Since its emergence in late 2019, the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) has infected 275 million worldwide and caused over 5.3 million deaths. The global public health burden of the coronavirus disease 2019 (COVID-19) has resulted in the rapid development of several vaccine candidates, some of which are currently being used for large-scale vaccinations.
However, the emergence of novel SARS-CoV2 variants of concern (VoC), as well as reduced efficacy of vaccines against these new viral strains, have raised concerns these VoCs are capable of evading vaccine-mediated immunity. Therefore, continued vaccine development activities are required to improve protection and reduce the transmission of SARS-CoV-2 variants.
Study: SARS-CoV2 variant-specific replicating RNA vaccines protect from disease and pathology and reduce viral shedding following challenge with heterologous SARS-CoV2 variants of concern. Image Credit: LookerStudio / Shutterstock.com

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
The HDT-301 vaccine
Previous studies have reported the development of a Venezuelan Equine Encephalitis Virus (VEEV)-based replicating ribonucleic acid RNA (repRNA) vaccine that encoded the spike protein of the original A.1 lineage of SARS-CoV-2 and was delivered with the help of a Lipid InOrganic Nanoparticle (LION).
This technology has been transitioned through the current good manufacturing practices (cGMP)-compliant, thus leading to a new drug status under the drug product name HDT-301. HDT-301 is a B.1.351-specific vaccine that is pending Phase I trials in the United States. It is also being evaluated in India under the drug product designation HGC019 in Phase I clinical trial.
A new study published on the preprint server bioRxiv* evaluated the repRNA vaccine expressing the SARS-CoV-2 spike protein for the B.1.1.7 and B.1.351 variants in mice and hamsters. The delivery of the vaccine was mediated by LION and was administered through an intramuscular injection.
About the study
The current study included both in vitro and in vivo assays. For the in vitro assays, the virus was cultured in two Vero cell lines followed by the construction of spike variants.
The mouse studies included 6-8 weeks old female C57BL/6 mice that were vaccinated with 1 microgram (µg) of the vaccine on days 0 and 28. The mice were then monitored for their neutralization activity against the SARS-CoV-2 A.1, B.1, B.1.1.7, and B.1.351 variants.
Comparatively, the hamster studies included 7 to 8-week-old male Syrian Golden hamsters that were acclimatized and assigned to study groups before vaccination. The hamsters were vaccinated with 20 µg of repRNA complexed to LION on day 0 and boosted four weeks later.
The antibody responses were monitored by the collection of blood 25 days after the first vaccination and 14 and 21 days after boost vaccination. Furthermore, hamsters were monitored for appetite, activity, and weight loss. The hamsters were also challenged with SARS-CoV-2 and orally swabbed days two and four post-infection (PI) to study the lung tissue and blood.
Thereafter, viral RNA quantification was conducted using the swabs followed by infectious virus titration using the tissue-culture infectious dose 50 assay (TCID50), plaque reduction neutralization tests (PRNTs), and histology and immunohistochemistry studies.
Study findings
The results of the current study indicated that in A.1 vaccinated mice, neutralization activity was similar against B.1 challenge virus, while 4.4-fold and 14.7 folds reduction in neutralization activity were observed against B.1.1.7 and B.1.351 challenge virus, respectively. In B.1.1.7 vaccinated mice, an 11.3-fold reduction in neutralization activity was observed against the B.1.351 variant. In B.1.351 vaccinated mice, 12.3 folds and a four-fold reduction in neutralization activity were observed against the B.1.1.7 and A.1 challenge virus, respectively.
In hamsters, it was observed that the neutralizing antibody (nAb) titers ranged from 1:320 to 1:10,240 against homologous viruses while 2- to 14-fold reductions were observed against heterologous viruses. Furthermore, 13- to 14-fold reductions in nAb titers were observed in hamsters that were immunized with A.1- or B.1.1.7-specific vaccines, respectively.
A 4-5 fold reduction in neutralization activity was observed against the A.1 or B.1.1.7 strains, respectively, when vaccinated with the B.1.351-specific vaccine. For the Delta variant, a 4- to 6-fold reduction in serum neutralization titers was observed in A.1 and B.1.1.7 spike vaccinated hamsters, respectively, while a 10-fold reduction was observed with the B.1.351 spike.
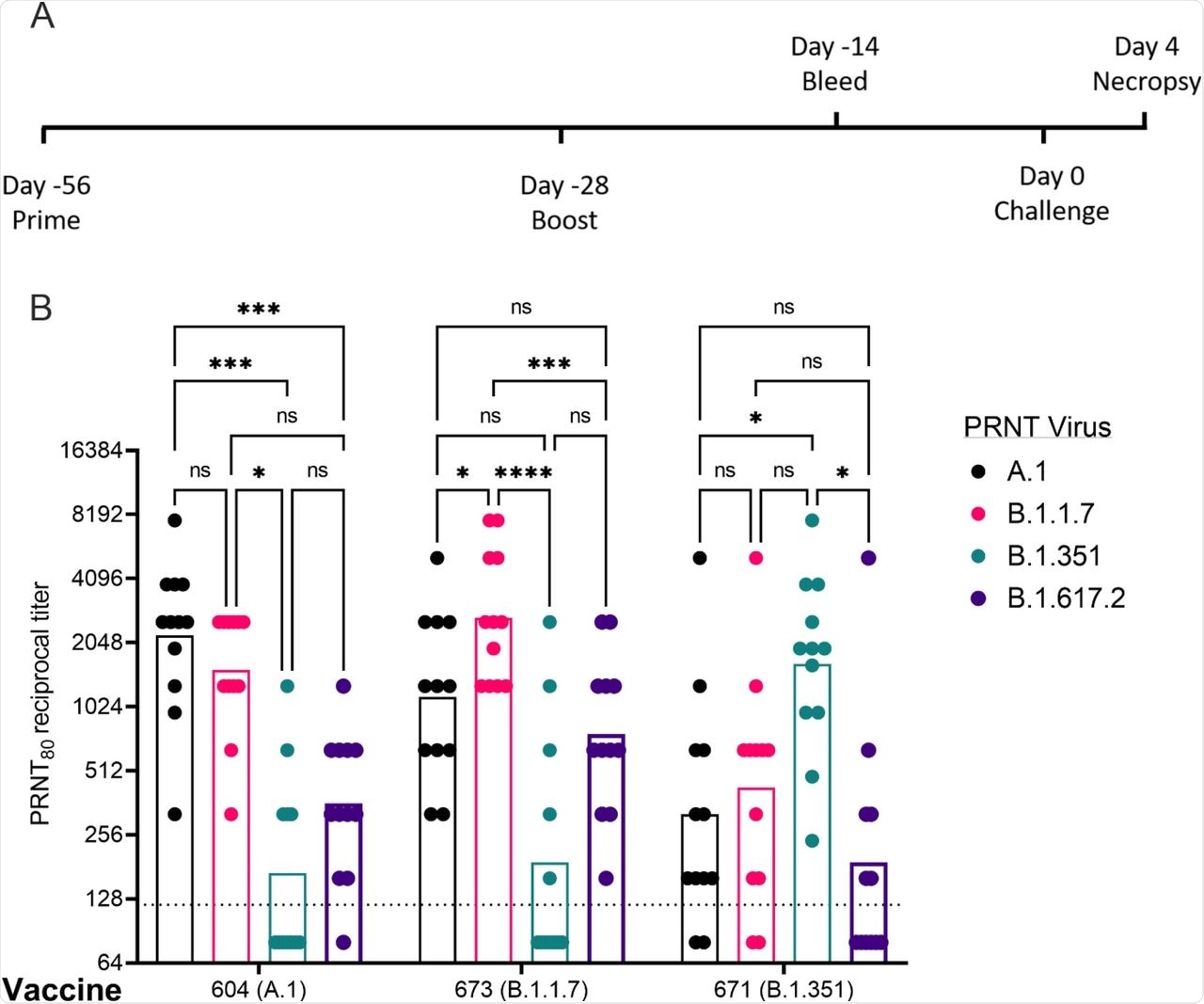
Post-boost neutralizing antibody responses in Syrian golden hamsters. Sera from animals immunized with LION complexed with repRNA vaccine variants A.1, B.1.1.7, or B.1.351 were incubated with live virus of variant A.1 (black), B.1.1.7 (pink), B.1.351 (green), or B.1.617.2 (purple) as indicated. Plaque-reduction neutralizing titers are indicated by individual symbols with geometric mean titers represented by the height of the bars. Indicated statistical comparisons performed using a two-way ANOVA with Tukey’s multiple comparisons test. ns P > 0.05, * P < 0.05, *** P < 0.001, **** P < 0.0001.
All three repRNAs led to the reduction in viral shedding at the oral cavity at day two post-injection, while A.1 and B.1.1.7 repRNAs reduced viral shedding at day 4 post-injection. No viral shedding was reported with the B.1.351 repRNA-vaccinated animals at days two and four post-injection.
Measurement of subgenomic RNA suggested that vaccination with any of the repRNAs led to a significant reduction of SARS-CoV-2 burden in the lungs. Evaluation of lung pathology determined the development of more severe lesions in hamsters challenged with the A.1 or B.1.1.7 variants as compared to the B.1.351 variant.
Both bronchiolitis and vasculitis were observed in most hamsters. However, those receiving the repRNAs did not show any lesions and had reduced viral antigen levels. Notably, hamsters challenged with either the A.1 or B.1.1.7 variant showed reduced lung-to-body weight ratios as compared to the mock hamsters. Comparatively, for the B.1.351 variant, only those hamsters vaccinated with the A.1 repRNA showed a reduced lung-to-body weight ratio.
