More than a year into the coronavirus disease 2019 (COVID-19) pandemic, caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the difference in the severity of the clinical syndrome by country-wide income levels has become clear. A new preprint, released on the medRxiv* server, examines some patterns revealed by a study of the factors operating in different regions and countries, to offer plausible hypotheses of these variations.
Several researchers have pointed to factors such as different scales and profiles of social interactions within households, endemic infections and median population age as affecting COVID-19 risk and mortality.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
However, these factors have not been found to form a connected framework whereby the risk of clinical severity can be predicted. The current study aimed to set up such a network of hypotheses linked to drivers of viral spread, age distribution among cases and fatalities, and population-level case fatality rates.
Ascertainment bias
The highest number of cases and of deaths due to COVID-19 has been reported in the Americas, while Africa contributes less than 3% of total deaths and an even smaller proportion of global cases, even though it harbors almost 15% of the world’s people.
The reasons offered for the seeming loss of clinical severity in low-income countries (LICs) include those relating to under-ascertainment (less testing, poor access to healthcare), and those relating to more effective or earlier control (proactive containment measures triggered by the warnings issuing from the earlier affected countries, and lower community transmission).
Reporting shortfalls in LICs are likely to affect the quality of data on reported cases and deaths by making it less representative. Testing rates and the prioritization of testing in scenarios marked by limited resources can thus significantly modify the apparent effects of age on the median age of cases and deaths, though not abolishing it.
Median population age
Demographic factors, including a larger proportion of younger people, and a larger proportion of healthy older people, could also have reduced the size of the fraction at risk for severe disease. Shielding of older people may also have been more effective due to the lower fraction of people in this age group.
Even after adjusting for population age structures, the researchers found that the distribution of cases and fatalities remained very different. The EURO and PAHO regions had a median case age of below 40 and a median age of case fatality of above 70. The reverse was the case in the AFRO region, with cases mostly above 40 and fatalities below 70.
Thus, the median population age was crucial to understanding differences in viral transmission, as well as infection and fatality rates. Even after age-standardization, the age structure of a population remains significantly associated with these rates.
In countries with an older population, the median age of death was higher, indicating the possible role of higher healthcare quality in minimizing avoidable deaths among younger people, even though they were more likely to be infected. Many deaths in such countries occurred among residents of long-term care facilities, who are mostly much older.
These countries also showed a younger age of death in people with chronic underlying comorbidities, perhaps mirroring the rise of such conditions in these countries at a lower age.
Household interactions
LICs are typically characterized by larger families, with several branches and generations sharing the same living space, which would be expected to counteract the favorable effects of the former, however.
While children appear to be both less susceptible and less infectious once infected with SARS-CoV-2, the role they play in its transmission is still unsettled. In the age group 11-18 years, children may drive transmission to a significant extent. However, this varies by age and geographical location.
In Europe, children and young people mix mostly with their peers, enabling the spread of disease across households. In south Asia, they mix with the rest of the family across generations. Overall, countries with a low median population age do not show the same profiles of household interactions as in Europe.
Such findings indicate lower risk in younger populations that a vulnerable older person in the family may come into contact with an infectious person. Thus, both the risk of infection, death and onward spread is lower.
Urban living and differences in the extent of urbanization could affect how much children mixed mostly with their peers compared to other age groups.
