Amongst the uncertainties related to the protection capability of existing severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccines against the Omicron variant, new research has surfaced which examines Immunoglobulin G (IgG) antibodies as well as the neutralizing antibodies (NAb) against the spike protein of SARS-CoV-2 Washington-1 (WA-1, vaccine strain) and the variants of concern (VOCs) including Beta, Delta, and Omicron in a longitudinal cohort.
The team from John Hopkins University, USA, observed little to no neutralizing capability following an initial two-dose messenger RNA (mRNA) vaccination against the Omicron variant. Also, neutralizing capacity to all SARS-CoV-2 variants was almost lost by 8-months post-second dose of primary vaccination. However, neutralizing titers significantly increased for all variants, including Omicron, after the third dose (post-boost) compared to the titers post-two-dose primary vaccination.
A pre-print version of the research paper is available on the medRxiv* server while the article undergoes peer review.
Study: Comparison of total and neutralizing SARS-CoV-2 spike antibodies against omicron and other variants in paired samples after two or three doses of mRNA vaccine. Image Credit: Kateryna Kon/Shutterstock

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Concerns regarding the waning anti-SARS-CoV-2 antibody levels
It is now a well-known fact that the anti-SARS-CoV-2 antibody levels, following primary two-dose vaccination, decline over time. Lower anti-SARS-CoV-2 levels are associated with breakthrough infections, which prompted the Food and Drug Administration (FDA) to approve a booster dose (third dose) for people 12 years and older in the USA. However, apart from the limited reports available on the protective efficacy, the uptake of the booster dose has remained low.
Therefore, the team undertook the current study to investigate the prophylactic potential of booster dose compared to that after the primary two-dose vaccination against SARS-CoV-2.
What did the researchers do?
A seroprevalence study was conducted on a longitudinal cohort of healthcare workers (HWs) from June 2020 to November 2021. Serum samples were longitudinally collected at three time points (TP).
- TP1: within 14-44 days post-second dose of an mRNA SARS-CoV-2 vaccine
- TP2: at least 8 months post-second dose
- TP3: within 14-44 days following an mRNA booster dose
Total antibody responses at the three TPs were measured by enzyme-linked immunosorbent assay (ELISA). To know whether increased antibody levels were associated with improved recognition of SARS-CoV-2 VOCs, neutralizing antibody titers (NT) against the vaccine strain and the Beta, Delta and Omicron VOC were measured in a subset of HWs (n=45), prioritizing the paired samples from TP1 and TP3.
Study findings
A total of 1,353 HWs (median age 41.8 years) contributed serum to at least one of the 3 TP groups. Compared to TP1, the team observed a significant decline in total antibody levels at TP2, which then elevated to much higher levels at TP3. Following a third dose (TP3), 94% of the individuals demonstrated spike IgG assay saturation compared to only 59% post-second dose (TP1).
