It is largely understood that the vaccination of a majority of the world’s population is the only measure that can stop the spread of the severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2). SARS-CoV-2 is a ribonucleic acid (RNA), positive sense, and respiratory virus that is responsible for the coronavirus disease 2019 (COVID-19).
As of August 20, 2021, SARS-CoV-2 has already claimed more than 4.4 million lives worldwide and infected over 211 million. Researchers around the world have worked at a record speed to develop vaccines and therapeutics to contain the pandemic.
 Study: Effectiveness of COVID-19 Vaccines among Incarcerated People in California State Prisons: A Retrospective Cohort Study. Image Credit: Dan Henson / Shutterstock.com
Study: Effectiveness of COVID-19 Vaccines among Incarcerated People in California State Prisons: A Retrospective Cohort Study. Image Credit: Dan Henson / Shutterstock.com

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Background
Several COVID-19 vaccines have received emergency use authorization from various global regulatory bodies, which has led to the quick initiation of vaccination programs in these countries across the globe. Of these include the two messenger RNA (mRNA) vaccines of BNT162b2 (Pfizer-BioNTech) and mRNA-1273 (Moderna), both of which have shown high efficacy in managing SARS-CoV-2 infection among various age groups, communities, and nursing facilities.
Despite these observations, there remains a gap in research related to the efficacy of vaccines in congregate settings such as prisons and jails, where the risks of transmission of SARS-CoV-2 are very high. Jails are often extremely populated with poor ventilation; therefore, maintaining physical distancing in these settings is often difficult.
Many prisoners have been reported to suffer from severe COVID-19. As a result, approximately half of the states in the United States have prioritized this group of individuals for COVID-19 vaccination. However, others states adopted a different approach and placed incarcerated people in the lowest priority tiers.
In December 2020, the California Department of Corrections and Rehabilitation (CDCR), which is known to be the second-largest state prison system in the U.S., commenced the COVID-19 vaccination program. CDCR soon scaled up the vaccination program across 35 prisons. Among these, 14 conducted COVID-19 testing and recorded data relevant to COVID-19.
About the study
In a new retrospective cohort study published on the preprint server medRxiv*, researchers determined the efficacy of mRNA vaccines (BNT162b2 and mRNA-1273) against confirmed COVID-19 infection among incarcerated people in California prisons. The authors of the current study collected COVID-19 data from CDCR that included approximately 61,000 incarcerated people between December 22, 2020, and March 1, 2021.
The data provided by the CDCR contained information on the demographic, clinical, and carceral characteristics of the subjects. It also included data on COVID-19 testing, vaccination status, and outcomes of these candidates.
The authors estimate vaccine efficacy using multivariable Cox models with time-varying covariates that have been adjusted for resident characteristics and infection rates across prisons.
Study findings
The current study revealed that both mRNA vaccines were highly effective against confirmed SARS-CoV-2 infection among the racially diverse population of the study cohort. Among the 60,707 residents, 49% received at least one dose of the BNT162b2 or mRNA-1273 vaccine during the study period.
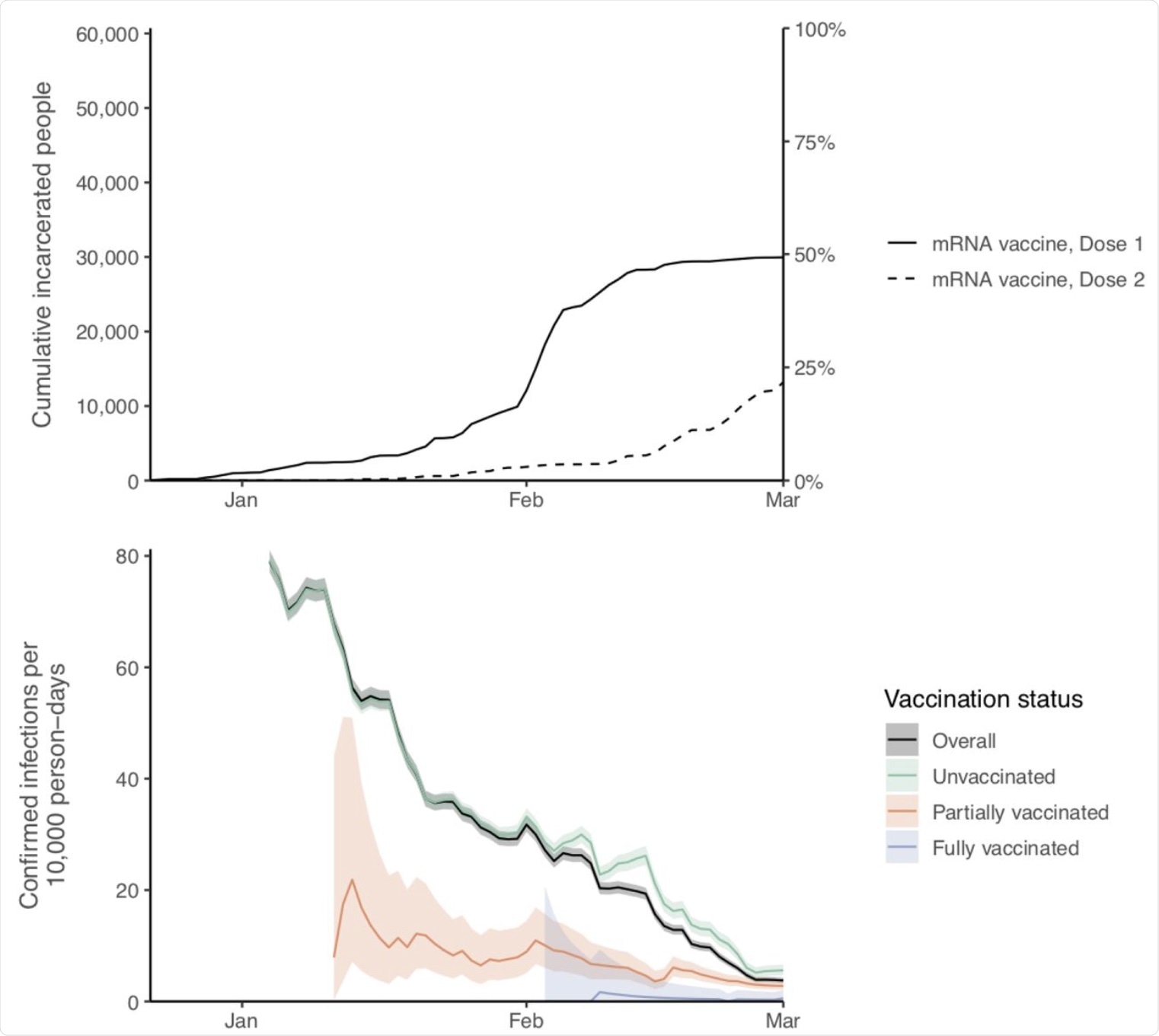 Cumulative vaccinations with one or two doses of mRNA vaccines (top panel) and 14-day rolling rates of confirmed infections per 10,000 person-days by vaccination status (bottom panel), among study cohort of incarcerated people in California state prisons.* Shaded areas represent 95% confidence intervals. Partially vaccinated status defined as ≥14 days after a first dose until receipt of a second dose; fully vaccinated status defined as ≥14 days after a second dose. * Time periods with fewer than 200 people tested were excluded.