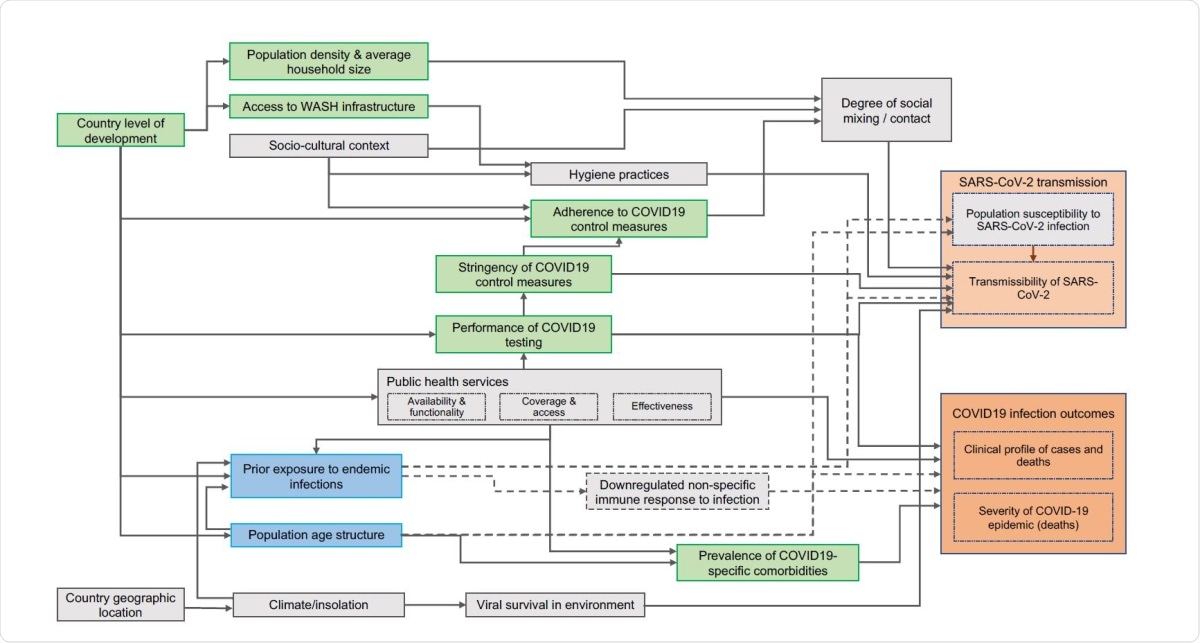
Proposed causal framework of factors determining SARS-CoV-2 transmissibility and COVID-19 disease outcomes. Boxes coloured in pink=outcome variables; boxes coloured in blue=exposures of interest; boxes coloured in green=covariates for which we obtained data; boxes coloured in grey=covariates and intermediate outcome variables for which we didn’t obtain data. Dotted lines represent hypotheses explored in this study.
Endemic infections
Pre-existing endemic infections may also have contributed to higher immunity against the virus at baseline, as they encourage a more diverse and competitive microbiome, higher innate immune responsiveness, and a reduced chance of the cytokine storm that characterizes severe COVID-19.
An earlier study links the relatively high protection against severe COVID-19 to widespread helmintic and parasitic infestations in many LIC populations, from early childhood onwards, with their strongly immunomodulatory effects. The immunomodulatory effects of endemic infestations and infections were discounted in the current study, though data is admittedly sparse.
Filariasis was globally observed to be inversely associated with COVID-19 prevalence. The researchers suggest the reason could be linked to the development of IgA (immunoglobulin A) antibodies, linked to mucosal immunity, in these individuals.
IgA antibodies drive a more anti-inflammatory response, as well as a skewed Th2 cellular immune response. This could counteract the tendency to develop a Th1-biased response in severe COVID-19 in those already exposed to filariasis. The result is a reduction in symptomatic SARS-CoV-2 infection and lower transmission rates.
Malaria prevalence, especially due to Plasmodium falciparum, also contributed to the risk of a transmissible infection, perhaps because this parasite causes the generation of antibodies that can interact with many different antigens. This may have allowed a greater level of pre-existing immunity to SARS-CoV-2 infection.
Further detailed research may help understand how pre-existing helmintic infestations affect COVID-19 disease risks.
What are the implications?
The study is limited by the poor ascertainment of cases in most LICs, for a variety of reasons, which leads to significant bias. The incomplete understanding of the virus itself further exacerbates the effect of such confounding factors.
Further studies on the sociodemographic correlates of COVID-19 are necessary to understand how it is transmitted across social interactions in a variety of settings, including urban and rural situations. This could help evolve broad guidelines to help with the timely management of future pandemics as well.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources