In the UK, researchers warn that a severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) variant of concern may cause epidemic outbreaks of similar magnitude to those associated with earlier waves of Coronavirus disease 2019 (COVID-19).
The team – from the University of Warwick, the University of Manchester, and the University of Oxford – say that a variant of concern that has either a substantial transmission advantage over resident lineages, or the ability to evade vaccine- or infection-induced immunity, can be expected to drive a wave of infections and hospitalizations similar to that seen in the country during the winter of 2020-21.
The team’s mathematical modeling analyses also revealed that a less transmissible variant than resident variants but exhibits even partial immune escape could trigger a wave of infections that would go undetected until non-pharmaceutical control measures were further relaxed.
“Given the prospect of the virus undergoing a continued accumulation of adaptive mutations, we should remain alert to all possible scenarios and continue the evidence-based analysis of evolutionary change so that public health measures can be adjusted in response to substantive changes in viral infectivity or severity of COVID-19,” warns Louise Dyson and colleagues.
A pre-print version of the research paper is available on the medRxiv* server, while the article undergoes peer review.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Concerns about current and future variants
As the COVID-19 pandemic continues globally, the ongoing incidence of SARS-CoV-2 infections increases the risk of the virus acquiring further advantageous mutations that could increase transmissibility, disease severity and escape from infection or vaccine-derived immunity. The number of countries reporting variants of concerns (VOCs) containing such mutations continues to increase.
The viral lineage B.1.1.7 that emerged in the UK in late Summer to early Autumn 2020 was designated a VOC on the 18th of December 2020.
“There is a consensus across multiple statistical and mechanistic modelling approaches that the B.1.1.7 variant has a substantial transmission advantage over pre-existing variants,” writes Dyson and colleagues.
More recently, the B.1.617.2 that emerged in India was designated a VOC in the UK on May 6th, 2021, based on assessments suggesting that it has “at least equivalent transmissibility to B.1.1.7.”
Even more worrying is the potential emergence of variants that evade previous immunity, thereby increasing the rates of re-infection and reducing the efficacy of vaccines.
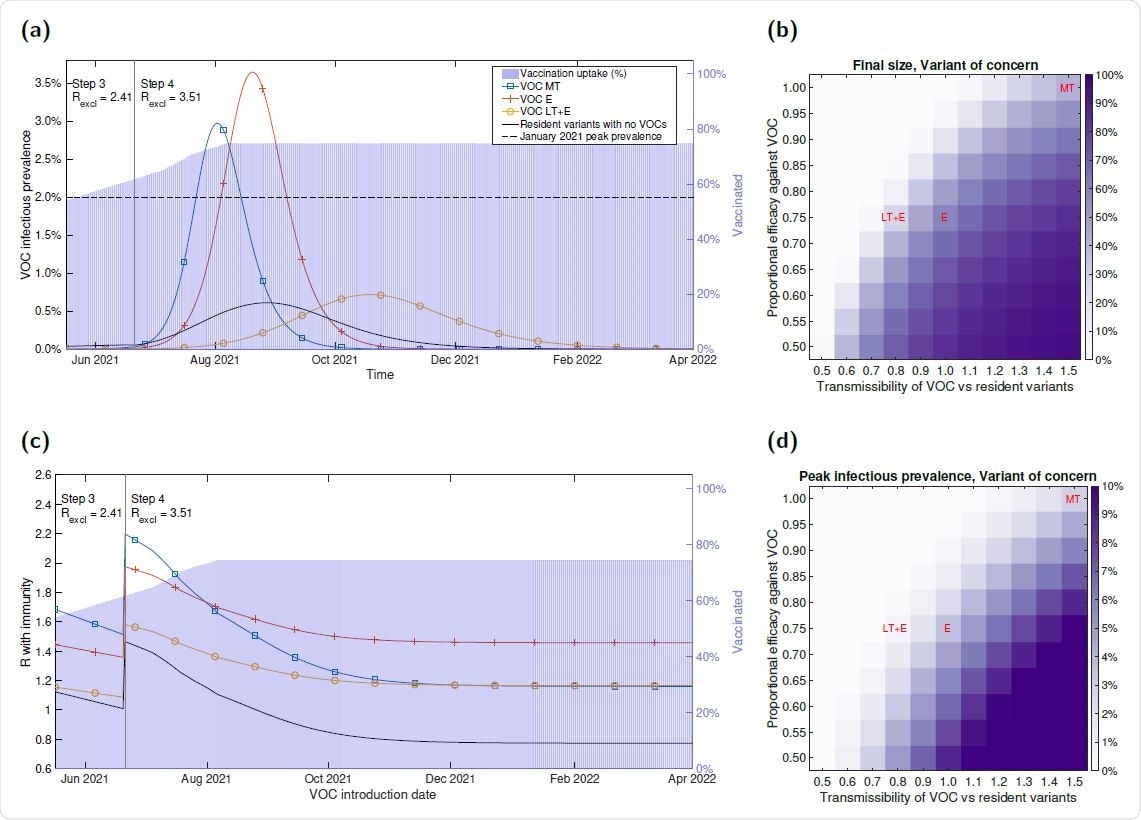
Infection burden for illustrative VOC scenarios, produced using the parsimonious SARS- CoV-2 transmission model. We considered three putative VOCs with differing transmissibility and immune escape characteristics: more transmissible (VOC MT, blue line with square markers), equal transmissibility with immune escape (VOC E, orange line with plus sign markers), less transmissible with immune escape (VOC LT+E, yellow line with circle markers), and resident variants alone in the absence of any VOC being introduced (black line with no markers). Additionally, in panels (a&c) we represent the vaccine uptake in the population through time via background shading, the transition time into Step 4 of the relaxation roadmap by the vertical solid line and we state the assumed R excluding immunity values for resident variants (Rexcl) throughout Steps 3 and 4, respectively. (a) Infectious prevalence over time. In each scenario, alongside resident variants, we introduced one of the VOCs on 17th May 2021 with 2,000 initial infections. (c) R with immunity (y-axis) with respect to the date of a VOC being introduced (x-axis). For the `Resident variants with no VOCs' scenario, the displayed profile corresponds to the instantaneous R with immunity of resident variants. In panels (b&d) we explore the sensitivity of three epidemiological outcomes to the relative transmissibility of the VOC compared to resident variant and the proportional efficacy (vaccine and natural-immunity) against the VOC: (b) outbreak final size; (d) peak in VOC infection cases.
Concerns are growing that as countries with high vaccine coverage begin to relax non-pharmaceutical interventions (NPIs), variants that escape existing immunity may arise and cause new waves of infection.
“The infectious disease dynamics of SARS-CoV-2 result from a complex interaction between the circulation of multiple variants, vaccination, NPI policy and adherence,” say the researchers.
“ Mathematical modeling approaches are an avenue for testing sensitivity of these dynamics to underlying assumptions and conveying uncertainty, with the caveat that models must balance biological realism with mathematical and computational tractability and parameter identifiability,” writes the team.
What did the researchers do?
The team used three mathematical models of SARS-CoV-2 variant dynamics to evaluate two scenarios for third waves of the pandemic: one that is driven by increased transmissibility and one that is driven by immune escape.
The models included a parsimonious susceptible-latent-infectious-recovered (SEIR) model, an age-structured transmission model and a stochastic importation model.
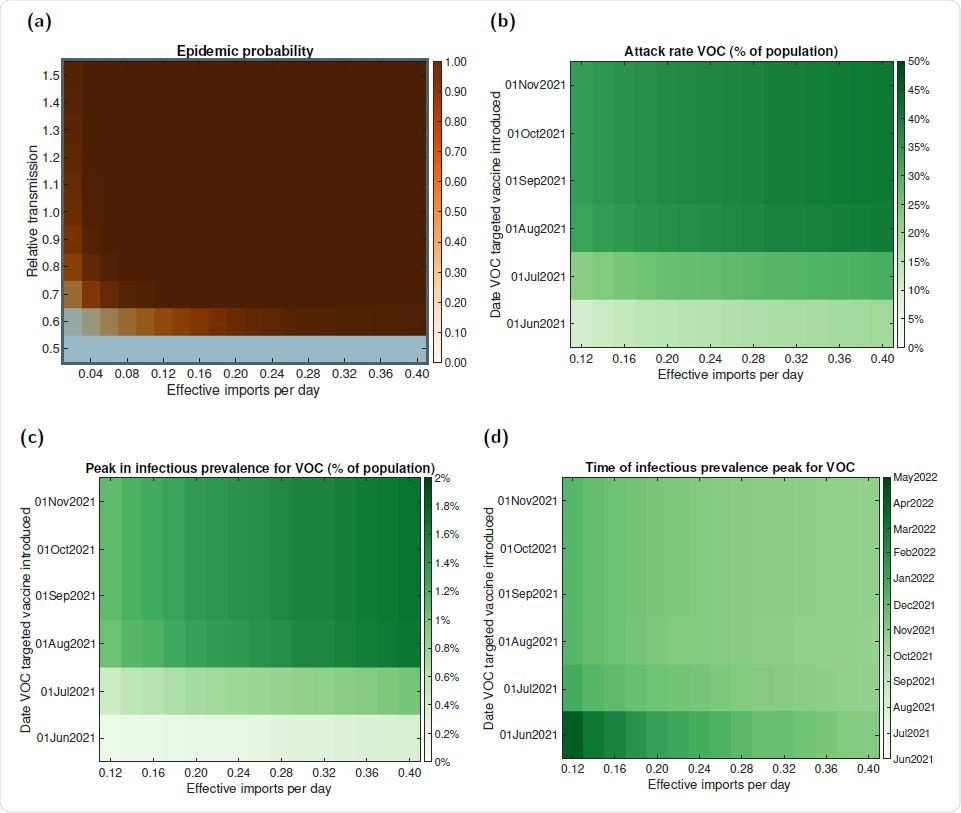
Outbreak potential and sensitivity of epidemic trajectories to the introduction time of a VOC targeted vaccine for VOC E. (a) The probability of an epidemic for varying relative transmissibilities (compared to resident variants) versus a given count of VOC effective imports per day (corresponding to the second generation cases that result from a single index case). In panels (b-d), we performed simulations using the parsimonious SARS-CoV-2 transmission model for differing effective VOC importation counts and introduction date of a VOC targeted vaccine and evaluated the following epidemiological summary statistics for the resultant VOC outbreak: (b) final size; (c) peak in infectious prevalence; (d) time of peak in infectious prevalence.
The models were calibrated to the situation in England in May 2021 so as to accurately capture infection dynamics and vaccination rates and to explore the potential impact of a putative new VOC-targeted vaccine.
What did they find?
