Presently, there is no licensed antiviral drug in the market that specifically treats the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. Considering the devastating effects caused by the coronavirus disease‐19 (COVID-19) pandemic, identifying antiviral drugs that are effective against SARS-CoV-2 is of the utmost importance.
Heme oxygenase-1 (HO-1) is a cytoprotective enzyme that can be induced by hemin, Cobalt protoporphyrin-9 (CoPP-9), and andrographolide. HO-1 acts by attenuating the inflammatory response and oxidative stress inside cells.
HO-1 catalyzes heme degradation to carbon monoxide, biliverdin, and iron, all of which have been reported to exert antiviral effects. The non-enzymatic action of HO-1 has been reported, wherein it inhibits influenza A infection through its interactions with interferon (IFN) regulatory factor 3, which results in the activation of the type I IFN response.
Hemin is an iron-containing porphyrin compound that is a known HO-1 inducer whose antiviral effects against viruses such as the hepatitis A virus have been already reported. HO-1 has been reported to exhibit an immunomodulatory and antiviral effect against viruses affecting the respiratory system, such as the influenza virus and respiratory syncytial virus.
Although an earlier publication has hypothesized that hemin-induced HO-1 may exert anti-viral activity against SARS-CoV-2, no evidence was presented. The current Scientific Reports study explores the antiviral activity of HO-1 induced by hemin against SARS-CoV-2 and attempts to describe the mechanism of its anti-viral action.
Hemin exhibited dose-dependent antiviral activity against SARS-CoV-2
Induction of HO-1 by hemin and the consequent antiviral effect against SARS-CoV-2 was assessed using in vitro assays on Vero 76 cells. Cells were infected with SARS-CoV-2 after pre-treatment with hemin.
A dose-dependent increase in HO-1 messenger ribonucleic acid (mRNA) and protein levels, accompanied by a dose-dependent decrease in SARS-CoV-2 RNA and viral nucleoprotein content, was subsequently observed, thereby indicating the antiviral activity exerted by hemin.
The scientists also performed time of addition experiments to determine at what stage of the viral infection hemin exerts its action. Suppression of viral protein expression was evident when cells were exposed to hemin, even after viral entry. This suggests that similar to remdesivir, hemin may inhibit the SARS-CoV-2 intracellular replication phase of its life cycle.
Additionally, immunofluorescence assays showed that there was a decrease in the number of Vero 76 cells containing the SARS-CoV-2 spike protein as the concentration of hemin increased at 24 hours post-infection.
Transient overexpression of monkey HO-1 in Vero-76 cells using expression vectors was found to inhibit SARS-CoV-2 replication in these cells. This indicates that HO-1 contributes to the observed antiviral effect and porphyrin in hemin may not be solely responsible as earlier reports suggest in the case of the human immunodeficiency virus (HIV).
Through cytotoxicity assays, it was found that hemin exhibited a 50% cytotoxicity concentration (CC50 ) of 169.9217 micromolar (μM), whereas its 50% effective concentration EC50 was found to be 0.6805 μM. Selectivity index (SI), which is the ratio of CC50 and EC50, gives the measure of the window between cytotoxicity and antiviral activity. The SI for hemin was found to be 249.7, thereby suggesting that CC50 was approximately 249 times higher than EC50 confirming that hemin is an effective and safe therapeutic agent.
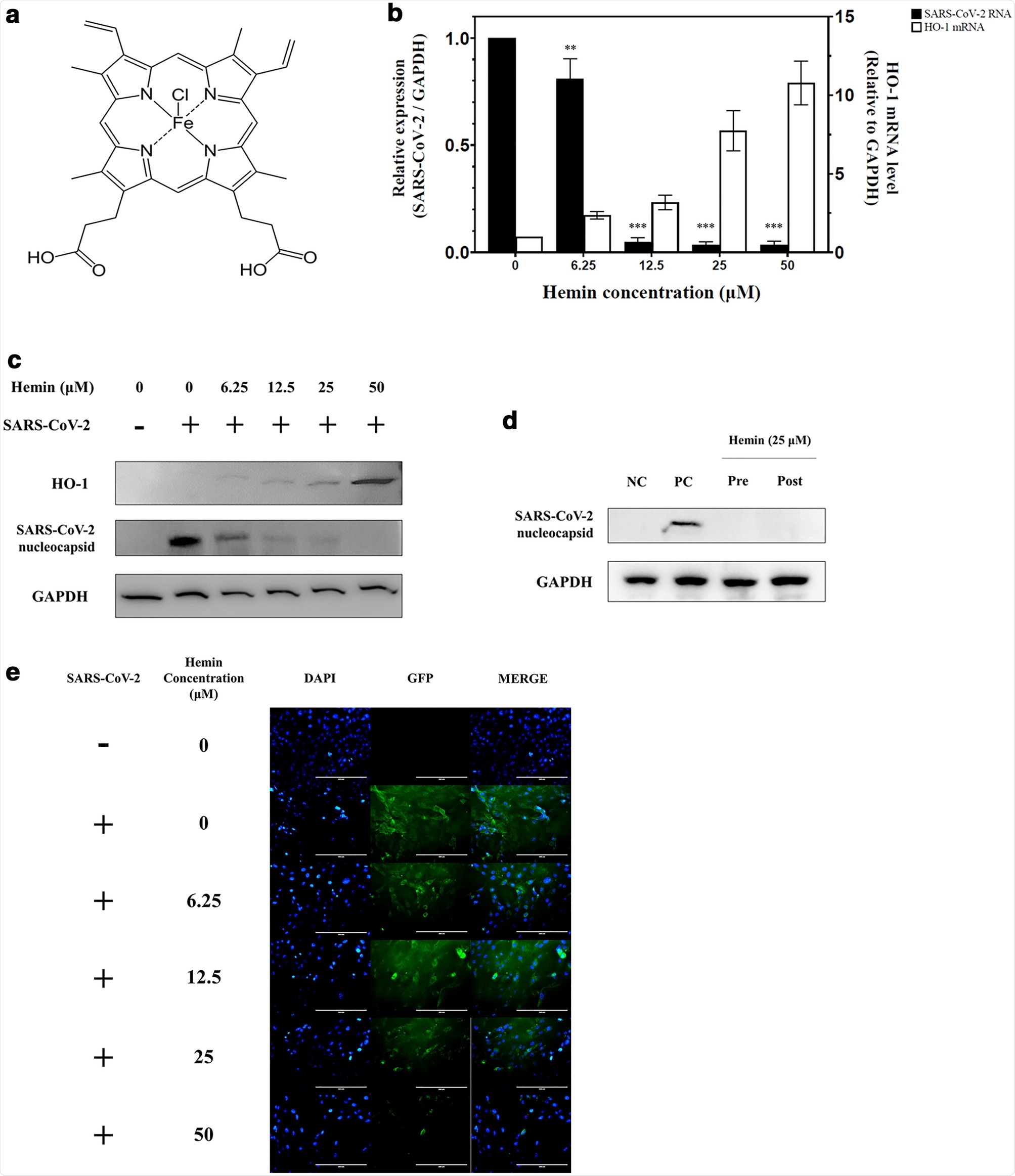 Changes in the induction of SARS-CoV-2 RNA and HO-1 expression in Vero76 cells at different concentrations of hemin. (a) Structure of hemin. The molecular formula of hemin is C34H32ClFeN4O4. (b) Vero76 cells infected with the SARS-CoV-2 strain NCCP43326 (multiplicity of infection (MOI): 0.001) were treated for 24 h without or with hemin at various concentrations. As the hemin concentration increased, the HO-1 mRNA expression increased, as expected, and the SARS-CoV-2 RNA expression decreased concomitantly. (c) Changes in the expression of HO-1 and SARS-CoV-2 nucleocapsid protein at different hemin concentrations. As the concentration of hemin increased, the width of the HO-1 protein band was observed to increase, and the viral protein content decreased. Comparison with the levels of GAPDH, a housekeeping protein, showed that equal quantities of cells were used in this experiment (also see Supplementary Fig. S4a). (d) Time-of-addition experiment with hemin. In the pre-treatment group (Pre), Vero76 cells were pre-treated with hemin for 1 h, and then infected with SARS-CoV-2. In the post-treatment group (Post), the cells were treated with hemin for 2 h after SARS-CoV-2 infection; NC (negative control): MOCK control, PC (positive control): only infected with SARS-CoV-2. (also see Supplementary Fig. S4b) (e) Cellular distribution of SARS-CoV-2 spike proteins. Vero76 cells pre-treated with hemin at various concentrations were infected with SARS-CoV-2 (MOI: 0.001). Immunofluorescence assay (IFA) for SARS-CoV-2 spike protein was conducted 24 h post-infection; green fluorescence: SARS-CoV-2 spike protein; blue fluorescence: DAPI staining for nuclei. Results of the IFA were analyzed using fluorescence microscopy. Original magnification: × 200. Data are presented as the mean ± standard deviation of at least three independent experiments. **P < 0.01, ***P < 0.001.
