COVID-19 manifests as a mild asymptomatic disease to acute respiratory disease syndrome (ARDS). The condition is often biphasic, with ARDS developing some days after mild symptoms due to pro-inflammatory cytokine release. Hence, it has been speculated that immune response mediated by antibodies might contribute to disease severity. Therefore, early assessment of COVID-19 severity and outcome prediction could help optimize medical care and resources.
 Study: Anti-SARS-CoV-2 Titers Predict the Severity of COVID-19. Image Credit: NIAID
Study: Anti-SARS-CoV-2 Titers Predict the Severity of COVID-19. Image Credit: NIAID
About the study
In the present observational study, researchers prospectively evaluated immunoglobulin G (IgG) response in a cohort of patients hospitalized with COVID-19. All patients hospitalized with acute respiratory symptoms or polymerase chain reaction (PCR)-confirmed COVID-19 were included. Those who acquired nosocomial SARS-CoV-2 infection and patients with a concomitant disease with COVID-19 were excluded.
The patient classification was based on disease severity: a) no oxygen therapy, b) oxygen through masks or nasal prongs, c) high-flow oxygen or non-invasive ventilation, d) mechanical ventilation, e) mechanical ventilation and organ support, and f) death. Patients were stratified into mild, moderate, and severe cases.
Patient-related clinical, demographic, and laboratory data were obtained from electronic health records (EHRs). In addition, serial serum samples were obtained from patients on days 0, 7, and 14 post-inclusion. Two serological tests assessed anti-SARS-CoV-2 IgG in samples: chemiluminescent immunoassay (CLIA) and an in-house Luminex assay.
The target proteins for anti-SARS-CoV-2 antibodies were nucleocapsid (N) protein and S1 monomer. CLIA results were reported as arbitrary units (AU)/ml, and Luminex results as median fluorescent intensity (MFI). Fischer’s exact or chi-square tests were performed for categorical variables and one-way analysis of variance (ANOVA) or Wilcoxon matched-pairs rank test for continuous variables.
Findings
The researchers enrolled 70 patients hospitalized with COVID-19 at Lausanne University Hospital and categorized 19 as mild cases, 29 as moderate cases, and 22 as severe cases. Half of the severe cases were directly admitted to the intensive care unit (ICU). Males outnumbered females in the cohort, and the median age of subjects was 60 years. More moderate cases showed dyslipidemia and obesity, while severe cases frequently showed dyspnea, cough, and radiological infiltrate.
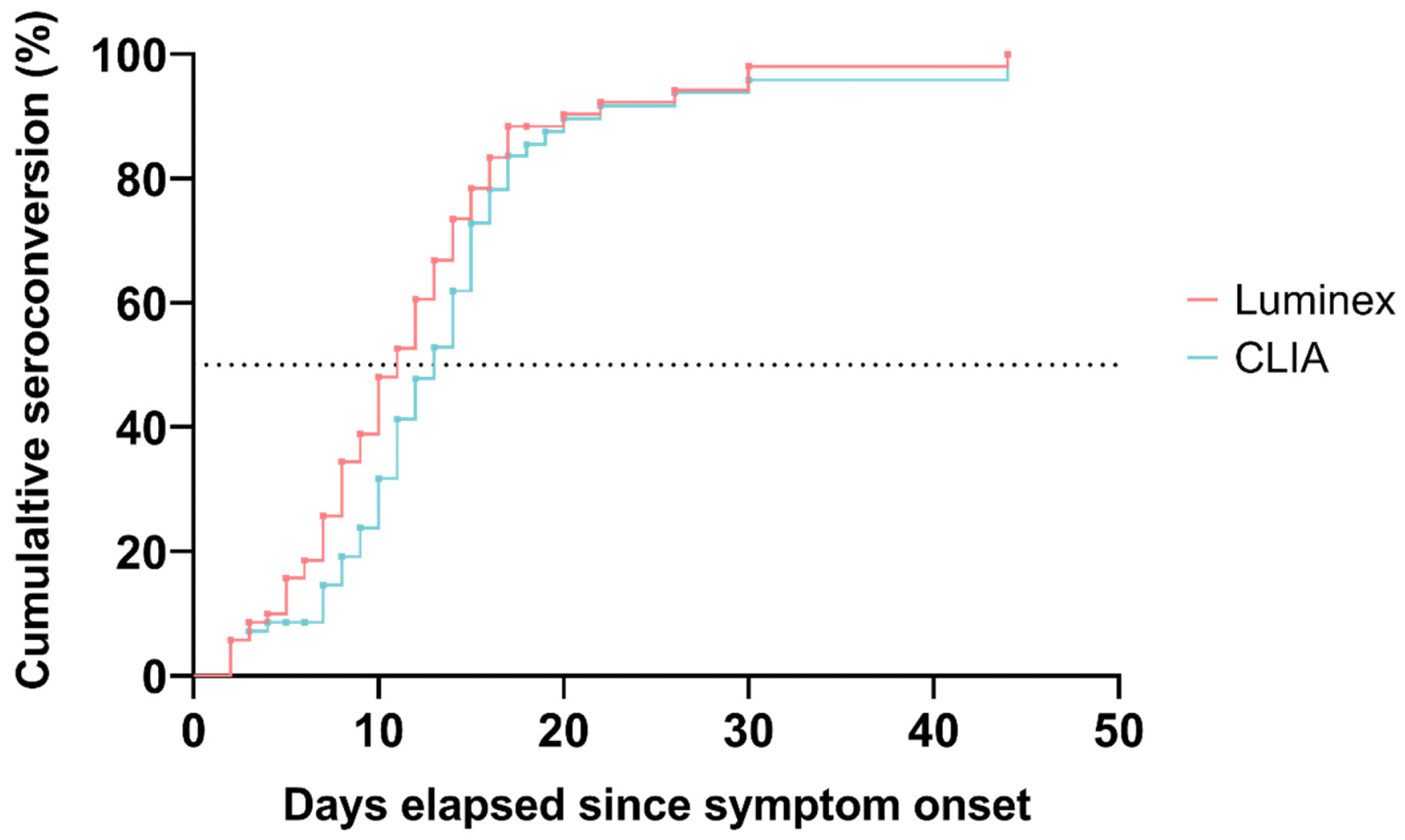 The cumulative proportion of patients who seroconverted according to both serological methods: Luminex (red), CLIA (blue). The dotted line represents the 50% cumulative seroconversion threshold.
The cumulative proportion of patients who seroconverted according to both serological methods: Luminex (red), CLIA (blue). The dotted line represents the 50% cumulative seroconversion threshold.
C-reactive protein and D-dimer levels were higher in severe cases. Severe cases presented symptoms for 13 median days after hospitalization, whereas moderate and mild cases had symptoms for eight and nine median days, respectively. Overall, 133 serum samples were tested with Luminex and CLIA tests. On day 0 (D0), 84% and 63% of patients showed positive results on Luminex and CLIA platforms. At D7, 97% of patients had positive results using either testing method.
Samples from all tested patients on D14 were positive for anti-SARS-CoV-2 antibodies with Luminex tests, while 97% of samples were positive with CLIA. The estimated cumulative seroconversion rate was 25%, 73%, and 98% for Luminex assay, and 14%, 62%, and 96% for CLIA on D7, D14, and D28, respectively. The median time from symptom onset until seroconversion was 11 days for Luminex assay and 13 days for CLIA.
The IgG titers differed significantly according to disease severity. In mild cases, the IgG titers remained relatively stable and low. In contrast, in moderate/severe cases, IgG titers measured using CLIA increased until 14 days (18 days with Luminex) since symptom onset, and high levels sustained after that.
The authors plotted a receiver operating characteristic (ROC) curve to assess the performance of serologic tests on D0 to predict the need for invasive ventilation. The ROC area under the curve (AUC) was comparable for two tests (0.78 for Luminex and 0.79 for CLIA). The optimal prediction was observed for CLIA IgG titers of 50 AU/ml at D0 with an 82% sensitivity, 79% specificity, 63% positive predictive value (PPV), and 91% negative predictive value (NPV).
However, the Luminex assay was also able to predict the need for invasive ventilation. Of the 28 patients with titers above 50.8 AU/ml at D0, eight were already intubated or required immediate intubation, 10 patients required ventilation late during their hospital stay, and the remaining did not require ventilation.
Conclusions
The research team observed that most patients had positive IgG titers at admission and found two distinct patterns of IgG titers over time. Moderate and severe cases had elevated and sustained serologic responses, whereas the titers were low in mild cases. Notably, severe cases had significantly increased IgG titers at admission than moderate/mild cases.
The optimal performance to predict the need to ventilate mechanically was obtained with CLIA tests with a 63% PPV and 91% NPV for the > 50.8 AU/ml cut-off. Moreover, one-third of patients with titers above the cut-off already presented with severe disease at admission and were immediately transferred to ICUs and ventilated invasively. These findings suggested that determining IgG titers at admission could help identify patients at risk of severe outcomes.
Journal reference:
- Anti‐SARS‐CoV‐2 Titers Predict the Severity of COVID‐19. Kritikos, A.; Gabellon, S.; Pagani, J.L.; Monti, M.; Bochud, P.Y.; Manuel, O.; Coste, A.; Greub, G.; Perreau, M.; Pantaleo, G.; et al. Viruses, 2022. DOI: https://doi.org/10.3390/v14051089, https://www.mdpi.com/1999-4915/14/5/1089