have pre-existing immunity against the virus, making it superior to other human viral vectors in current use, including the human adenovirus, measles virus, or the modified vaccinia Ankara virus vectors.
The NDV replicates in the respiratory tract, making it ideal to mount a protective mucosal response against SARS-CoV-2 infection. Finally, NDV replicates efficiently in chicken embryos and in other cell lines approved by the US Food and Drug Administration (FDA), allowing for rapid production.
Due to all these advantages, non-pathogenic strains of NDV have been used in multiple live attenuated vaccines (LAVs), with proven safety and efficacy in a host of animal models.
The researchers in the current study have already developed one such vaccine against SARS-CoV-2, expressing the full-length spike protein. This paper describes its results in a test of its immunogenicity and safety in mice and hamsters.
Intranasal vaccination leads to efficient non-pathogenic viral replication
The researchers found that the rNDV expressing the full-length SARS-CoV-2 spike glycoprotein replicated as efficiently as the wildtype virus in eggs and cell lines, and spreads within the infected cells.
When inoculated in mice, no adverse effects were found. Replication of the S-expressing rNDV or wildtype rNDV was less in the mouse gut compared to the respiratory tract.
The researchers found that in hamsters, earlier safety results in mice were confirmed, with no lasting weight loss or other adverse effects in vaccinated animals.
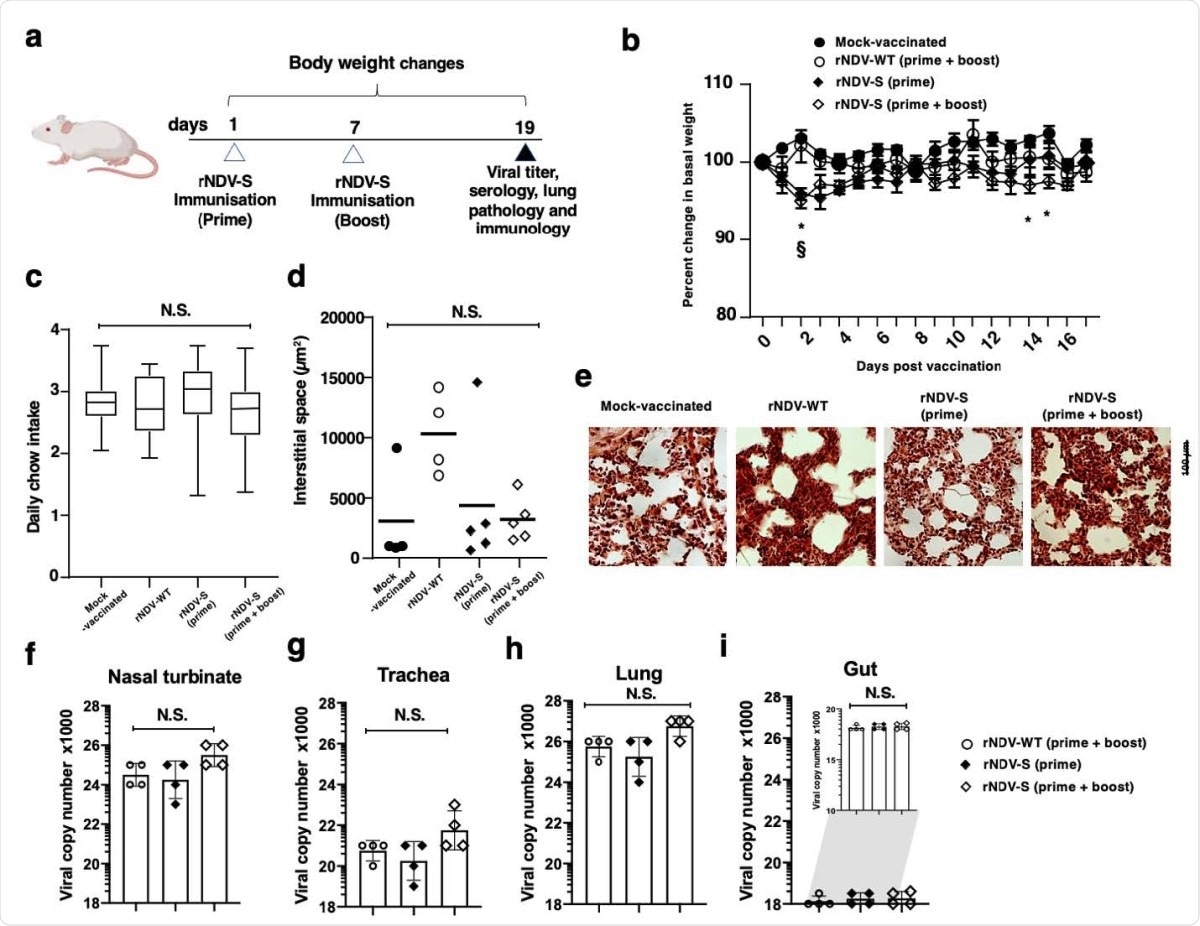
No adverse pathology results following rNDV-S intranasal vaccination. (a) Experimental model for the prime and prime + boost intranasal vaccination of mice. (b) Percentage weight change of mice following instillation of PBS or indicated rNDV constructs over the experimental time-course. (c) Daily chow intake by mock-vaccinated or rNDV-S vaccinated mice. Mean interstitial space size of right lung following H&E staining and assessed via ImageJ (d) and representative images (e). Viral replication of intranasally administered rNDV in nasal turbinate (f), trachea (g), lung (h) and gut (i). Data (n = 4–5 mice/group); *, P<0.05 and NS, non-significant between naïve and vaccinated groups, error bars represent SE of means via repeated t-test or ANOVA with Dunnett’s post-test.
Antibody response
They tested mice vaccinated with a prime-boost regimen of rNDV-S for both the total immunoglobulin G (IgG) antibody titers and neutralizing antibody titers, measured using microneutralization and pseudo-particle entry inhibition assays, respectively. They found robust neutralizing activity against an S-expressing pseudovirus, following rNDV-S vaccination with either one or both doses of vaccine.
The high IgA levels in lungs and serum, as well as IgA-secreting B cells, indicate robust induction of mucosal immunity after intranasal vaccination. The higher levels of IgG2a in the pleural fluid seem to suggest Fc-mediated antibody-dependent cellular cytotoxicity (ADCC), resulting in the killing of the infected cells.
In hamsters, too, effective antibody production was observed after the booster dose, indicating that both doses would be required for neutralization.
Absence of inflammatory changes
The researchers found that there were no signs of systemic inflammation, such as, for instance, spleen hypercellularity, or an increase in the number of myeloid cells. Dendritic cells were increased in mice immunized with wildtype or rNDV-S, but this was significant only in the former group. Lung cellularity was higher in the latter group, but again, there was a significant difference in myeloid cells, except in one subset of monocytes and macrophages. Overall, they found no evidence of long-term myeloid inflammation at the vaccination site or systemically after intranasal administration of this vaccine.
Lung inflammation was much lower in the vaccinated hamsters than in the controls, after a two-dose regimen. No infectious virus was detected in the respiratory tract from four days after the first dose of the vaccine. “These results demonstrate that vaccination with rNDV-S can protect from SARS-CoV-2 infection, resulting in lower SARS-CoV-2 titers and protection from lung damage.”
T cell responses
Intranasal rNDV-S vaccine administration showed a marked increase in the number of splenic CD4+ IFNγ-producing T-cells and NK TNF-producing cells, indicating a systemic T cell response to the S antigen. Within lung tissue, a significant decrease in CD4+ T cells and an increase in CD8+ T cells was seen in vaccinated animals. Memory T cells also appeared to be induced in lung tissue.
In both vaccinated and control groups (the latter having received wildtype rNVD), TNFα levels were reduced, while IFNγ production went up in the vaccinated group after the booster dose. The CD4 T cell population was reduced, but this was not reflected in the cells producing IL-17 or TNF. In fact, the vaccinated group showed a rise of over sixfold in the IFNγ-producing CD4+ T cell subset. This indicates that both locally and systemically, a specific IFNγ response was induced by intranasal vaccination.
Neutralizing response
The researchers observed an increase in fat associated lymphoid clusters (FALCs), which are locations in the pericardium and mediastinum that respond early to intranasal vaccination by local antibody production. Cellular proliferation marker Ki67 also showed an increase. Both IgA and IgG2a targeting SARS-CoV-2 spike receptor-binding domain (RBD) were increased significantly in the pleural fluid of vaccinated mice, indicating that a class switch of antibody production had occurred.
Since there was no increase in the number of B cells in pericardial or pleural fluid, these antibodies appear to have been produced earlier or resulted from a current systemic B cell response occurring within the primary lymphoid organs. Circulating antibodies that neutralized both full-length spike and RBD were observed.
In hamsters, two doses appear to be required to elicit a neutralizing response.
Implications
The rNDV-S induced robust systemic humoral and cell-mediated immune responses with two intranasal vaccine doses in mice and fully protected against lung infection, inflammation, and pathology after SARS-CoV-2 challenge in hamsters.”
By preventing infection at the site of viral entry into the body, this vaccine candidate may be able to prevent both disease and transmission, making it an ideal option for the achievement of sterilizing immunity against COVID-19.
Th1 cellular immunity appears to be essential in successful recovery from SARS-CoV-2 infection. In this study, spleen IFNγ-producing CD4+ T cells were increased after vaccination, while in the lungs, both CD4 and CD8 T cells produced more IFNγ. IFNγ is known to activate antiviral immunity. On the other hand, TNF secretion from lung CD8 T cells appeared to be reduced, even as the number of CD8 T cells went up. Apparently, this vaccine induces TNF-independent CD8 T cell-mediated cytotoxicity.
Such vaccines do not require needles, and may even be self-administered. Moreover, they may elicit mucosal immunity in the gut and genital tract, besides the respiratory tract. Further study may be needed to determine the optimal dose between the prime and booster doses, so as to achieve a robust memory response and higher antibody levels.
The researchers comment: “Based on these pre-clinical results in two rodent models, future clinical evaluation of rNDV-S in humans for the treatment of SARS-CoV-2 infection is urgently warranted.”

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Park, J.-G. et al. (2021). Immunogenicity and Protective Efficacy of an Intranasal Live-attenuated Vaccine Against SARS-CoV-2 in Preclinical Animal Models. bioRxiv preprint. doi: https://doi.org/10.1101/2021.01.08.425974,
https://www.biorxiv.org/content/10.1101/2021.01.08.425974v1
- Peer reviewed and published scientific report.
Park, Jun-Gyu, Fatai S. Oladunni, Mohammed A. Rohaim, Jayde Whittingham-Dowd, James Tollitt, Matthew D.J. Hodges, Nadin Fathallah, et al. 2021. “Immunogenicity and Protective Efficacy of an Intranasal Live-Attenuated Vaccine against SARS-CoV-2.” IScience 24 (9): 102941. https://doi.org/10.1016/j.isci.2021.102941. https://www.cell.com/iscience/fulltext/S2589-0042(21)00909-3.