The epidemiological trajectories for the VOCs were shown to be wide-ranging and highly dependent on transmissibility, immune escape properties, and the time at which a VOC-targeted vaccine may be introduced.
The modeling revealed that a VOC could generate a wave of infections and hospitalizations comparable in magnitude to that seen in the winter of 2020-21 if it possesses either a considerable transmission advantage over resident variants or the ability to evade prior immunity.
Furthermore, even when a variant is less transmissible than resident variants, just partial immune escape could trigger a wave of infection and hospitalizations.
In addition, the reduced transmissibility may mean the VOC would remain difficult to detect until NPIs were relaxed.
“As we continue with the vaccination rollout, reducing the level of NPIs could reveal less-transmissible immune-escape variants, which were previously kept in check by control measures,” says the team.
The timing of a VOC-targeted vaccine is important
The study also pointed to a critical interplay between the timing of a VOC-targeted vaccine and the number of VOC imports.
When the number of imports per day was sufficiently low (less than 0.5 per day), introducing a new vaccine early enough had an appreciable effect on the VOC epidemic curve.
Generally, any single cluster of VOC infections was most easily controlled when the case count was low.
“Early detection and efforts to extinguish infection clusters is therefore paramount, as increased importation rates seed more clusters and will necessitate additional resources to keep under control,” writes Dyson and colleagues.
Close monitoring of SARS-CoV-2 evolution is essential
The team says there is no reason to believe that the SARS-CoV-2 virus has yet settled at its fitness optimum in terms of replication and transmission capabilities.
“A concerted international COVID-19 pandemic response requires global situational awareness of how the virus is mutating and identification of emergent variants that are of concern,” says Dyson and colleagues.
“Close monitoring of the evolution of SARS-CoV-2 across a range of geographical scales is needed to enhance local situational awareness and quantify risk from variants that may be of concern, with reliable and accurate data ensuring outputs from models of infectious disease dynamics are as informative as possible,” they conclude.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Dyson L, et al. Possible future waves of SARS-CoV-2 infection generated by variants of concern with a range of characteristics. medRxiv, 2021. doi: https://doi.org/10.1101/2021.06.07.21258476, https://www.medrxiv.org/content/10.1101/2021.06.07.21258476v1
- Peer reviewed and published scientific report.
Dyson, Louise, Edward M. Hill, Sam Moore, Jacob Curran-Sebastian, Michael J. Tildesley, Katrina A. Lythgoe, Thomas House, Lorenzo Pellis, and Matt J. Keeling. 2021. “Possible Future Waves of SARS-CoV-2 Infection Generated by Variants of Concern with a Range of Characteristics.” Nature Communications 12 (1). https://doi.org/10.1038/s41467-021-25915-7. https://www.nature.com/articles/s41467-021-25915-7.