A majority of individuals who experience certain respiratory infections like the novel coronavirus disease 2019 (COVID-19) may only experience mild symptoms, whereas others can experience much more severe complications of the infection.
To date, there has been limited information that can predict who will have an asymptomatic presentation of these infections and those who will become seriously ill. Recent findings from a paper published in the Journal of Clinical Investigations describe how the presence of certain antibodies and antibody-secreting cells within the lungs can quickly induce an immune response to minimize the effects of these respiratory infections.
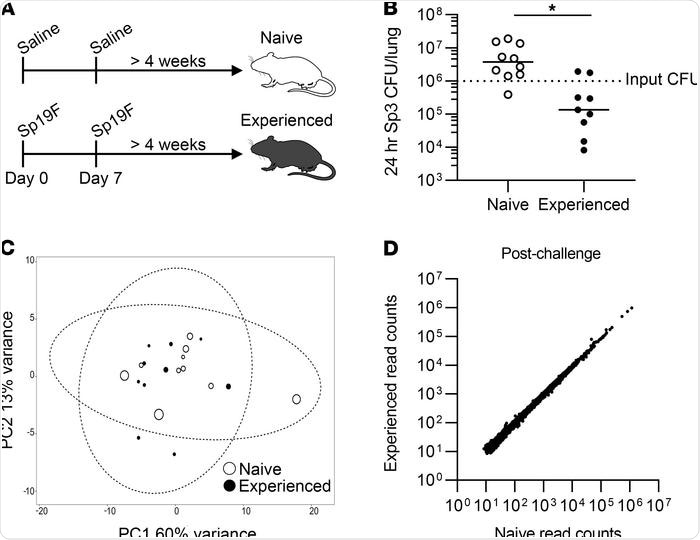
Pneumococcal exposures provide lung protection without extensive changes to the blood transcriptome. Image Credit: https://www.jci.org/articles/view/141810/figure/1
The immune cells of the lungs
Throughout the body, tissue-resident immune cells function as the frontline defense against potential pathogens, particularly at mucosal sites of constant environmental antigen exposure, like the lungs. Adaptive memory cells, in particular, play a significant role in supportive long-term protection against pathogens that have previously entered the host.
Within mucosal sites throughout the body, resident memory T (TRM) cells are the earliest immune cells to respond to bacterial and viral pathogens arriving at mucosal surfaces. Streptococcus pneumoniae, which is the viral infection that is responsible for most community-acquired pneumonia cases in the United States. Following a lung infection by this pneumococcus, heterotypic antibodies, long-lived plasma cells (LLPCs), and lung TRM cells will play pivotal roles in developing acquired immunity to this infection.
How does the immune response differ in viral and bacterial lung infections?
Previous work has confirmed that memory B cells (MBCs) can harbor cross-reactive antibodies against mutated strains of certain viruses, a group of researchers at the Boston University School of Medicine hypothesized that MBCs will play a similar protective role in the lungs following infection by multiple pneumococcal subtypes.
In their work, the researchers gave mice two exposures to pneumococcal infections and assessed the bacterial burdens in the animals’ lungs at the end of the treatment. The lungs of mice that recovered from pneumococcal infections were to contain BRM cells which provided serotype-independent anti-pneumococcal protection. A similar BRM phenotype was also observed in disease-free human lung tissues following their exposure to various respiratory pathogens such as pneumococcus and influenza. Between 50% and 70% of the human lung B cells identified on these tissue sections expressed CD27, which is a memory marker of B cells.
The stimulation of BRM cell production within the lungs, as well as several other types of B cells in the pleural fluid surrounding the lungs, and their subsequent production of neutralizing antibodies, was found to correlate with a reduced infection severity. More specifically, lungs that had recovered from infections that exhibited these cell populations were found to be better equipped at fighting microbes.
Improving the clinical management of lung infections
The findings of the current study provide more information on the components of lung immunity. As a result, this information could be used to predict whether certain patients are more susceptible to severe infections.
The identification of these individuals will better equip clinicians to treat patients who are likely to experience severe symptoms early on in the infection. Furthermore, clinicians could also use this information to determine which patients are more likely to require extensive care.
The authors of this study also believe that the production of novel types of vaccines for future influenza and coronaviruses could be developed based on their ability to induce this specific type of lung immunity.
Source:
- Barker, K. A., Etesami, N. S., Shenoy, A. T., et al. (2021). Lung-resident memory B cells protect against bacterial pneumonia. Journal of Clinical Investigation. doi:10.1172/JCI141810.