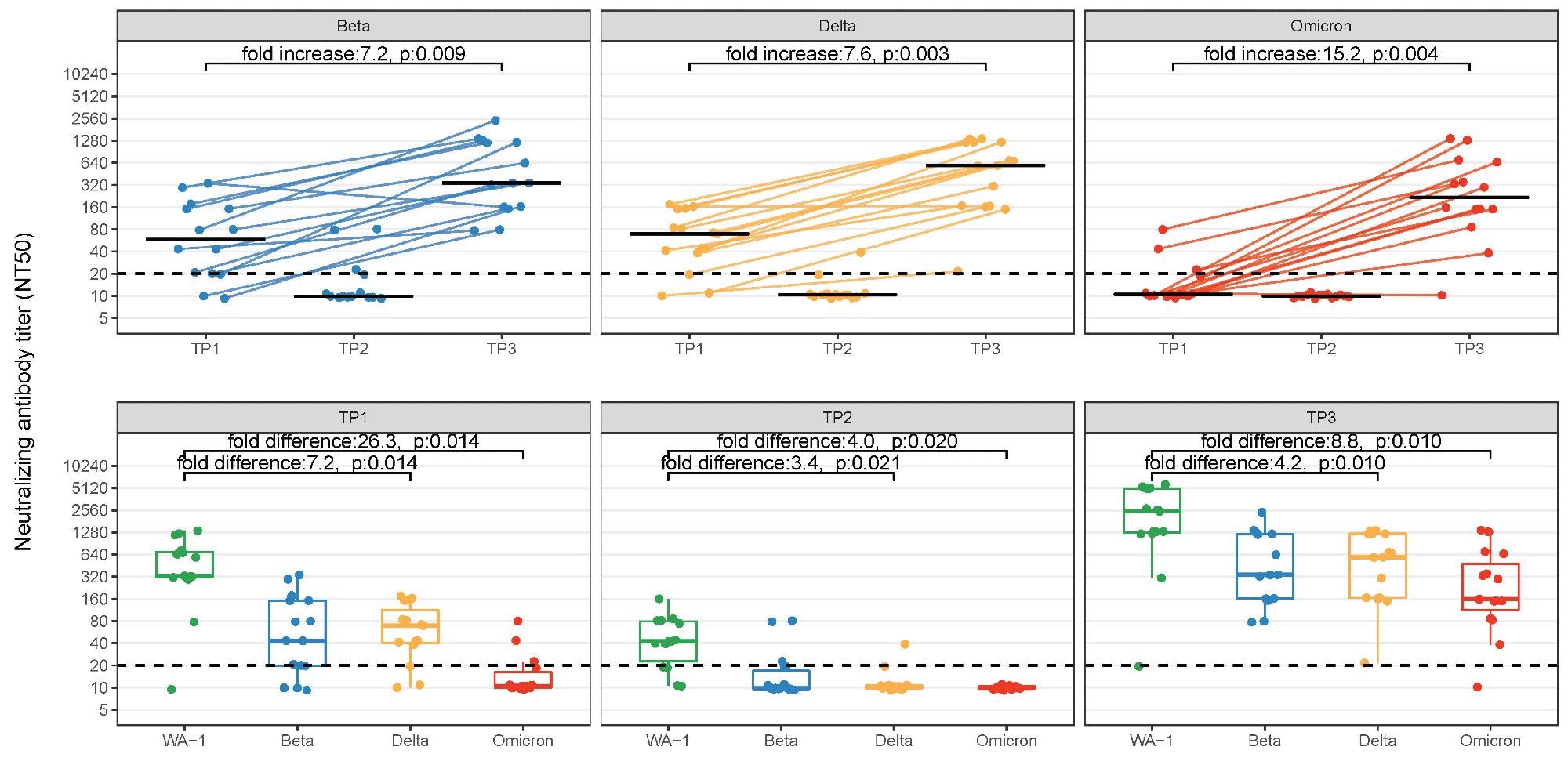 Comparison of neutralizing antibody titers (NT) to SARS-CoV-2 vaccine strain (WA-1), Beta, Delta, and Omicron VOC from healthcare workers with paired serum samples in a longitudinal cohort. Data shown at three time points: within 14-44 days post-dose-2 (Timepoint 1), at least 8 months post-dose-2 (Timepoint 2), and within 14-44 post-booster (Timepoint 3). The top panel shows NT titer for each variant across the three time points with connecting lines illustrating 15 paired samples in Timepoints 1 and 3. Bottom panel shows NT at each timepoint for each VOC. Fold change (increase/difference) represents geometric median fold change. P207 values have been corrected for multiple comparisons using Bonferroni methods.
Comparison of neutralizing antibody titers (NT) to SARS-CoV-2 vaccine strain (WA-1), Beta, Delta, and Omicron VOC from healthcare workers with paired serum samples in a longitudinal cohort. Data shown at three time points: within 14-44 days post-dose-2 (Timepoint 1), at least 8 months post-dose-2 (Timepoint 2), and within 14-44 post-booster (Timepoint 3). The top panel shows NT titer for each variant across the three time points with connecting lines illustrating 15 paired samples in Timepoints 1 and 3. Bottom panel shows NT at each timepoint for each VOC. Fold change (increase/difference) represents geometric median fold change. P207 values have been corrected for multiple comparisons using Bonferroni methods.
Spike IgG antibody profiles were found to correlate with the NTs. However, when compared to WA-1, lower NT activity was observed against all VOC at TP1. By 8-month post-second dose (TP2), total antibody levels had waned with only little NT observed against Beta and Delta, and none against Omicron.
However, the booster dose improved NT activity against all strains, including a >15-fold increase in neutralization against Omicron in paired samples, and thus eliminated the immune escape observed for Omicron following two-dose primary mRNA vaccination. Moreover, following a booster dose, the fold reductions between WA-1 and VOCs were also less than those observed at TP1 among paired samples.
“This study demonstrates that in paired samples an mRNA vaccine booster produces greater quantity and function of spike antibodies and NT as compared to primary SARS-CoV-2 mRNA immunization and was necessary to restore measurable NT to VOC”, concludes the team.

 This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
This news article was a review of a preliminary scientific report that had not undergone peer-review at the time of publication. Since its initial publication, the scientific report has now been peer reviewed and accepted for publication in a Scientific Journal. Links to the preliminary and peer-reviewed reports are available in the Sources section at the bottom of this article. View Sources
Journal references:
- Preliminary scientific report.
Debes A., K., et al. (2022) Comparison of total and neutralizing SARS-CoV-2 spike antibodies against Omicron and other variants in paired samples after two or three doses of mRNA vaccine. medRxiv. doi: https://doi.org/10.1101/2022.01.26.22269819, https://www.medrxiv.org/content/10.1101/2022.01.26.22269819v1
- Peer reviewed and published scientific report.
Debes, Amanda K., Shaoming Xiao, Emily R. Egbert, Patrizio Caturegli, Avinash Gadala, Elizabeth Colantuoni, Ioannis Sitaras, Andrew Pekosz, and Aaron M. Milstone. 2022. “Neutralizing SARS-CoV-2 Spike Antibodies against Omicron in Paired Samples after Two or Three Doses of MRNA Vaccine.” Edited by Daniel R. Perez. Microbiology Spectrum 10 (5). https://doi.org/10.1128/spectrum.02046-22. https://journals.asm.org/doi/10.1128/spectrum.02046-22.