Cumulative vaccinations with one or two doses of mRNA vaccines (top panel) and 14-day rolling rates of confirmed infections per 10,000 person-days by vaccination status (bottom panel), among study cohort of incarcerated people in California state prisons.* Shaded areas represent 95% confidence intervals. Partially vaccinated status defined as ≥14 days after a first dose until receipt of a second dose; fully vaccinated status defined as ≥14 days after a second dose. * Time periods with fewer than 200 people tested were excluded.
The researchers estimated the effectiveness of the COVID-19 vaccines at 14 days after receiving the first dose of the vaccine and 14 days after the second dose of the vaccine. In the first scenario, the vaccine effectiveness was estimated to be 74%. Additionally, vaccine efficacy increased to 97% after full vaccination, which is 14 days after receiving the second dose of the vaccine.
The researchers of the current study also estimated the vaccine effectiveness among the high-risk residents, owing to their medical history and pre-existing conditions. The effectiveness of BNT162b2 was 74% 14 days after the first dose and 92% after these individuals were completely vaccinated. Similarly, the effectiveness of the mRNA-1273 vaccine was estimated to be 71% at 14 days after the first dose and 96% upon complete vaccination.
Significance
The results obtained in the current study align with previous studies conducted in the United Kingdom and Israel related to the effectiveness of mRNA vaccines among the residents and staff in skilled nursing facilities and other frontline workers. Comparably, this is possibly the first study that evaluates the effectiveness of a COVID-19 vaccination program in a carceral setting.
The main strength of this study is the incorporation of detailed daily information on vaccination status and COVID-19 outcomes of each resident. Another strong point is the large sample size that helped evaluate the vaccine effectiveness within particular subgroups of interest, such as medically vulnerable candidates.
Limitations
The researchers have pointed out several limitations of this study. For instance, vaccine effectiveness has been focused on confirmed infections; however, other important outcomes, such as symptomatic infections or severe disease were not considered.
During the study period, the rate of hospitalizations and deaths was low; therefore, relating these aspects with vaccine effectiveness would not be accurate. Another limitation of this study is that the effectiveness of the vaccine against specific SARS-CoV-2 variants was not assessed. This is because CDCR conducted limited viral genome sequencing during the study period. Additionally, the antigen test kits used by CDCR to detect COVID-19 symptoms have low sensitivity; therefore, there is an increased likelihood that case rates were under-detected.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Chin, T. E., Leidner, D., Zhang, Y., et al. (2021) Effectiveness of COVID-19 Vaccines among Incarcerated People in California State Prisons: A Retrospective Cohort Study. medRxiv. doi:10.1101/2021.08.16.21262149. https://www.medrxiv.org/content/10.1101/2021.08.16.21262149v1.
- Peer reviewed and published scientific report.
Chin, Elizabeth T, David Leidner, Yifan Zhang, Elizabeth Long, Lea Prince, Stephanie J Schrag, Jennifer R Verani, et al. 2022. “Effectiveness of Coronavirus Disease 2019 (COVID-19) Vaccines among Incarcerated People in California State Prisons: Retrospective Cohort Study.” Clinical Infectious Diseases 75 (1): e838–45. https://doi.org/10.1093/cid/ciab1032. https://academic.oup.com/cid/article/75/1/e838/6515675.