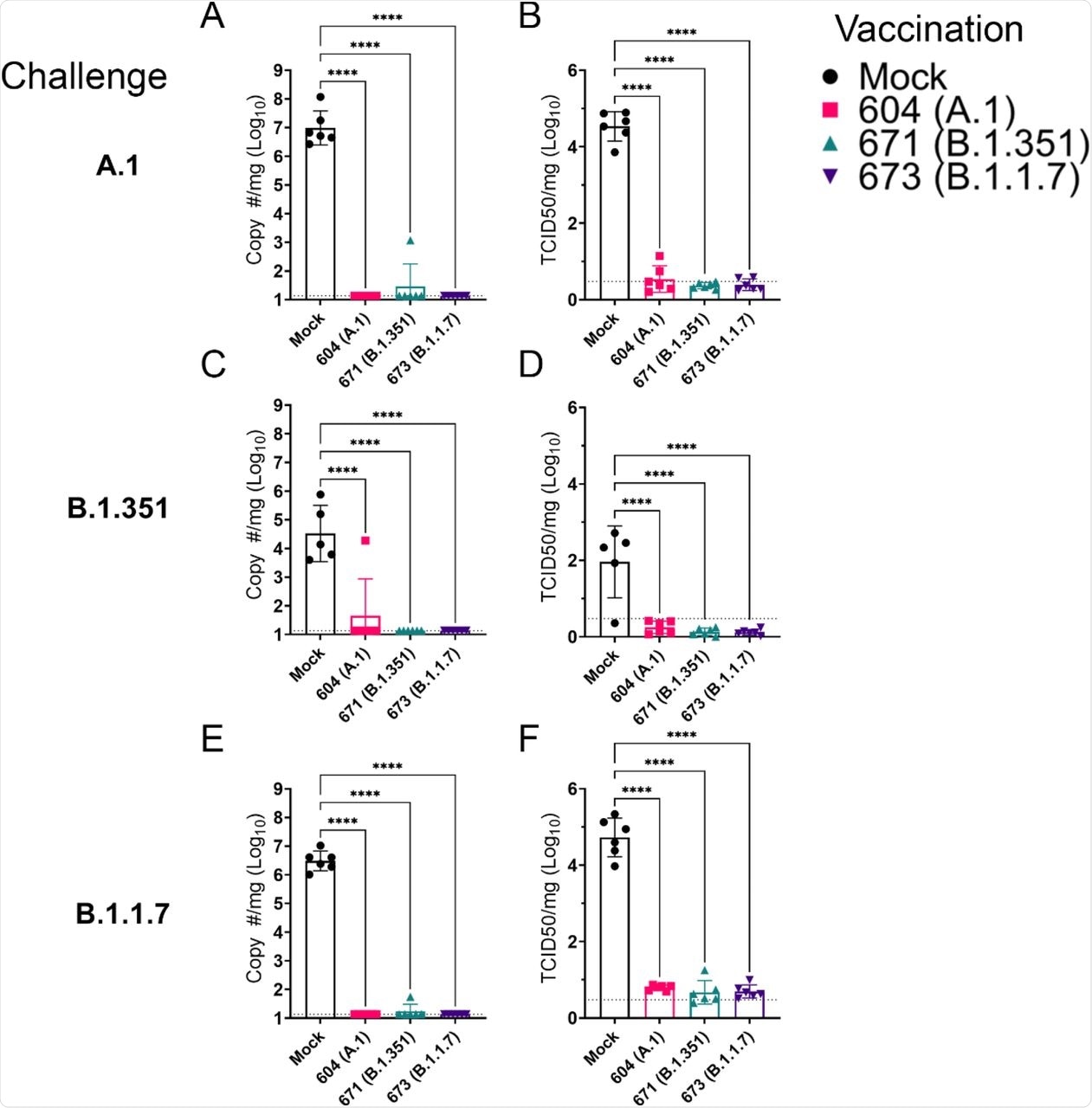
repRNA vaccination significantly reduces the viral burden in the lungs. Mock or repRNA vaccinated hamsters were challenged with 1000 TCID50 of the indicated SARS-CoV2 strains via the IN route. At day 4 PI, hamsters were euthanized, and lung tissue collected. SARS-CoV2 burden in the lung was quantified by qRT-PCR specific for the SgE RNA (A, C, E). Infectious virus in the lungs was quantified by TCID50 assay (B, D, F). Indicated statistical comparisons were performed using a One-way ANOVA with Dunnett’s multiple comparison test against mock-vaccinated hamsters. * P < 0.05, **** P < 0.0001. Comparisons without indicated P-values were non-significant (P > 0.05)
Conclusions
The current study demonstrates that the LION/repRNA vaccines are capable of inducing protective immunity against several SARS-CoV-2 variants. The tested vaccines were capable of reducing the viral burden at both the upper and lower respiratory tract, as well as protecting the lung pathology against the variants. Therefore, the current study supports further development of the LION/repRNA vaccine platform to protect against the current and future SARS-CoV-2 variants.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources