Using publicly available coronavirus disease 2019 (COVID-19) data from Israel, a study suggests the BioNTech-Pfizer BNT162b2 vaccine is proving highly effective in the real world.
Several vaccines have now been approved for COVID-19, and many countries have started intensive vaccination programs. In Israel, vaccination started on December 20, 2020. By the end of January, 33% of the population had received the first dose of the BioNTech-Pfizer vaccine, also know as BNT162b2, and 19% had received their second dose.
Just as the vaccinations began, the country was in the midst of a third wave of COVID-19, with cases and hospitalizations more than doubling by mid-January 2021. In response, the country declared a lockdown on January 8, 2021. But, cases did not come down, and there were questions about the effectiveness of the vaccine.
However, it is challenging to estimate how effective vaccines are in the real world. Vaccination uptake is enmeshed with the socioeconomics and demographics of a population, along with disparities among groups in infection rates. Such confounding factors are not present in randomized clinical trials because of blinding. In the real world, clinical and demographic data at the individual level is needed to determine vaccine effectiveness.
Estimating vaccine effectiveness
In a paper published on the medRxiv* preprint server, Assistant Professor Dvir Aran, from Technion-Israel Institute of Technology, used publicly available data on COVID-19 cases and hospitalization after vaccination by the Pfizer vaccine. The author provides estimates of the effectiveness of vaccines in reducing cases.
The author used the daily positive cases and hospitalization data from the Israeli Ministry of Health COVID-19 public database, including hospitalization after vaccination up to January 31, counted the number of vaccinated individuals each day and multiplied that with the daily case rate of the whole population, adjusting for the difference in the number of cases between the vaccinated and general population. Using these parameters, the author estimated the vaccine effectiveness.
The analysis found there were 3,082,190 people who were vaccinated with a first dose between December 20, 2020, and January 31, 2021, and 1,789,836 had also received their second dose. Of the total vaccinated, 1,215,797 were above 60 years.
Among the vaccinated individuals, 31,810 tested positive for SARS-CoV-2 and 1,525 were hospitalized or died. The analysis indicates there was reduction of about 28% in the number of cases for those above 60 years by day 13 after the first dose, 43% reduction between days 14 and 21, and more than 80% reduction after the second dose.
This analysis is based on the assumption that all people who received their vaccines early on had the same level of positive cases as the general population. But, this is not true in the real world. Older people have lower positivity and lower socio-economic groups have higher positivity rates.
Adjusting for this, the author found if it is assumed that the vaccinated population has half the number of cases as the general population, there is no decrease in the number of cases up to 21 days after the first dose. The number of cases reduced by 66% seven days after the second dose.
For people over 60 years, who comprise a major proportion of the severely ill cases, the analysis suggests a strong effect of the vaccine, reducing severe cases by about 60% after the first dose and up to 94% seven days after the second dose.
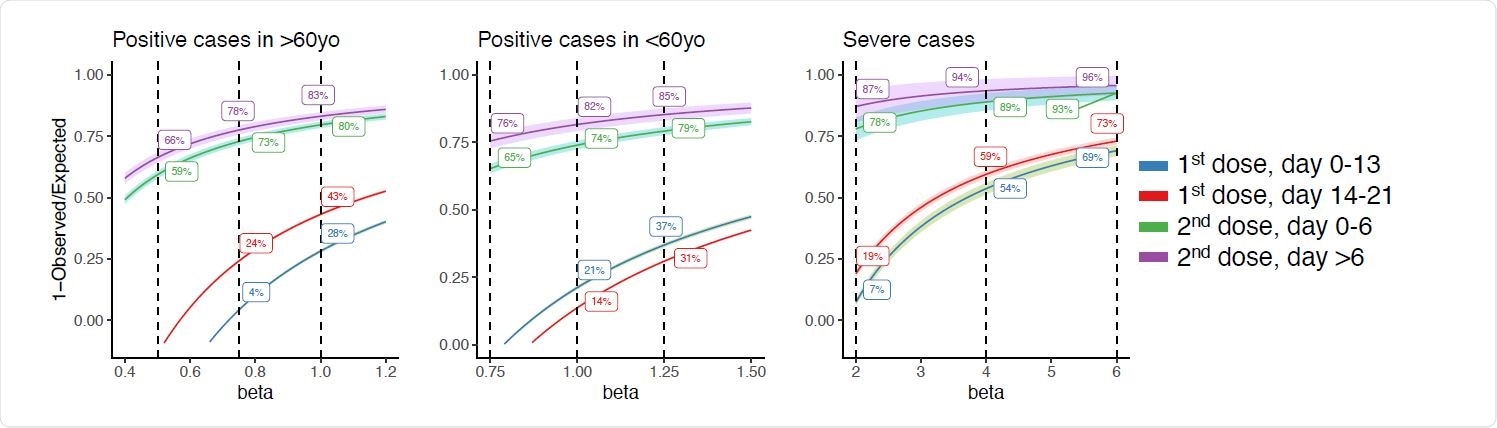
Effectiveness rate estimations of the vaccination by different levels of beta values. Standard errors are in shade.
Vaccine highly effective in real world
Overall, the analysis suggests a reduction in positive cases of 66-83% in people older than 60 years, 76-85% for those below 60 years, and 87-96% effective in preventing severe cases.
The Pfizer vaccine is reported to have an efficacy of 95% one week after the second dose based on the clinical trials, although efficacy before that is not clear. Analysis of the real-world data from Israel, which includes about 140-fold more individuals than the trial, provides an estimate of the effectiveness of the vaccine in reducing cases and the disease severity.
The author acknowledges some limitations of the analysis. These include delays in reporting cases, the fact that hospitalization may increase observed case counts, and limitations on making individual-level inference as the analysis used to aggregate counts. As the incidence in the general population is also affected by vaccination, the real effectiveness may be higher. The author writes the analysis provides “strong reassurance” that the vaccine is highly effective.

 *Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.
*Important notice: medRxiv publishes preliminary scientific reports that are not peer-reviewed and, therefore, should not be regarded as conclusive, guide clinical practice/health-related behavior, or treated as established information.