Changes in the induction of SARS-CoV-2 RNA and HO-1 expression in Vero76 cells at different concentrations of hemin. (a) Structure of hemin. The molecular formula of hemin is C34H32ClFeN4O4. (b) Vero76 cells infected with the SARS-CoV-2 strain NCCP43326 (multiplicity of infection (MOI): 0.001) were treated for 24 h without or with hemin at various concentrations. As the hemin concentration increased, the HO-1 mRNA expression increased, as expected, and the SARS-CoV-2 RNA expression decreased concomitantly. (c) Changes in the expression of HO-1 and SARS-CoV-2 nucleocapsid protein at different hemin concentrations. As the concentration of hemin increased, the width of the HO-1 protein band was observed to increase, and the viral protein content decreased. Comparison with the levels of GAPDH, a housekeeping protein, showed that equal quantities of cells were used in this experiment (also see Supplementary Fig. S4a). (d) Time-of-addition experiment with hemin. In the pre-treatment group (Pre), Vero76 cells were pre-treated with hemin for 1 h, and then infected with SARS-CoV-2. In the post-treatment group (Post), the cells were treated with hemin for 2 h after SARS-CoV-2 infection; NC (negative control): MOCK control, PC (positive control): only infected with SARS-CoV-2. (also see Supplementary Fig. S4b) (e) Cellular distribution of SARS-CoV-2 spike proteins. Vero76 cells pre-treated with hemin at various concentrations were infected with SARS-CoV-2 (MOI: 0.001). Immunofluorescence assay (IFA) for SARS-CoV-2 spike protein was conducted 24 h post-infection; green fluorescence: SARS-CoV-2 spike protein; blue fluorescence: DAPI staining for nuclei. Results of the IFA were analyzed using fluorescence microscopy. Original magnification: × 200. Data are presented as the mean ± standard deviation of at least three independent experiments. **P < 0.01, ***P < 0.001.
Metabolite byproducts of heme degradation catalyzed by HO-1 exert antiviral activity
HO-1 catalyzes the breakdown of heme to metabolites like free iron, biliverdin, and carbon monoxide (CO). The induction of HO-1 expression by hemin may lead to an increase in metabolite byproducts due to heme breakdown.
The scientists evaluated the antiviral activity of CO-releasing molecule (CORM), which induces CO production, iron III chloride (FeCl3), and biliverdin against SARS-CoV-2. The findings suggested that FeCl3 and biliverdin significantly inhibited SARS-CoV-2 replication, while CORM did not have an effect. This finding confirms the antiviral effect of the enzymatic action of HO-1.
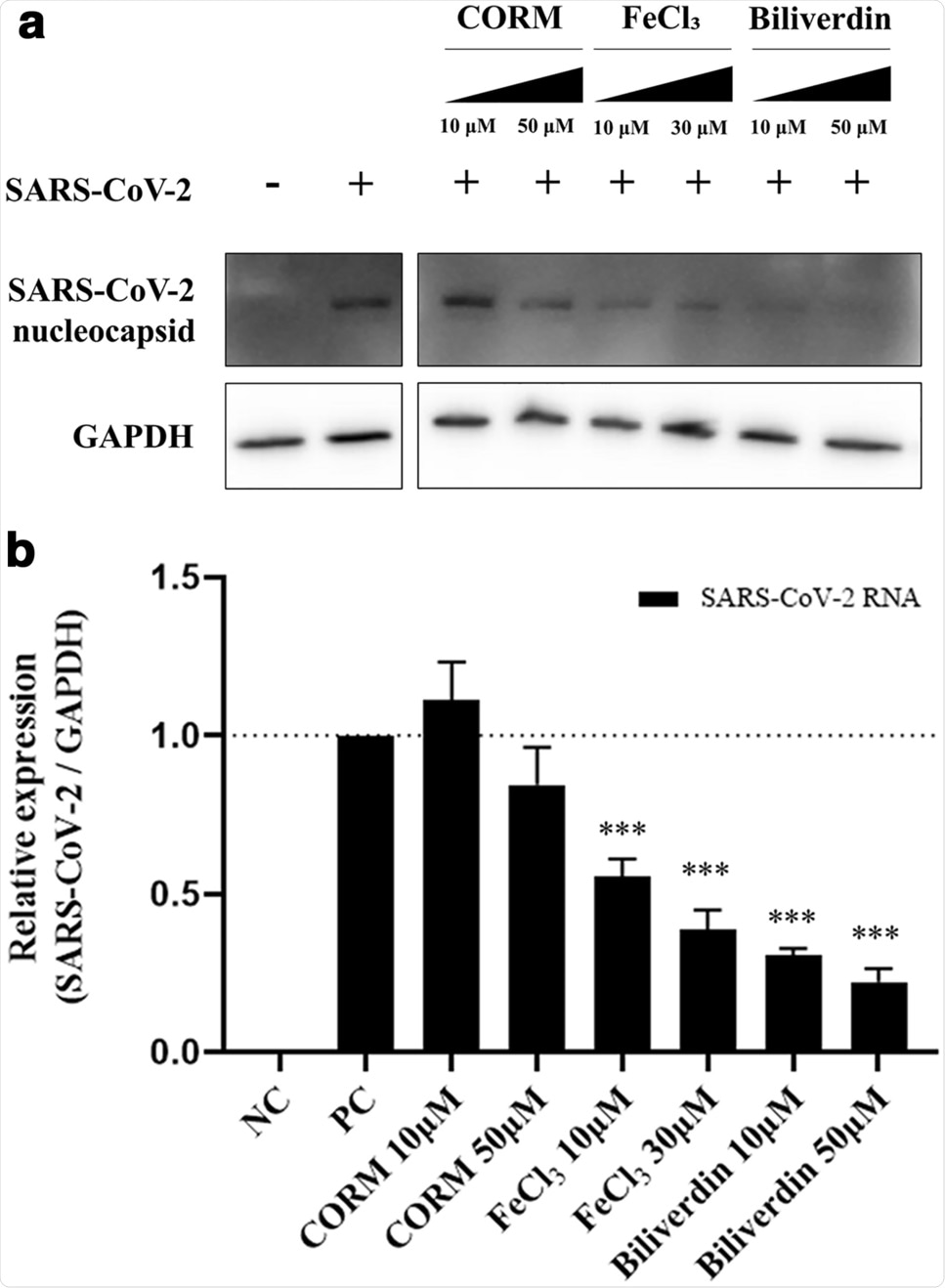 Antiviral efficacy of the metabolites produced by HO-1. The inhibition of SARS-CoV-2 proliferation by carbon monoxide, iron ions, and biliverdin produced by HO-1 was confirmed. Treatment with CORM (releases carbon monoxide), FeCl3, and biliverdin was performed 1 h before SARS-CoV-2 infection. At 24 h post-infection, (a) western blot assay (also see Supplementary Fig. S4d) and (b) qPCR were performed to compare the changes in the viral protein and RNA contents. ***P < 0.001.
Antiviral efficacy of the metabolites produced by HO-1. The inhibition of SARS-CoV-2 proliferation by carbon monoxide, iron ions, and biliverdin produced by HO-1 was confirmed. Treatment with CORM (releases carbon monoxide), FeCl3, and biliverdin was performed 1 h before SARS-CoV-2 infection. At 24 h post-infection, (a) western blot assay (also see Supplementary Fig. S4d) and (b) qPCR were performed to compare the changes in the viral protein and RNA contents. ***P < 0.001.
Additional antiviral activity against SARS-CoV-2 by HO-1 that is independent of its enzymatic activity
The current study further explored if the enzymatic activity of HO-1 played a role in its antiviral effect against SARS-CoV-2. Assays were performed, wherein Vero 76 cells were treated with both hemin and zinc protoporphyrin-9 (ZnPP-9), which is a known inhibitor of HO-1 enzymatic activity.
When compared to cells treated with hemin alone, there was a higher reduction of SARS-CoV-2 RNA and protein in the cells that received both ZnPP-9 and hemin. However, when HO-1 expression was knocked down using short interfering RNA against HO-1, the antiviral activity was significantly inhibited. The findings indicate an additional antiviral effect of HO-1, which may be attributed to activities that are distinct from its enzymatic actions.
Interestingly, the scientists also observed that in cells treated with just ZnPP-9, there was a low level of HO-1 expression and inhibition of SARS-CoV-2 replication. Previous reports suggest that though ZnPP-9 inhibits HO-1 enzymatic activity, it increases the expression of HO-1 proteins. The elevated levels of HO-1 may exert their antiviral effect indirectly through the IFN regulatory factor-3 (IRF3)-mediated pathway.
The study further explored if the hemin-induced expression of HO-1 results in an increase in levels of IFN-stimulated gene (ISG) proteins such as OAS1, Mx1, and ISG15. It was found that the expression of ISG proteins was increased in Vero 76 cells, irrespective of whether they were infected with SARS-CoV-2 in the presence or absence of hemin pre-treatment.
Cells that were pre-treated with hemin and subsequently infected with SARS-CoV-2 showed elevated expression of ISG proteins, while the viral nucleocapsid protein was not detected in these cells. This indicates the antiviral activity of ISG proteins.
Conversely, ISG proteins induced by the viral infection alone in the absence of hemin did not exert an inhibitory effect on viral replication, which was evident by the detection of viral nucleocapsid protein in these cells. This finding suggests that ISG proteins induced by SARS-CoV-2 infection do not effectively inhibit viral replication.
A possible explanation for this effect is that SARS-CoV-2 induces ISG proteins at a later stage when the viral replication is complete, while HO-1 induces ISG protein expression through the IRF3-mediated pathway at an earlier stage, effectively inhibiting viral replication.
Conclusions
The hemin product that was used in this study to induce HO-1 is commercially available in the market as PANHEMATIN® and has a well-established safety profile. The findings from this present study demonstrate that hemin exhibits significant in vitro antiviral activity against SARS-CoV-2 by inducing the expression of HO-1.
Hemin may prove to be a safe and effective antiviral drug therapy for SARS-CoV-2 infection. The findings from this study may pave the way for developing effective therapeutics that can combat COVID-19.